Global GMO Testing Market - Key Trends and Drivers Summarized
Why Is GMO Testing Crucial for Modern Agriculture, Food Safety, and Regulatory Compliance?
GMO testing is becoming increasingly important in modern agriculture, food safety, and regulatory compliance as genetically modified organisms (GMOs) become more prevalent in global food supply chains. But why is GMO testing so essential today? GMO testing refers to the scientific process of identifying and quantifying the presence of genetically modified DNA in crops, food products, animal feed, and other agricultural products. This testing is critical for ensuring that food producers, suppliers, and consumers are aware of the genetic modifications present in the products they are consuming.The rise of genetically modified crops, such as corn, soybeans, and cotton, has revolutionized agriculture by providing plants with traits like herbicide resistance and pest tolerance, leading to higher yields and reduced reliance on chemical pesticides. However, as the use of GMOs increases, so does the demand for accurate testing to ensure food safety, protect consumer rights, and meet the stringent regulations imposed by governments worldwide. Countries have different regulations regarding the labeling and use of GMOs, and many require testing to verify compliance with their laws. Moreover, consumers are increasingly concerned about the environmental and health impacts of GMOs, making GMO testing critical for building trust in food products and ensuring transparency in food labeling.
How Are Technological Advancements Enhancing the Accuracy and Scope of GMO Testing?
Technological advancements are significantly improving the accuracy, speed, and scope of GMO testing, enabling better detection and quantification of genetically modified DNA in various products. One of the most impactful advancements is the development of polymerase chain reaction (PCR) technology, specifically quantitative PCR (qPCR) and digital PCR (dPCR). These techniques allow for highly sensitive and specific detection of even minute amounts of genetically modified DNA in complex samples. qPCR enables rapid screening by amplifying specific DNA sequences associated with genetically modified traits, providing results in a fraction of the time it would take using older methods like enzyme-linked immunosorbent assays (ELISA). Digital PCR (dPCR) offers even greater sensitivity and accuracy, allowing for the precise quantification of GMO content in food or agricultural products.Another important advancement is the use of next-generation sequencing (NGS) technologies. NGS allows for a comprehensive analysis of entire genomes, making it possible to detect both known and unknown genetic modifications in a single test. This is particularly valuable in identifying new or unapproved GMOs that may have been introduced into the food supply. With NGS, researchers can compare the genetic sequence of a product to reference databases, allowing for a broader and more detailed investigation into the presence of GMOs. This technology is expanding the scope of GMO testing beyond simple detection to include detailed genetic profiling, which is essential for regulatory compliance and intellectual property management in the biotech industry.
In addition to laboratory-based testing, portable and field-ready GMO testing kits are becoming more advanced, allowing for on-site testing by farmers, food producers, and regulatory bodies. These portable devices, often based on lateral flow immunoassay technology, can provide rapid results on the presence of specific GMO traits, such as herbicide tolerance or insect resistance. This is particularly useful for farmers who need to verify the genetic status of crops before planting or harvest. These field-ready tests complement laboratory-based methods by providing a quick and cost-effective way to monitor GMO content at various stages of the agricultural supply chain.
Advances in bioinformatics are also playing a key role in improving GMO testing. With the growing volume of genetic data available, bioinformatics tools are essential for analyzing and interpreting the results of GMO tests. These tools help researchers identify and characterize the specific genetic modifications present in a sample, while also comparing them against known databases of approved and unapproved GMOs. By integrating data from multiple testing platforms, bioinformatics enhances the accuracy and reliability of GMO detection, making it easier for regulatory authorities and food producers to meet compliance requirements.
These technological innovations are driving the evolution of GMO testing, ensuring that it keeps pace with the rapid development of new GMOs and the increasing complexity of global food supply chains.
Why Is GMO Testing Critical for Ensuring Transparency, Consumer Choice, and Food Safety?
GMO testing is critical for ensuring transparency, consumer choice, and food safety because it provides clear, scientifically verified information about the presence of genetically modified organisms in food and agricultural products. Transparency in food labeling is becoming increasingly important as consumers demand more information about the origins and composition of the food they eat. Accurate and reliable GMO testing allows food producers to label their products correctly, whether they contain GMOs or are GMO-free, empowering consumers to make informed decisions based on their preferences or dietary restrictions. This is particularly important in regions where GMO labeling is mandatory or where consumers prefer organic or non-GMO products.From a regulatory perspective, GMO testing is essential for compliance with national and international standards. Many countries have strict regulations regarding the import, export, and sale of genetically modified foods, requiring producers to verify that their products meet these standards. For example, the European Union (EU) has stringent GMO labeling and traceability regulations, mandating that food products containing more than 0.9% GMO content must be labeled accordingly. Without accurate GMO testing, it would be impossible for companies to comply with such regulations, leading to potential legal consequences and loss of consumer trust.
Food safety is another key area where GMO testing plays a crucial role. While GMOs themselves are subject to rigorous safety evaluations before they are approved for commercial use, there is still ongoing debate and research into their long-term effects on human health and the environment. Accurate testing ensures that only approved GMOs are present in the food supply and helps monitor for unintended cross-contamination between GMO and non-GMO crops. This is particularly important for protecting vulnerable populations, such as individuals with allergies, who may need to avoid specific genetically modified ingredients. By identifying and quantifying GMOs in food products, testing can help mitigate risks and ensure the safety of the global food supply.
In the agricultural sector, GMO testing is also vital for maintaining the integrity of seed production and preventing the unintentional spread of genetically modified crops. Farmers who wish to grow organic or non-GMO crops must ensure that their seeds and fields are free from genetically modified contaminants. Testing helps verify that seeds are compliant with certification standards, protecting the organic food industry and ensuring that farmers can meet consumer demand for non-GMO products.
Beyond consumer choice and safety, GMO testing is critical for protecting intellectual property in the biotech industry. Many genetically modified crops are patented by the companies that develop them, and testing is necessary to ensure that these patented traits are not being used without authorization. This helps safeguard the intellectual property rights of biotech companies and ensures that farmers and food producers are complying with licensing agreements related to the use of GMO technology.
What Factors Are Driving the Growth of the GMO Testing Market?
Several key factors are driving the rapid growth of the GMO testing market, including the increasing demand for transparency in food labeling, the expansion of global regulations on GMOs, the rise of consumer preference for non-GMO and organic products, and advancements in testing technologies. First, the growing consumer demand for transparency in food labeling is a major driver of the GMO testing market. As more consumers seek information about the ingredients in their food and how it is produced, food companies are under pressure to provide clear and accurate labeling. This has led to an increased need for GMO testing, as companies must verify whether their products contain genetically modified ingredients to meet labeling requirements.Second, the expansion of global regulations on GMOs is fueling demand for testing services. Many countries, particularly in Europe and Asia, have implemented strict regulations regarding the sale and labeling of genetically modified foods. These regulations require rigorous testing to ensure compliance, driving the need for more advanced and reliable GMO testing methods. Additionally, the international trade of agricultural products often requires GMO certification, as different countries have varying levels of tolerance for GMO content in imported goods. As trade grows and regulatory frameworks become more complex, the need for accurate testing continues to rise.
Third, the increasing consumer preference for non-GMO and organic products is contributing to the growth of the GMO testing market. Organic food sales have surged worldwide, driven by consumer concerns about the potential health and environmental impacts of GMOs. To meet the growing demand for organic and non-GMO products, food producers must ensure that their supply chains are free from genetically modified contamination. This requires regular testing at multiple stages of production, from seed selection to final product distribution, to ensure that non-GMO claims are substantiated.
Advancements in GMO testing technologies, such as qPCR, dPCR, and next-generation sequencing, are also driving market growth by making testing more accurate, efficient, and accessible. As testing technologies become more advanced, the costs of testing have decreased, making it more affordable for small and medium-sized food producers to invest in GMO testing. Additionally, the rise of portable testing kits and field-ready devices has made it easier for farmers and food producers to test for GMOs at various points in the supply chain, further driving the adoption of testing solutions.
The increasing focus on food safety is another key factor driving the growth of the GMO testing market. With global food supply chains becoming more complex and interconnected, the risk of cross-contamination between GMO and non-GMO crops is higher. GMO testing is essential for maintaining the integrity of non-GMO and organic products, ensuring compliance with food safety standards, and preventing unintended GMO contamination in the food supply.
In conclusion, the growth of the GMO testing market is driven by rising consumer demand for transparency in food labeling, the expansion of global GMO regulations, increasing preference for non-GMO and organic products, and technological advancements in testing methods. As industries across the food and agriculture sectors prioritize food safety, regulatory compliance, and consumer trust, GMO testing will continue to play a central role in ensuring the integrity of food products and supporting the development of sustainable agricultural practices.
Report Scope
The report analyzes the GMO Testing market, presented in terms of market value (US$). The analysis covers the key segments and geographic regions outlined below:- Segments: Trait (Stacked, Herbicide Tolerance, Insect Resistance); Technology (Polymerase Chain Reaction (PCR), Immunoassay); Food Type (Crop, Processed Food).
- Geographic Regions/Countries: World; United States; Canada; Japan; China; Europe (France; Germany; Italy; United Kingdom; Spain; Russia; and Rest of Europe); Asia-Pacific (Australia; India; South Korea; and Rest of Asia-Pacific); Latin America (Argentina; Brazil; Mexico; and Rest of Latin America); Middle East (Iran; Israel; Saudi Arabia; United Arab Emirates; and Rest of Middle East); and Africa.
Key Insights:
- Market Growth: Understand the significant growth trajectory of the Polymerase Chain Reaction Technology segment, which is expected to reach US$1.8 Billion by 2032 with a CAGR of 5.3%. The Immunoassay Technology segment is also set to grow at 4.8% CAGR over the analysis period.
- Regional Analysis: Gain insights into the U.S. market, valued at $1.0 Billion in 2025, and China, forecasted to grow at an impressive 8.3% CAGR to reach $1.1 Billion by 2032. Discover growth trends in other key regions, including Japan, Canada, Germany, and the Asia-Pacific.
Why You Should Buy This Report:
- Detailed Market Analysis: Access a thorough analysis of the Global GMO Testing Market, covering all major geographic regions and market segments.
- Competitive Insights: Get an overview of the competitive landscape, including the market presence of major players across different geographies.
- Future Trends and Drivers: Understand the key trends and drivers shaping the future of the Global GMO Testing Market.
- Actionable Insights: Benefit from actionable insights that can help you identify new revenue opportunities and make strategic business decisions.
Key Questions Answered:
- How is the Global GMO Testing Market expected to evolve by 2032?
- What are the main drivers and restraints affecting the market?
- Which market segments will grow the most over the forecast period?
- How will market shares for different regions and segments change by 2032?
- Who are the leading players in the market, and what are their prospects?
Report Features:
- Comprehensive Market Data: Independent analysis of annual sales and market forecasts in US$ Million from 2025 to 2032.
- In-Depth Regional Analysis: Detailed insights into key markets, including the U.S., China, Japan, Canada, Europe, Asia-Pacific, Latin America, Middle East, and Africa.
- Company Profiles: Coverage of players such as ALS Ltd., AsureQuality, Bio-Rad Laboratories, Inc., Bureau Veritas SA, Eurofins Scientific SE and more.
- Complimentary Updates: Receive free report updates for one year to keep you informed of the latest market developments.
Some of the companies featured in this GMO Testing market report include:
- ALS Ltd.
- AsureQuality
- Bio-Rad Laboratories, Inc.
- Bureau Veritas SA
- Eurofins Scientific SE
- Intertek Group PLC
- Merieux NutriSciences Corporation
- Microbac Laboratories, Inc.
- SGS SA
- Thermo Fisher Scientific, Inc.
Domain Expert Insights
This market report incorporates insights from domain experts across enterprise, industry, academia, and government sectors. These insights are consolidated from multilingual multimedia sources, including text, voice, and image-based content, to provide comprehensive market intelligence and strategic perspectives. As part of this research study, the publisher tracks and analyzes insights from 1,313 domain experts. Clients may request access to the network of experts monitored for this report, along with the online expert insights tracker.Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- ALS Ltd.
- AsureQuality
- Bio-Rad Laboratories, Inc.
- Bureau Veritas SA
- Eurofins Scientific SE
- Intertek Group PLC
- Merieux NutriSciences Corporation
- Microbac Laboratories, Inc.
- SGS SA
- Thermo Fisher Scientific, Inc.
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 400 |
| Published | June 2026 |
| Forecast Period | 2025 - 2032 |
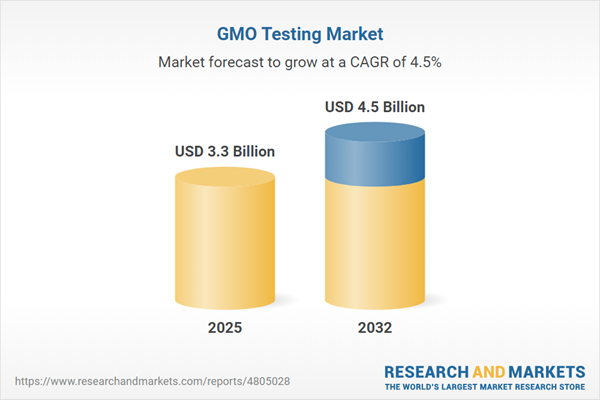
| Estimated Market Value ( USD | $ 3.3 Billion |
| Forecasted Market Value ( USD | $ 4.5 Billion |
| Compound Annual Growth Rate | 4.5% |
| Regions Covered | Global |