COVID-19 impacted the European general surgical devices market during the initial phase as elective and general surgeries were delayed due to lockdowns. Over the previous two years, the market recovery has been led by increased surgical device sales, new product launches, and increased minimally invasive surgeries.
Modern technological innovations, such as minimally invasive surgery equipment and robotic and power-aided systems, are primarily propelling the surgical devices industry. Surgical equipment has wide applications in distinctive surgical techniques, like neurosurgery, plastic and reconstructive surgeries, wound closure, urology, obstetrics and gynecology, thoracic surgery, micro-vascular, cardiovascular, orthopedic surgery, laparoscopy, and others. Furthermore, per the WHO June 2021 update, the percentage of Caesarean sections by 2030 is likely to be 47% in Southern Europe. Additionally, as per the Eurostat 2022 update, most EU Member States have reported significant positive rates of change in recent years for keyhole surgery used to perform hysterectomies, appendectomies, and repairs of inguinal hernias. Furthermore, as per a study published in July 2021 by BMJ journal, musculoskeletal problems accounted for more than 25% of all surgical procedures performed in the United Kingdom by the NHS.
Moreover, according to the report from Brake, a road safety charity organization, 24,530 people were seriously injured in 2021 in the United Kingdom. Thus, with the increase in the number of injuries and accidents, the demand for surgical devices is expected to rise. This is anticipated to impact the growth of the market studied significantly.
Furthermore, rising demand for minimally invasive devices, technological advancements, and product launches are likely to improve the market growth. For instance, in February 2021, Cambridge-based medical device company CMR Surgical (CMR) launched Versius Robotic System in German hospitals. The robotics tool is likely to be used at Klinikum Chemnitz to aid in laparoscopic surgeries.
Hence, as per the factors mentioned above, the European general surgical devices market is expected to witness growth during the forecast period. However, stringent government regulations and poor reimbursement restrain the market growth.
Europe General Surgical Devices Market Trends
Handheld Device is Expected to Witness a Growth in the Europe General Surgical Devices Market During the Forecast Period
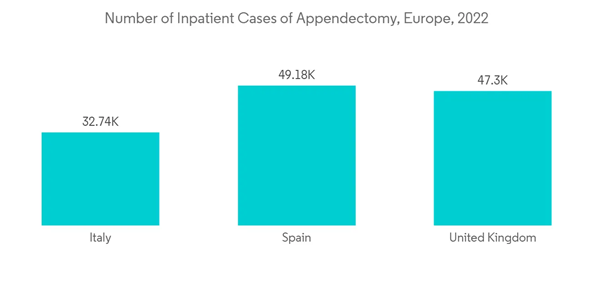
Handheld surgical devices are non-powered, handheld, or hand-manipulated devices such as scalpels, forceps, needle holders, and retractors used in various general surgical operations. Needle holders secure the needle in position for suturing in various tissues. Needle holders are widely used in minimally invasive surgeries as minimally invasive surgery uses smaller surgical incisions, and it is generally less risky than traditional surgery. Hence, the market is anticipated to witness growth in the handheld surgical devices segment due to the rise in minimally invasive surgical procedures.The increasing burden of injuries and accidents leads to minimally invasive surgeries, which is likely to boost segment growth. For instance, as per the 2021 update from Istat, 151,875 accidents occurred in Italy in 2021, in which 204,728 were injured. The same source showed that 561 people were estimated to be injured per day in Italy in 2021. Hence, the high number of injuries is contributing to the market growth. Moreover, increased R&D in handheld devices is likely to boost the market growth. For instance, according to an article published in April 2022 by UPIS journal, a study was conducted to determine the current level of interest in using handheld steerable devices. This study surveyed members of the EAES. The study assessed how steerable devices are now perceived and used in routine practice and what kind of training was provided before using the instruments in laparoscopic surgery. Such studies focusing on the utilization of handheld surgical devices in Europe are expected to drive the growth of the handheld devices segment during the forecast period.
Furthermore, strategic initiatives by key players, such as mergers, acquisitions, and collaboration, are likely to accelerate market growth. In November 2021, Smith+Nephew expanded the reach of its Cori handheld robotic system to Europe. The company's system was made available for use in the United Kingdom's National Health Service.
Hence, as per the abovementioned factors, the handheld surgical devices segment is expected to grow during the forecast period.
The United Kingdom is Likely to Witness a Growth in the Europe General Surgical Devices Market During the Forecast Period
The United Kingdom is expected to hold a significant market share in the European general surgical devices market due to the rising demand for minimally invasive devices and a growing number of surgeries, injuries, and accidents. According to Brake, a road safety charity organization, 24,530 people were seriously injured (KSI) in the United Kingdom in 1,19,850 casualties of all severities in 2021. The need for surgical equipment increases with increasing injuries and accidents. These factors are likely to drive the growth of the market studied.Furthermore, product launches, approvals, and R&D in the country's surgical devices field are likely to boost market growth during the forecast period. In June 2022, Advanced Medical Solutions, a UK-based company focused on tissue-healing technologies, received FDA 510(k) approval for LiquiBand XL. This new device can close longer wounds than existing LiquiBand products. Since its European approval in 2021, the device has been successfully used in various orthopedic procedures in many countries. It is easy to use, effective and reduces pain and scarring compared to other closure methods like stapling.
Moreover, as per the September 2021 news release by the UK government, the implementation of surgical hubs, new technology, and innovative ways of working is likely to help tackle waiting lists and treat around 30% more elective care patients by 2023 to 2024. Surgical hubs, which are already being tested in several cities, including London, are assisting in the speed of several planned surgeries, such as cataract removal, hysterectomies, and hip and knee replacements, and will be expanded across the country. Surgical hubs in existing hospital campuses bring together talents and resources under one roof while lowering infection risk.
Hence considering the above-mentioned factors, the United Kingdom is expected to hold a significant market share in the European general surgical devices market.
Europe General Surgical Devices Market Competitor Analysis
The European general surgical devices market is fragmented due to the presence of many large, medium, and small players. Companies are focusing on new developments and advancements in general surgical devices, which is expected to increase competition in the area further. Existing players are dominating the market by adopting new strategies. Many companies are focusing on mergers, acquisitions, and collaboration to expand their product availability. Some of the major players in the market are B. Braun Melsungen AG, Boston Scientific Corporation, Conmed Corporation, and Johnson & Johnson, among others.Additional benefits of purchasing the report:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- B. Braun Melsungen AG
- Boston Scientific Corporation
- Stryker Corporation
- Olympus Corporation
- Medtronic
- Integer Holdings Corporation
- Conmed Corporation
- Johnson & Johnson