Global Clinical Data Management Systems Market - Key Trends & Drivers Summarized
Why Are Clinical Data Management Systems Becoming Indispensable in Modern Clinical Research?
Clinical Data Management Systems (CDMS) have become a cornerstone in modern clinical trials, ensuring the collection, validation, integration, and analysis of clinical data with speed, accuracy, and regulatory compliance. With the increasing complexity of clinical trials, stringent regulatory standards, and the growing importance of real-time data insights, CDMS solutions are no longer auxiliary tools but essential components of clinical research infrastructure. These systems support the entire lifecycle of a clinical trial, from study design and data entry to cleaning and statistical analysis. As pharmaceutical, biotechnology, and contract research organizations (CROs) face mounting pressure to bring therapies to market faster and more efficiently, CDMS offers a structured and auditable platform for managing diverse and voluminous clinical datasets. The rise of multi-site, global studies, and decentralized clinical trials (DCTs) has further intensified the need for robust, centralized systems that can ensure data consistency, traceability, and integrity across geographies. As the clinical research ecosystem evolves, CDMS plays an increasingly vital role in enabling timely decision-making and supporting evidence-based drug development.How Are Advanced Technologies and Platform Integrations Transforming CDMS Capabilities?
The clinical data management landscape is undergoing a significant shift, driven by the integration of advanced technologies and platform interoperability. Traditional CDMS platforms have evolved into sophisticated, cloud-based, AI-powered ecosystems capable of handling complex trial designs and heterogeneous data sources. Artificial Intelligence and Machine Learning algorithms are now being applied to automate data cleaning, identify anomalies, and enhance predictive analytics, thereby reducing human errors and accelerating timelines. Electronic Data Capture (EDC) systems, eSource integrations, wearable devices, ePRO (electronic patient-reported outcomes), and mobile health applications are feeding real-time data into CDMS platforms, creating more dynamic and continuous data streams. Furthermore, seamless integration with other clinical trial technologies - such as CTMS (Clinical Trial Management Systems), eTMF (electronic Trial Master File), randomization tools, and electronic consent platforms - is enabling end-to-end trial data visibility and control. Cloud-based deployment models are enhancing scalability and accessibility, while embedded compliance features such as 21 CFR Part 11, GDPR, and HIPAA readiness are ensuring adherence to global regulatory norms. These innovations are transforming CDMS into an agile, intelligent, and collaborative platform for data-driven clinical research.What Role Do Industry Needs, Regulatory Pressures, and Globalization Play in Market Evolution?
The clinical data management systems market is being shaped by diverse industry-specific needs, evolving regulatory frameworks, and the globalization of clinical trials. Pharmaceutical and biotechnology companies are demanding faster, more accurate data management solutions to cope with increasing trial volumes and shrinking development timelines. CROs, which play a crucial role in outsourced clinical research, are adopting CDMS platforms to streamline workflows, standardize deliverables, and meet client expectations for faster turnaround times and cost efficiency. Regulatory bodies such as the FDA, EMA, and ICH are enforcing stricter guidelines on data integrity, audit trails, and electronic record management, making compliance-ready CDMS platforms a necessity rather than an option. As clinical trials expand into emerging markets across Asia-Pacific, Latin America, and Eastern Europe, the demand for multi-language support, remote data access, and standardized global operations is rising. Moreover, industry standards like CDISC (Clinical Data Interchange Standards Consortium) are influencing system design and data format requirements. The rise of virtual and hybrid trials, accelerated by the COVID-19 pandemic, has further amplified the need for flexible, remote-compatible, and interoperable data management systems that can support decentralized data sources and digital patient interactions.What Are the Key Factors Driving the Accelerated Growth of the CDMS Market?
The growth in the clinical data management systems market is driven by several factors directly tied to technological advancements, end-user demand dynamics, and the evolving nature of clinical research. The increasing adoption of decentralized and hybrid clinical trial models is generating demand for digital platforms that can manage data from diverse, remote, and real-time sources. Technological progress in EDC systems, eSource capture, remote monitoring tools, and AI-powered data analytics is enhancing the efficiency and scalability of CDMS platforms. The growing complexity of clinical trial protocols, including adaptive designs and biomarker-driven studies, is creating a need for more flexible and intelligent data handling systems. Pharmaceutical and biotech companies are prioritizing CDMS solutions to reduce development cycles, lower operational costs, and mitigate compliance risks. The rising outsourcing trend to CROs is expanding the market for CDMS vendors offering customizable and integrated solutions. Additionally, regulatory requirements for data transparency, traceability, and audit readiness are compelling research organizations to upgrade legacy systems. Cloud-based CDMS deployments are further driving adoption due to their ease of access, scalability, and lower IT infrastructure costs. Lastly, the increasing emphasis on real-world data integration, patient-centric trial models, and data-driven decision-making is positioning CDMS as a strategic asset in the global clinical research ecosystem.Report Scope
The report analyzes the Clinical Data Management Systems market, presented in terms of market value (US$). The analysis covers the key segments and geographic regions outlined below:- Segments: Delivery Mode (Licensed Enterprise Solutions, Cloud-Based Solutions, Wed-Hosted Solutions); End-Use (Contract Research Organization End-Use, Medical Device Companies End-Use, Pharma/Biotech Companies End-Use, Other End-Uses).
- Geographic Regions/Countries: World; United States; Canada; Japan; China; Europe (France; Germany; Italy; United Kingdom; Spain; Russia; and Rest of Europe); Asia-Pacific (Australia; India; South Korea; and Rest of Asia-Pacific); Latin America (Argentina; Brazil; Mexico; and Rest of Latin America); Middle East (Iran; Israel; Saudi Arabia; United Arab Emirates; and Rest of Middle East); and Africa.
Key Insights:
- Market Growth: Understand the significant growth trajectory of the Licensed Enterprise Solutions segment, which is expected to reach US$2.8 Billion by 2032 with a CAGR of 12.8%. The Cloud-Based Solutions segment is also set to grow at 10.3% CAGR over the analysis period.
- Regional Analysis: Gain insights into the U.S. market, valued at $679.6 Million in 2025, and China, forecasted to grow at an impressive 15.7% CAGR to reach $1.2 Billion by 2032. Discover growth trends in other key regions, including Japan, Canada, Germany, and the Asia-Pacific.
Why You Should Buy This Report:
- Detailed Market Analysis: Access a thorough analysis of the Global Clinical Data Management Systems Market, covering all major geographic regions and market segments.
- Competitive Insights: Get an overview of the competitive landscape, including the market presence of major players across different geographies.
- Future Trends and Drivers: Understand the key trends and drivers shaping the future of the Global Clinical Data Management Systems Market.
- Actionable Insights: Benefit from actionable insights that can help you identify new revenue opportunities and make strategic business decisions.
Key Questions Answered:
- How is the Global Clinical Data Management Systems Market expected to evolve by 2032?
- What are the main drivers and restraints affecting the market?
- Which market segments will grow the most over the forecast period?
- How will market shares for different regions and segments change by 2032?
- Who are the leading players in the market, and what are their prospects?
Report Features:
- Comprehensive Market Data: Independent analysis of annual sales and market forecasts in US$ Million from 2025 to 2032.
- In-Depth Regional Analysis: Detailed insights into key markets, including the U.S., China, Japan, Canada, Europe, Asia-Pacific, Latin America, Middle East, and Africa.
- Company Profiles: Coverage of players such as Castor EDC, Crucial Data Solutions, Dassault Systemes SE, eClinical Solutions LLC, IBM Corporation and more.
- Complimentary Updates: Receive free report updates for one year to keep you informed of the latest market developments.
Some of the companies featured in this Clinical Data Management Systems market report include:
- Castor EDC
- Crucial Data Solutions
- Dassault Systemes SE
- eClinical Solutions LLC
- IBM Corporation
- IQVIA, Inc.
- LabKey Corporation
- Merative L.P.
- Novatech CRO
- Oracle Corporation
- PAREXEL International Corporation
- Veeva Systems, Inc.
Domain Expert Insights
This market report incorporates insights from domain experts across enterprise, industry, academia, and government sectors. These insights are consolidated from multilingual multimedia sources, including text, voice, and image-based content, to provide comprehensive market intelligence and strategic perspectives. As part of this research study, the publisher tracks and analyzes insights from 43 domain experts. Clients may request access to the network of experts monitored for this report, along with the online expert insights tracker.Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Castor EDC
- Crucial Data Solutions
- Dassault Systemes SE
- eClinical Solutions LLC
- IBM Corporation
- IQVIA, Inc.
- LabKey Corporation
- Merative L.P.
- Novatech CRO
- Oracle Corporation
- PAREXEL International Corporation
- Veeva Systems, Inc.
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 282 |
| Published | May 2026 |
| Forecast Period | 2025 - 2032 |
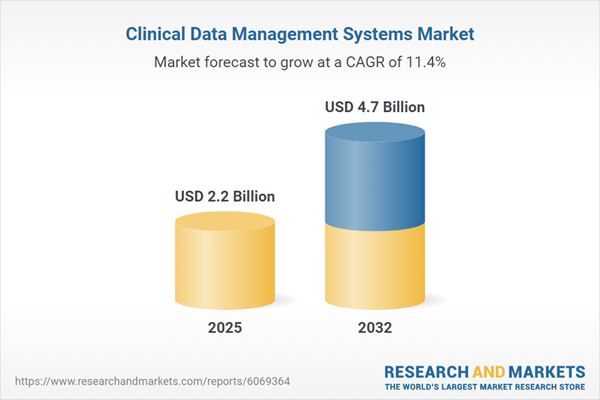
| Estimated Market Value ( USD | $ 2.2 Billion |
| Forecasted Market Value ( USD | $ 4.7 Billion |
| Compound Annual Growth Rate | 11.4% |
| Regions Covered | Global |