Global Surgical Kits Market - Key Trends & Drivers Summarized
Surgical kits are pre-packaged sets of medical instruments and supplies designed to facilitate various types of surgical procedures. These kits are meticulously assembled to include everything a surgical team might need, from scalpels, forceps, and sutures to drapes, gloves, and antiseptics. The components are often organized in a specific order to match the procedural steps, ensuring that the surgical team can work efficiently and effectively without interruption. The standardization and convenience provided by surgical kits significantly reduce preparation time, improve sterility, and enhance overall surgical outcomes. They are used in diverse medical settings, including hospitals, outpatient surgery centers, and emergency rooms, tailored for general surgeries, orthopedic procedures, cardiovascular operations, and more.Technological advancements and the increasing complexity of surgical procedures have driven the evolution of surgical kits. Modern kits are now often customized for specific types of surgeries, reflecting advances in surgical techniques and technologies. For example, minimally invasive surgeries require specialized instruments that are included in these kits. Innovations in sterilization methods, such as gamma radiation and ethylene oxide gas, ensure that all components are free from contaminants. Additionally, the use of high-quality, disposable instruments has become more prevalent, addressing concerns about cross-contamination and infection control. The integration of digital checklists and inventory management systems has also streamlined the process of assembling and distributing these kits, ensuring that they meet the highest standards of quality and safety.
The growth in the surgical kits market is driven by several factors, including the rising number of surgical procedures, increasing focus on infection control, and advancements in surgical technologies. The global increase in elective and emergency surgeries, driven by an aging population and the prevalence of chronic diseases, has significantly boosted demand for surgical kits. The heightened emphasis on preventing hospital-acquired infections has led to greater adoption of sterile, single-use kits. Technological advancements in surgery, such as robotic-assisted procedures and laparoscopic techniques, require highly specialized and reliable instruments, further driving the need for customized surgical kits. Moreover, the growing trend towards outpatient and ambulatory surgeries has increased the demand for portable and comprehensive surgical solutions. Additionally, healthcare providers' focus on cost-efficiency and operational productivity has spurred the adoption of surgical kits that streamline surgical workflows and reduce preparation time.
Report Scope
The report analyzes the Surgical Kits market, presented in terms of market value (USD). The analysis covers the key segments and geographic regions outlined below.- Segments: Type (Disposable, Reusable); Procedure (General Surgery, Cardiac Surgery, Ophthalmology, Orthopedic, Neurosurgery, Gynecology, Other Procedures); End-Use (Hospitals, Specialty Clinics, Other End-Uses).
- Geographic Regions/Countries: World; USA; Canada; Japan; China; Europe; France; Germany; Italy; UK; Spain; Russia; Rest of Europe; Asia-Pacific; Australia; India; South Korea; Rest of Asia-Pacific; Latin America; Argentina; Brazil; Mexico; Rest of Latin America; Middle East; Iran; Israel; Saudi Arabia; UAE; Rest of Middle East; Africa.
Key Insights:
- Market Growth: Understand the significant growth trajectory of the Disposable Surgical Kits segment, which is expected to reach US$22.6 Billion by 2030 with a CAGR of 8.9%. The Reusable Surgical Kits segment is also set to grow at 4.1% CAGR over the analysis period.
- Regional Analysis: Gain insights into the U.S. market, valued at $7.6 Billion in 2024, and China, forecasted to grow at an impressive 10.7% CAGR to reach $2.9 Billion by 2030. Discover growth trends in other key regions, including Japan, Canada, Germany, and the Asia-Pacific.
Why You Should Buy This Report:
- Detailed Market Analysis: Access a thorough analysis of the Global Surgical Kits Market, covering all major geographic regions and market segments.
- Competitive Insights: Get an overview of the competitive landscape, including the market presence of major players across different geographies.
- Future Trends and Drivers: Understand the key trends and drivers shaping the future of the Global Surgical Kits Market.
- Actionable Insights: Benefit from actionable insights that can help you identify new revenue opportunities and make strategic business decisions.
Key Questions Answered:
- How is the Global Surgical Kits Market expected to evolve by 2030?
- What are the main drivers and restraints affecting the market?
- Which market segments will grow the most over the forecast period?
- How will market shares for different regions and segments change by 2030?
- Who are the leading players in the market, and what are their prospects?
Report Features:
- Comprehensive Market Data: Independent analysis of annual sales and market forecasts in US$ Million from 2024 to 2030.
- In-Depth Regional Analysis: Detailed insights into key markets, including the U.S., China, Japan, Canada, Europe, Asia-Pacific, Latin America, Middle East, and Africa.
- Company Profiles: Coverage of players such as Advin Health Care, 3M Company, B. Braun Melsungen AG, Arthrex, Inc., Acumed LLC and more.
- Complimentary Updates: Receive free report updates for one year to keep you informed of the latest market developments.
Some of the 252 companies featured in this Surgical Kits market report include:
- Advin Health Care
- 3M Company
- B. Braun Melsungen AG
- Arthrex, Inc.
- Acumed LLC
- Balton Sp z o.o.
- Bicon LLC
- Aurolab
- AxoGen, Inc.
- 3D Diagnostix, Inc.
- 3DIEMME srl
- ACTEON Group
- Anatomics Pty Ltd.
- Asa Dental S.P.A.
- Beijing Demax Medical Technology Co.,LTD
This edition integrates the latest global trade and economic shifts into comprehensive market analysis. Key updates include:
- Tariff and Trade Impact: Insights into global tariff negotiations across 180+ countries, with analysis of supply chain turbulence, sourcing disruptions, and geographic realignment. Special focus on 2025 as a pivotal year for trade tensions, including updated perspectives on the Trump-era tariffs.
- Adjusted Forecasts and Analytics: Revised global and regional market forecasts through 2030, incorporating tariff effects, economic uncertainty, and structural changes in globalization. Includes historical analysis from 2015 to 2023.
- Strategic Market Dynamics: Evaluation of revised market prospects, regional outlooks, and key economic indicators such as population and urbanization trends.
- Innovation & Technology Trends: Latest developments in product and process innovation, emerging technologies, and key industry drivers shaping the competitive landscape.
- Competitive Intelligence: Updated global market share estimates for 2025, competitive positioning of major players (Strong/Active/Niche/Trivial), and refined focus on leading global brands and core players.
- Expert Insight & Commentary: Strategic analysis from economists, trade experts, and domain specialists to contextualize market shifts and identify emerging opportunities.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Advin Health Care
- 3M Company
- B. Braun Melsungen AG
- Arthrex, Inc.
- Acumed LLC
- Balton Sp z o.o.
- Bicon LLC
- Aurolab
- AxoGen, Inc.
- 3D Diagnostix, Inc.
- 3DIEMME srl
- ACTEON Group
- Anatomics Pty Ltd.
- Asa Dental S.P.A.
- Beijing Demax Medical Technology Co.,LTD
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 919 |
| Published | February 2026 |
| Forecast Period | 2024 - 2030 |
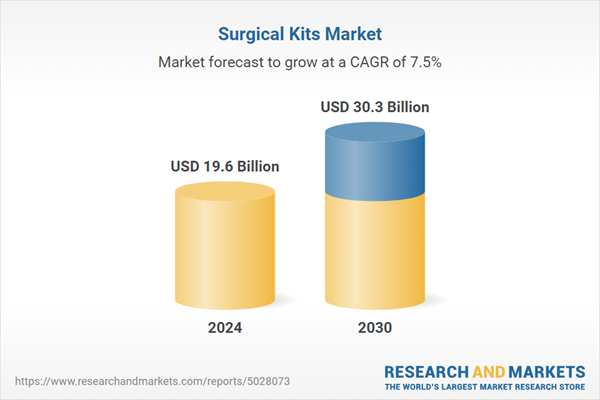
| Estimated Market Value ( USD | $ 19.6 Billion |
| Forecasted Market Value ( USD | $ 30.3 Billion |
| Compound Annual Growth Rate | 7.5% |
| Regions Covered | Global |