The COVID-19 pandemic is expected to have a significant impact on the growth of the studied market. The coronavirus enters the cell when viral spike protein binds to the ACE2 protein present on the surface of human cells. Immunity among the COVID-19 recovered patients is predicted to develop due to the presence of neutralizing antibodies that block this binding. Hence, researchers study the protein interactions and action of neutralizing antibodies. For instance, as per the research study published in Science Daily in December 2020, titled 'Plasma From Recovered COVID-19 Patients Inhibits Spike Protein Binding to ACE2 in a Microsphere-Based Inhibition Assay', researchers applied immunoprecipitation associated with flow cytometry (IP-FCM) in studying the interactions of proteins in SARS-CoV2 infection and the role of antibodies in blocking the virus from binding to cells. Thus, given the ongoing pandemic and increasing research activities, the studied market is expected to be significantly impacted by the current crisis.
The immunoprecipitation testing market is increasing due to the prevalence of autoimmune disorders, which require antibody-antigen interaction testing. Additionally, the growing opportunities in funding and investments from the government and non-government organizations toward the research on next-generation DNA sequencing and genomics are fueling the market growth. In February 2021, the National Institute of Allergy and Infectious Diseases (NIAID) initiated a Phase 1 clinical study to evaluate the safety and immunogenicity of an Epstein-Barr Virus (EBV) gp350-Ferritin Nanoparticle Vaccine, in which the secondary immunogenicity endpoints are measured using luciferase immunoprecipitation assay.
The growing applications of immunoprecipitation testing in central nervous disorders, cancers, and cardiovascular diseases are also boosting the growth of the immunoprecipitation testing market. The tumor cells resistant to drugs exhibit dynamic protein-protein interactions; immunoprecipitation testing aids in studying these interactions among cancer patients. According to the Globocan 2020 fact sheet, an estimated 19,292,789 new cancer cases were diagnosed worldwide, with nearly 9,958,133 deaths from cancer globally. According to estimates from the International Agency for Research on Cancer (IARC), by 2040, the global burden of cancers is expected to grow to 27.5 million new cancer cases and 16.3 million deaths worldwide. Thus, the growing burden of cancers is also expected to drive market growth.
Additionally, in May 2019, Merck & Co. Inc. established a new life science facility that is majorly aimed at research and development activities related to oncology, immunology, and immuno-oncology.
However, limitations in sample size and variations in lipid content might hamper the wide application of this test, which is expected to impede the market growth.
Immunoprecipitation Testing Market Trends
Chromatin Immunoprecipitation (ChIP) is Expected to Dominate the Market by Type Over the Forecast Period
Chromatin immunoprecipitation (ChIP) is a type of immunoprecipitation technique used to investigate the regions of the genome associated with target DNA-binding protein, or to identify the specific proteins associated with a specific region of the genome. Chromatin immunoprecipitation is commonly used in epigenetics research.ChIP is expected to experience significant growth owing to technological advances. For instance, the Chromatrap technology enables data accuracy and delivers quick results without any pipetting risks, allowing researchers to simultaneously analyze multiple antibodies and gene targets.
In the current COVID-19 pandemic, ChIP plays a vital role in determining the protein-DNA interactions, which is very important in understanding the target binding sequences for the SARS-Cov2 Virus. Thus, as the research progresses, the studied segment is expected to witness significant growth during the pandemic.
Additionally, the key players are involved in research activities to develop improved protocols for chromatin immunoprecipitation, which aids in the growth of the studied market. For instance, in the study published in Bio-Protocol in December 2020, titled 'Chromatin Immunoprecipitation (ChIP) to Assess Histone Marks in Auxin-treated Arabidopsis thaliana Inflorescence Tissue' chromatin immunoprecipitation, coupled with quantitative PCR (Chip-qPCR), was used in the identification of binding sites of DNA binding proteins and the detection of histone modification on locus-specific areas of the genome. Hence, the extensive application of ChIP in various genomic studies is a major factor driving the studied segment over the forecast period.
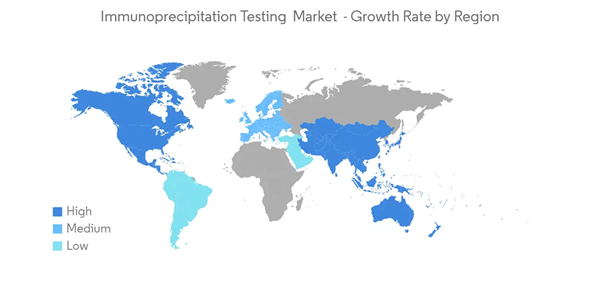
North America Holds the Major Share in Immunoprecipitation Testing Market and is Expected to Dominate the Market Over the Forecast Period
North America holds the major share in the immunoprecipitation testing market owing to the increasing investments and government funding, especially in biotechnology research, and growing awareness regarding personalized medicine. Additionally, growing collaborations and partnerships between organizations and universities and a high concentration of key regional players are expected to drive the studied market in the region. Various government bodies, like NHGRI (National Human Genome Research Institute), have supported the work on sequencing of the human genome and funded the research that involves the study of the genome's structure, function, and role in human health and various disease.In April 2020, GlaxoSmithKline invested USD 250 million in Vir Biotechnologies, a San Francisco-based clinical-stage immunology company, to support its antibody-drug development program for COVID-19. Similarly, in April 2020, the federal government granted USD 14.5 million to Emergent BioSolutions-a Maryland-based biopharmaceutical company-to working on an antibody-drug trial for COVID-19. However, this pandemic has slowed the discovery and development activities of biological products for other applications, such as oncology and rare diseases, which may somewhat hamper growth.
With the growing number of COVID-19 cases in the region, particularly in the United States, extensive research employs advanced tools such as immunoprecipitation sequencing to study the DNA-protein interactions and genome analysis. Additionally, according to the research article published by the National Institutes of Health (NIH) in June 2020, titled 'Increasing Prevalence of Antinuclear Antibodies in the United States' the prevalence of antinuclear antibodies (ANA), which is the common biomarker of autoimmunity, was significantly increasing in the United States over the years. Thus, given the aforementioned factors, the North American region is expected to witness significant growth over the forecast period.
Immunoprecipitation Testing Industry Overview
The immunoprecipitation testing market has rapidly evolved over the last few years. In recent years, the major players in the studied market are expanding their product portfolio through strategies such as product developments, mergers, and acquisitions. Thus, the immunoprecipitation testing market has become a moderately competitive industry. The major players in the studied market include Merck KGaA, Thermo Fisher Scientific, Inc., Bio-rad laboratories, GenScript Biotech Corporation, and Biologics International Corp, among others.Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Thermo Fisher Scientific Inc.
- Abcam Plc
- BioLegend
- Bio-Rad Laboratories
- Biologics International Corp
- Creative Proteomics
- GenScript Biotech Corporation
- Merck KGaA
- Novus Biologicals
- Takara Bio Inc.
- Rockland Immunochemicals, Inc.
- Cell Signalling Technology, Inc.
- Geno Technology , Inc.
- Abbkine Scientific Co., Ltd
- ProteinTech Group
- Labome