Key Highlights
- The COVID-19 pandemic significantly impacted the global healthcare systems, with substantial consequences for COVID-19-infected patients and others, resulting in the cancellation of treatment and surgical procedures to manage and reserve resources; for instance, in September 2021, WHO stated that people who already had neurological conditions were disproportionately affected by COVID-19. In hospitalized COVID-19 patients, chronic neurological conditions were revealed to be independently linked with higher mortality. Routine care interruptions, delayed treatment due to concerns about infectious risks, disruptions in the supply chains for medicines, and the resulting stock-outs had an impact on those with pre-existing neurological problems. The resumed services, treatment, surgical procedures, and increased hospital visits will likely increase the demand for neurology devices.
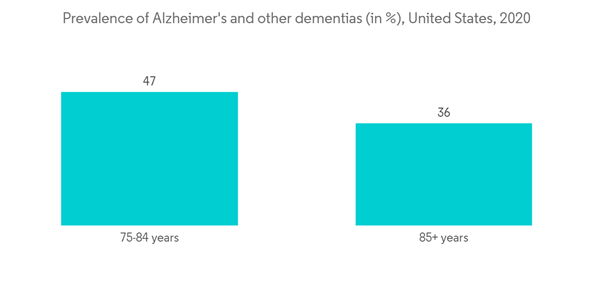
- The rising aging population and increase in the incidence of neurological disorders, huge investments by private players in neurology devices, and growth in R&D in the field of neuro therapies are expected to drive the market studied in North America. For instance, according to a report published by Alzheimer’s Association in March 2022, a vast majority of people who develop Alzheimer’s dementia are aged 65 years or older. An estimated 6.5 million Americans aged 65 and older are living with Alzheimer's dementia. This number is assumed to grow to 13.8 million by 2060, barring the development of medical breakthroughs to prevent, slow, or cure Alzheimer's disease. In the United States, nearly 5.3% of people aged 65 to 74 years, 13.8% of people aged 75 to 84 years, and 34.6% of people aged 85 years or older have Alzheimer’s dementia.
- In addition, the growing geriatric population and the presence of robust healthcare infrastructure for neurological diagnostics and treatment are also contributing to the growth of the neurology devices market over the forecast period. For instance, the World Population Ageing 2021 report states that more than 46 million adults aged 65 and older live in the United States. By 2050, that number is expected to grow to almost 90 million. Similarly, the Mexican population aged 65 is projected to reach 9.31 million by 2030. Thus, the increasing aging population is more prone to develop neurological disorders, which are expected to increase the demand for various neurology devices for diagnosing and treatment of the conditions, which is expected to fuel the market growth in North America.
- Moreover, the rising company initiatives to develop neurology devices in the region contribute to the market's growth. For instance, in January 2021, the United States FDA granted clearance to ClearMind Biomedical’s neuro endoscope, the Axonpen System. This system can illuminate and visualize intracranial tissue and fluids and the controlled tissue and fluid aspiration during surgery. In April 2022, Abbott launched an upgraded version of its digital health app, NeuroSphere myPath. The app's functionality has been enhanced to better assist physicians in monitoring their patients as they utilize Abbott neurostimulation devices to treat their chronic pain. Thus, the North American market is expected to gain traction due to the significant increase in company initiatives to develop neurology devices.
- Therefore, the abovementioned factors are attributed collectively to the studied market growth over the forecast period. However, the high equipment cost, stringent FDA validation, and new device guidelines are expected to hinder market growth over the forecast period.
North America Neurology Devices Market Trends
Cerebrospinal Fluid Management Devices is Expected to Witness High Growth over the Forecast Period
Cerebrospinal fluid (CSF) devices drain a buildup of CSF in the brain (hydrocephalus). Cerebrospinal fluid (CSF) devices treat hydrocephalus, a standard medical procedure and life-saving treatment for many patients. These shunt systems drain excess fluid from the brain to another part of the body, where the fluid is absorbed as part of the circulatory process. If left unchecked, CSF imbalance can lead to elevated intracranial pressure (ICP), leading to various complications. CSF shunts can be used to alleviate these problems in patients who suffer from hydrocephalus. According to the Cleveland Clinic April 2022 report, one to two out of every 1,000 newborns are born with hydrocephalus in the United States. Additionally, according to Hydrocephalus Association 2022 report, hydrocephalus affects over 1 million Americans, ranging from infants and older children to young and middle-aged adults and seniors. As the number of patients increases, the shunt systems are used, and so does the segment's growth.Product launches, approval, and increased focus by several players on their expanding product portfolios in neurosurgery will propel the North American neurology devices market. For instance, in July 2021, Medtronic launched the Percept PC Deep Brain Stimulation (DBS) system. The system integrated with BrainSense technology is made to administer therapeutic stimulation inside and outside the clinic while simultaneously capturing brain signals. The Percept PC device is indicated for individuals with Parkinson's disease, tremors, dystonia, and epilepsy and provides enhanced comfort. An increase in product launches and approval is emerging as a critical trend that is likely to drive the growth of the neurology devices market in North America.
The United States is Expected to Register Significant Growth in the North American Neurology Devices Market
The United States is expected to have a significant share in the neurology devices market owing to the factors such as the rising prevalence of neurological disorders and the rising geriatric population in the country.The increasing burden of neurological disorders such as Alzheimer's disease, epilepsy, Parkinson's disease, neuromuscular disorders, and others is the key factor driving the market's growth. For instance, according to data from Alzheimers.org, 6.2 million Americans aged 65 and older were thought to have Alzheimer's-related dementia in the United States in 2021. Thus, the rising burden of neurological diseases among the United States population is expected to increase the demand for early diagnosis and treatment, further increasing the demand for neurology devices, thereby propelling market growth. Moreover, as per the 2022 report from Cancer.net, approximately 5,900 brain and other CNS tumors are anticipated to be diagnosed in children ages 0 to 19 in the United States. After leukemia, brain and other CNS tumors are the second most common childhood cancers, accounting for about 26% of cancer in children younger than 15 and 21% in teens ages 15 to 19 in the United States.
In addition, the growing geriatric population and the presence of robust healthcare infrastructure for neurological diagnostics and treatment are also contributing to the growth of the neurology devices market over the forecast period. For instance, according to the March 2021 report of the Alzheimer’s Association, approximately 6.2 million people of age 65 years and more are living with Alzheimer’s-related dementia in the United States, and it is projected that this number will increase to 13.8 million people by 2060. Thus, the increasing aging population is more prone to develop neurological disorders. This is expected to increase the demand for various neurology devices for diagnosing and treating the conditions, which is expected to fuel market growth.
Thus, the increasing prevalence of neurological disorders and the growing senior population in the United States is expected to augment the market growth over the forecast period.
North America Neurology Devices Market Competitor Analysis
The North American neurology devices market is consolidated in nature due to the presence of a few companies operating. The competitive landscape includes an analysis of a few North American companies which hold market shares and are well known, including Abbott Laboratories, B. Braun SE, Boston Scientific Corporation, Medtronic PLC, Stryker, and Nihon Kohden Corporation.Additional benefits of purchasing the report:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Abbott Laboratories
- B. Braun SE
- Boston Scientific Corporation
- Johnson and Johnson
- MicroPort Scientific Corporation
- Medtronic PLC
- Nihon Kohden Corporation
- Penumbra Inc.
- Smith & Nephew
- Stryker Corporation