Speak directly to the analyst to clarify any post sales queries you may have.
On the other hand, the COVID-19 pandemic has brought significant challenges to the entire healthcare industry and impacted the growth of the U.S. vital signs monitoring devices market. This has accelerated the adoption of vital signs monitoring devices in the patient population and end-user. Similarly, most people are opting for home precautions with the help of temperature monitoring devices to measure their body temperature amid the pandemic. Also, amid COVID-19, vital signs monitoring devices, such as standalone or wearable devices, have gained significant traction in clinical trial studies over patients and support providers with systematic, real-time information.
Vital signs patient monitors are used to monitor patients' vital signs in emergency care settings and for continuous management of chronic diseases. There is rising demand for various vital signs monitoring devices to treat this increasing patient population. As the demand for these devices has increased, vendors are constantly focusing on increasing their production capacity to meet the increasing demand and provide quality care to patients in the U.S. vital signs monitoring devices market. Further, patients’ vital signs monitors are the most required products, along with portable ventilators and medical consumables for non-invasive and invasive ventilation to treat a broad range of respiratory conditions.
Also, the increased awareness of telemedicine is anticipated to drive the U.S. vital signs monitoring devices market during the forecast period. Further, the increase in demand for technologically advanced products due to benefits over traditional ones is anticipated to fuel the growth of the US vital signs monitoring devices market. Companies are investing in the development of advanced products to improve treatment.
MARKET TRENDS & DRIVERS
Increasing Need for wireless Patient Monitoring at Home
Over the past few years, home patient monitoring has become a standard of care among adult patients who require a long-term alternative to hospital-based care. The trend toward using a wireless patient monitoring system to monitor vital signs continues to grow. Among the many benefits being brought to the healthcare service by wireless patient monitoring is the facility to escalate home care for both chronic and acute conditions. Various pandemic-related factors have created an opportunity to integrate care at home. The emergence of new technologies and capabilities makes taking care of one at home possible.Advances in Remote Patient Monitoring (RPM)
The COVID-19 pandemic has accelerated this trend by highlighting the need and value of RPM. RPM has caught the attention of many medical professionals. More recently, IoT-enabled monitoring devices have added even more functionality to RPM. Physicians using IoT-enabled RPM systems benefit more from their patients. By combining AI with the Internet of Things (IoT) and RPM, healthcare organizations and practitioners can better serve their patients and provide opportunities for improved healthcare outcomes.Upcoming Technologies in Vital Sign Monitoring
IoT technologies have penetrated several healthcare fields, from follow-up to rehabilitation therapies, personal monitoring of daily activities, reminders of medical appointments and medication intakes, and remote monitoring of patient's vital signs. Recently, advances in sensors and mobile devices have led to the development of wearable vital signs monitoring devices that connect with smartphones. This help to analyze the data obtained from people who use them to monitor their health, suggest improvements, and even predict hidden diseases through intelligent algorithms applied to the data sensed from devices such as Bracelets and smartwatches. Through medical IoT technology, physicians or caregivers responsible for patients' health can remotely know, in real-time, the physical condition of people.Rise of Virtual Clinical Trials
In remote and decentralized trials, collecting this data directly from patients at home is critical. As known during the pandemic, the monitoring activity at clinical sites has been strongly impacted and revolutionized. Virtual clinical trials (VCT) rely on technologies such as apps, web-based platforms, wearable devices, and remote monitoring. Decentralized clinical trials (DCTs) are being increasingly implemented and have gained more attention due to the COVID-19 pandemic.Rising Prevalence of Chronic Disorders Augmenting the Need for Continuous Monitoring
Chronic diseases have significant health and economic costs in the U.S. Around 90% of the nation’s USD 4.1 trillion annual healthcare expenditures are for people with chronic and mental health conditions. Seventy-one percent of U.S. healthcare spending goes to treating people with multiple chronic conditions. As the American workforce grows and ages, so do its health needs. Addressing employee health is no longer an option; it’s a priority. As healthcare costs continue to rise, organizations are taking bigger steps to ensure the sustainability of their business and the growing need to manage both people and profit effectively.Chronic disease management and home health initiatives can reduce care costs and promote better patient outcomes. A crucial tool in these programs - remote patient monitoring is helping one academic medical center succeed in these efforts.
Growing Preference for Integrated Vital Signs Monitors
In recent years, it has become much more common to find continuous, low-acuity monitoring at the bedside to proactively manage patients at risk of adverse events in which spot-checks may miss important markers. It is observed that there is a high requirement for integrated vital signs monitors outside hospitals. For instance, in the ambulance, the multi-parameter monitors need to continuously monitor the vital sign of the patient during emergency transport. The increased demand for integrated vital signs monitors with remote monitoring platforms encourages vendors to develop and commercialize such devices catering to end-user requirements.Manufacturers’ Increasing Focus on Developing Software & Apps for Vital Signs Monitoring
Manufacturers are focusing on developing software solutions as there is an increasing focus on automated documentation - that is, the electronic transfer of data from the vital signs monitor to the hospital’s electronic medical record (EMR) system. This can reduce some of the potential for errors resulting from manual documentation of patient data. Some vital signs monitor vendors to provide specialized connectivity solutions/software that accomplish this. At the same time, some of these solutions are specific to the vendor’s vital signs monitors and may be compatible with vital signs monitors from multiple vendors.SEGMENTATION INSIGHTS
INSIGHTS BY PRODUCT
The U.S. vital signs monitoring devices market by product is segmented into standalone and integrated vital signs monitoring devices. The standalone segment accounted for 63.52% in 2022, followed by the integrated segment. Standalone vital signs monitoring devices include blood pressure, body temperature, pulse oximeters, and heart rate/cardiac monitors. These devices measure blood pressure, temperature, saturated oxygen levels, and pulse rates. Standalone vital sign monitors are easy to transfer from one location to another. They are typically lightweight and compact, making it easy to keep tabs on a given patient’s vital signs wherever they are. The standalone segment further comprises heart rate/cardiac monitors, pulse oximeters, blood pressure monitors, and body temperature monitors. Under which the heart rate/cardiac monitors segment accounted for the major market share of 61.93%.Segmentation by Product
- Standalone Vital Sign Monitoring Devices
- Heart Rate/Cardiac Monitors
- Pulse Oximeters
- Blood Pressure Monitors
- Body Temperature Monitors
- Integrated Vital Sign Monitoring Devices
INSIGHTS BY PATIENT GROUP
The geriatric patient group segment accounted for 53.23% of the U.S. vital signs monitoring devices market share and dominated the patient group segment. With the rising elderly population, there is a surge in various chronic disease prevalence rates. Also, it has been reported that the major chronic illnesses that burden the country's aging population include hypertension, high cholesterol, arthritis, diabetes, heart disease, cancer, and dementia. Vital signs of this aging population need to be monitored on time to provide them with high-quality care and avoid fatal conditions, enhancing market growth.The pediatric patient group in the U.S. vital signs monitoring devices market is growing quickly and is likely to grow at a CAGR of 8.10% during the projected period. Infectious diseases have been significantly reduced, and child survival rates for cancer, congenital heart disease, leukemia, etc., have greatly improved. However, over the past few years, there has been a steady increase in chronic health conditions and disabilities in children and adolescents, primarily in four common disorders, including asthma, obesity, psychiatric disorders, and neurodevelopmental disorders. This has increased the demand for vital signs monitoring devices to support these pediatric patient populations and provide them with high-quality care.
Segmentation by Patient Group
- Geriatric Patient Group
- Adult Patient Group
- Pediatric Patient Group
INSIGHTS BY APPLICATION
The U.S. vital signs monitoring devices market is segmented into chronic care and acute care by application, and in 2022, the chronic care segment dominated the segment accounting for 73.62%. Chronic diseases are physical or mental illnesses such as hypertension, diabetes, cardiovascular disease, obesity, and stroke. These diseases represent the most health risks and are responsible for over two-thirds of deaths in the U.S. There is an increasing need to develop vital signs monitoring solutions to manage chronic diseases. Such systems or solutions are used for continuous monitoring, diagnosis, prognosis, and treatment. Hence, they help to reduce healthcare costs and allow patients to go about smoothly with their daily lives while their vital signs are fully monitored.Segmentation by Application
- Chronic Care
- Acute Care
INSIGHTS BY END-USER
The U.S. vital signs monitoring devices market is segmented into hospitals, ambulatory care settings, home care settings & individuals, and others by end-user. The hospital's segment accounted for the largest share of the end-user segment in 2022. Vital signs are essential to monitoring adult or child patients’ progress during hospitalization, as they allow for prompt detection of delayed recovery or adverse events. Vital signs are measured to obtain the basic parameters of a patient's health status. The most common intervention in the hospital is the measurement of vital signs, which traditionally consist of blood pressure, temperature, pulse rate, and respiratory rate.The ambulatory care setting segment in the U.S. vital signs monitoring devices market is growing at a CAGR of 8.57% during the forecast period. Ambulatory care centers are cost-effective and offer quality care with a minimal indirect cost of treatment. The requirement for vital signs monitors, especially integrated ones, is relatively less in ASCs than in hospitals as most procedures are daycare ones, and the patient stay is less. As bulky and fixed integrated patient monitors are relatively less, ambulatory care settings prefer portable and compact vital signs and standalone devices for monitoring patients' vital signs.
Segmentation by End-user
- Hospitals
- Homecare Settings & Individuals
- Ambulatory Care Settings
- Others
VENDOR LANDSCAPE
The U.S. vital signs monitoring devices market is highly competitive and consists of several major players. The industry is fragmented, with various global, local, and regional manufacturers. Owing to continuous technological advancements and product innovations, mid-sized and emerging vendors are increasing their market presence by introducing new products at competitive prices. Also, the market potential drives several players to expand their share through acquisitions. Many U.S. vital signs monitoring devices market vendors are adopting various strategies to increase their presence in many major markets.A&D Company, Baxter, CONTEC MEDICAL SYSTEMS, GE Healthcare, ICU Medical, Koninklijke Philips, Masimo, Medtronic, NIHON KOHDEN, Nonin, OMRON, and OSI Systems are some of the major vendors offering a wide range of both standalone and integrated vital signs monitors in the U.S. vital signs monitoring devices market. However, many local vendors are providing products with similar specifications at lower prices, which has resulted in a price war among vendors. Major vendors continuously compete for the leading position in the market, with occasional competition from other local vendors.
Recent Vendor's Activities in the U.S. vital signs monitoring devices market
- In January 2022, ICU Medical acquired Smiths Medical. The addition of vital care products expanded the business range of ICU Medical with a robust reach.
- In December 2021, Baxter acquired Hillrom, a medical technology leader. Before that, in January 2021, Hillrom acquired Bardy Diagnostics, an innovator in digital health and a leading provider of ambulatory cardiac monitoring technologies.
- In February 2021, Koninklijke Philips completed the acquisition of BioTelemetry. The acquisition of BioTelemetry is a strong fit with Philips’ cardiac care portfolio. The combination results in the business expansion of patient care management solutions for the hospital and homes.
Key Company Profiles
- A&D Company
- Baxter
- CONTEC MEDICAL SYSTEMS
- GE Healthcare
- ICU Medical
- Koninklijke Philips
- Masimo
- Medtronic
- NIHON KOHDEN
- Nonin
- OMRON
- OSI Systems
Other Prominent Vendors
- 100-Plus
- Advanced Instrumentations
- American Diagnostic
- Bittium
- Cardinal Health
- Compass Health Brands
- Drägerwerk AG & Co.
- EDAN Instruments
- FUKUDA DENSHI
- Halma
- Infinium Medical
- Mennen Medical
- Microlife
- NURECA
- Oxitone
- Qardio
- Rossmax International
- Solaris Medical Technology
- Sunset Healthcare Solutions
- Viatom Technology
- VitalConnect
- VivaLNK
- VYAIRE MEDICAL
KEY QUESTIONS ANSWERED
- How big is the U.S. vital signs monitoring devices market?
- What is the growth rate of the U.S. vital signs monitoring devices market?
- What are the growing trends in the U.S. vital signs monitoring devices market?
- Which product segment dominates the U.S. vital signs monitoring devices market?
- Who are the key players in the U.S. vital signs monitoring devices market?
Table of Contents
1 Research Methodology2 Research Objectives
3 Research Process
4 Scope & Coverage
4.1 Market Definition
4.1.1 Inclusions
4.1.2 Exclusions
4.1.3 Market Estimation Caveats
4.2 Base Year
4.3 Scope of the Study
4.3.1 Market Segmentation by Product
4.3.2 Market Segmentation by Patient Group
4.3.3 Market Segmentation by Application
4.3.4 Market Segmentation by End-User
4.3.5 Market Segmentation by Geography
5 Report Assumptions & Caveats
5.1 Key Caveats
5.2 Currency Conversion
5.3 Market Derivation
6 Market at a Glance
7 Premium Insights
7.1 Market Overview
8 Introduction
8.1 Overview
8.1.1 Covid-19 Impact
8.1.2 Reimbursement Scenario for Vital Signs Monitors
8.1.3 Wearable Technology in Vital Signs Monitoring Devices
9 Market Opportunities & Trends
9.1 Increasing Demand for Wireless Patient Monitoring at Home
9.2 Rise of Virtual Clinical Trials
9.3 Advances in Remote Patient Monitoring Technology
9.4 Upcoming Technologies in Vital Signs Monitoring
10 Market Growth Enablers
10.1 Increasing Prevalence of Chronic Disorders Augmenting Continuous Monitoring Demand
10.2 Enchancements in Vital Signs Monitors
10.3 Increased Preference for Integrated Vital Signs Monitors
10.4 Increasing Focus on Developing Software & Apps for Vital Signs Monitoring by Manufacturers
11 Market Restraints
11.1 Cybersecurity & Related Limitations With Vital Signs Monitors
11.2 False Alarms & Alarm Fatigue Associated With Vital Signs Monitors
11.3 Intense Competition Coupled With Pricing Pressures
12 Market Landscape
12.1 Market Overview
12.2 Market Size & Forecast
12.2.1 Product Segmentation Insights
12.2.2 Patient Group Segmentation Insights
12.2.3 Application Segmentation Insights
12.2.4 End-User Segmentation Insights
12.3 Five Forces Analysis
12.3.1 Threat of New Entrants
12.3.2 Bargaining Power of Suppliers
12.3.3 Bargaining Power of Buyers
12.3.4 Threat of Substitutes
12.3.5 Threat of Rivalry
13 Product
13.1 Market Snapshot & Growth Engine
13.2 Market Overview
13.3 Standalone Vital Signs Monitors
13.3.1 Market Overview
13.3.2 Market Size & Forecast
13.3.3 Heart Rate/Cardiac Monitors
13.3.4 Pulse Oximeters: Market Size and Forecast
13.3.5 Blood Pressure Monitors: Market Size and Forecast
13.3.6 Body Temperature Monitors
13.4 Integrated Vital Signs Monitors
13.4.1 Market Overview
13.4.2 Market Size & Forecast
14 Patient Group
14.1 Market Snapshot & Growth Engine
14.2 Market Overview
14.3 Geriatric Patient Group
14.3.1 Market Overview
14.3.2 Market Size & Forecast
14.4 Adult Patient Group
14.4.1 Market Overview
14.4.2 Market Size & Forecast
14.5 Pediatric Patient Group
14.5.1 Market Overview
14.5.2 Market Size & Forecast
15 Application
15.1 Market Snapshot & Growth Engine
15.2 Market Overview
15.3 Chronic Care
15.3.1 Market Overview
15.3.2 Market Size & Forecast
15.4 Acute Care
15.4.1 Market Overview
15.4.2 Market Size & Forecast
16 End-User
16.1 Market Snapshot & Growth Engine
16.2 Market Overview
16.2.1 Key Buying Criteria and Buying Behavior Patterns
16.3 Hospitals
16.3.1 Market Overview
16.3.2 Market Size & Forecast
16.4 Ambulatory Care Settings
16.4.1 Market Overview
16.4.2 Market Size & Forecast
16.5 Homecare Settings & Individuals
16.5.1 Market Overview
16.5.2 Market Size & Forecast
16.6 Others
16.6.1 Market Overview
16.6.2 Market Size & Forecast
17 Competitive Landscape
17.1 Competition Overview
17.1.1 Vendors Recent Activities: M&As and Product Launches
17.2 Market Share Analysis
17.2.1 A&D Company
17.2.2 Baxter
17.2.3 Contec Medical Systems
17.2.4 GE Healthcare
17.2.5 Icu Medical
17.2.6 Koninklijke Philips
17.2.7 Masimo
17.2.8 Medtronic
17.2.9 Nihon Kohden
17.2.10 Nonin
17.2.11 Omron
17.2.12 Osi Systems
18 Key Company Profiles
18.1 A&D Company
18.1.1 Business Overview
18.1.2 Product Offerings
18.1.3 Key Strategies
18.1.4 Key Strengths
18.1.5 Key Opportunities
18.2 Baxter
18.2.1 Business Overview
18.2.2 Product Offerings
18.2.3 Key Strategies
18.2.4 Key Strengths
18.2.5 Key Opportunities
18.3 Contec Medical Systems
18.3.1 Business Overview
18.3.2 Product Offerings
18.3.3 Key Strategies
18.3.4 Key Strengths
18.3.5 Key Opportunities
18.4 GE Healthcare
18.4.1 Business Overview
18.4.2 Product Offerings
18.4.3 Key Strategies
18.4.4 Key Strengths
18.4.5 Key Opportunities
18.5 Icu Medical
18.5.1 Business Overview
18.5.2 Product Offerings
18.5.3 Key Strategies
18.5.4 Key Strengths
18.5.5 Key Opportunities
18.6 Koninklijke Philips
18.6.1 Business Overview
18.6.2 Product Offerings
18.6.3 Key Strategies
18.6.4 Key Strengths
18.6.5 Key Opportunities
18.7 Masimo
18.7.1 Business Overview
18.7.2 Product Offerings
18.7.3 Key Strategies
18.7.4 Key Strengths
18.7.5 Key Opportunities
18.8 Medtronic
18.8.1 Business Overview
18.8.2 Product Offerings
18.8.3 Key Strategies
18.8.4 Key Strengths
18.8.5 Key Opportunities
18.9 Nihon Kohden
18.9.1 Business Overview
18.9.2 Product Offerings
18.9.3 Key Strategies
18.9.4 Key Strengths
18.9.5 Key Opportunities
18.10 Nonin
18.10.1 Business Overview
18.10.2 Product Offerings
18.10.3 Key Strategies
18.10.4 Key Strengths
18.10.5 Key Opportunities
18.11 Omron
18.11.1 Business Overview
18.11.2 Product Offerings
18.11.3 Key Strategies
18.11.4 Key Strengths
18.11.5 Key Opportunities
18.12 Osi Systems
18.12.1 Business Overview
18.12.2 Product Offerings
18.12.3 Key Strategies
18.12.4 Key Strengths
18.12.5 Key Opportunities
19 Other Prominent Vendors
19.1 100-Plus
19.1.1 Business Overview
19.1.2 Product Offerings
19.2 Advanced Instrumentations
19.2.1 Business Overview
19.2.2 Product Offerings
19.3 American Diagnostic
19.3.1 Business Overview
19.3.2 Product Offerings
19.4 Bittium
19.4.1 Business Overview
19.4.2 Product Offerings
19.5 Cardinal Health
19.5.1 Business Overview
19.5.2 Product Offerings
19.6 Compass Health Brands
19.6.1 Business Overview
19.6.2 Product Offerings
19.7 Drägerwerk AG & Co.
19.7.1 Business Overview
19.7.2 Product Offerings
19.8 Edan Instruments
19.8.1 Business Overview
19.8.2 Product Offerings
19.9 Fukuda Denshi
19.9.1 Business Overview
19.9.2 Product Offerings
19.1 Halma
19.10.1 Business Overview
19.10.2 Product Offerings
19.11 Infinium Medical
19.11.1 Business Overview
19.11.2 Product Offerings
19.12 Mennen Medical
19.12.1 Business Overview
19.12.2 Product Offerings
19.13 Microlife
19.13.1 Business Overview
19.13.2 Product Offerings
19.14 Nureca
19.14.1 Business Overview
19.14.2 Product Offerings
19.15 Oxitone
19.15.1 Business Overview
19.15.2 Product Offerings
19.16 Qardio
19.16.1 Business Overview
19.16.2 Product Offering
19.17 Rossmax International
19.17.1 Business Overview
19.17.2 Product Offerings
19.18 Solaris Medical Technology
19.18.1 Business Overview
19.18.2 Product Offerings
19.19 Sunset Healthcare Solutions
19.19.1 Business Overview
19.19.2 Product Offerings
19.20 Viatom Technology
19.20.1 Business Overview
19.20.2 Product Offerings
19.21 Vitalconnect
19.21.1 Business Overview
19.21.2 Product Offerings
19.22 Vivalnk
19.22.1 Business Overview
19.22.2 Product Offerings
19.23 Vyaire Medical
19.23.1 Business Overview
19.23.2 Product Offerings
20 Report Summary
20.1 Key Takeaways
20.2 Strategic Recommendations
21 Quantitative Summary
21.1 Market by Product
21.2 Market by Patient Group
21.3 Market by Application
21.4 Market by End-User
22 Appendix
22.1 Abbreviations
Companies Mentioned
- A&D Company
- Baxter
- CONTEC MEDICAL SYSTEMS
- GE Healthcare
- ICU Medical
- Koninklijke Philips
- Masimo
- Medtronic
- NIHON KOHDEN
- Nonin
- OMRON
- OSI Systems
- 100-Plus
- Advanced Instrumentations
- American Diagnostic
- Bittium
- Cardinal Health
- Compass Health Brands
- Drägerwerk AG & Co.
- EDAN Instruments
- FUKUDA DENSHI
- Halma
- Infinium Medical
- Mennen Medical
- Microlife
- NURECA
- Oxitone
- Qardio
- Rossmax International
- Solaris Medical Technology
- Sunset Healthcare Solutions
- Viatom Technology
- VitalConnect
- VivaLNK
- VYAIRE MEDICAL
Methodology
Our research comprises a mix of primary and secondary research. The secondary research sources that are typically referred to include, but are not limited to, company websites, annual reports, financial reports, company pipeline charts, broker reports, investor presentations and SEC filings, journals and conferences, internal proprietary databases, news articles, press releases, and webcasts specific to the companies operating in any given market.
Primary research involves email interactions with the industry participants across major geographies. The participants who typically take part in such a process include, but are not limited to, CEOs, VPs, business development managers, market intelligence managers, and national sales managers. We primarily rely on internal research work and internal databases that we have populated over the years. We cross-verify our secondary research findings with the primary respondents participating in the study.

LOADING...
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 260 |
| Published | February 2023 |
| Forecast Period | 2022 - 2028 |
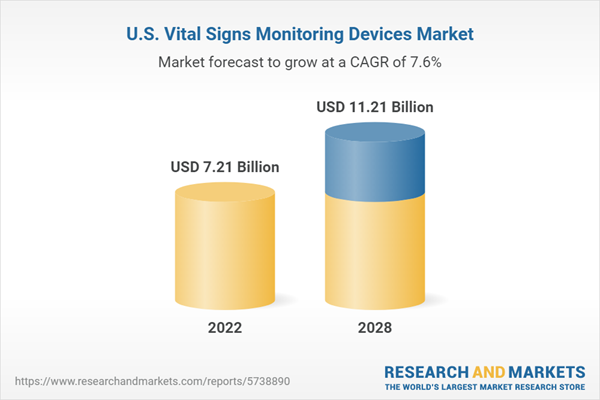
| Estimated Market Value ( USD | $ 7.21 Billion |
| Forecasted Market Value ( USD | $ 11.21 Billion |
| Compound Annual Growth Rate | 7.6% |
| Regions Covered | United States |
| No. of Companies Mentioned | 35 |