Cancer diagnosis and treatment care were impacted by the sudden emergence of COVID-19 infection due to the halt in diagnostic procedures and surgical procedures. Moreover, as per many studies, cancer patients, especially prostate cancer patients, are more susceptible to coronavirus than individuals without cancer, as they are in an immunosuppressive state caused by malignancy and anticancer treatment. Thus, they are majorly suggested to stay at home and avoid all non-emergent diagnoses and treatment. According to the Union for International Cancer Control (UICC) in May 2020, there is a significant decrease in cancer diagnostics rates in the United States during the pandemic. Such decreases in cancer diagnostics are expected to have a short-term decline in demand during the pandemic period. However, the relaxation of the strict regulations is expected to contribute to the reopening of diagnostic clinics and research activities for prostate cancer, thereby contributing to the growth of the market during the post-pandemic period.
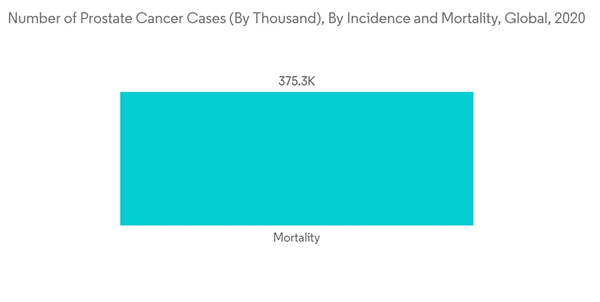
The major factor attributing to the growth of the market studied is an aging population and a high prevalence rate of prostate cancer. According to Globocan 2020, prostate cancer is the second most common type of cancer in the male population. As per the data published by the same source, the prevalence of prostate cancer was found to be 1,414,259, or 7.3% of the total cancer cases worldwide. As prostate cancer is more common in the elderly population, the rising aging population in the world is also expected to propel market growth. For instance, according to the World Population Prospects 2022 published by the United Nations, the share of global population aged 65 years or above is projected to rise from 10% in 2022 to 16% in 2050. Such an increase in the geriatric population who are prone to developing prostate cancer is expected to drive the growth of the market over the forecast period.
Furthermore, the rising product approvals and developments are expected to contribute to the growth of the studied market. For instance,in March 2022, the United States Food and Drug Administration approved Novartis's Pluvicto (lutetium Lu 177 vipivotide tetraxetan) for the treatment of adult patients with a certain type of advanced cancer called prostate-specific membrane antigen-positive metastatic castration-resistant prostate cancer. Therefore, these will lead to increased adoption of prostate cancer therapies, driving market growth.
Hence, owing to all the aforementioned factors, the market is anticipated to propel at high growth rates. However, the high cost of prostate cancer drugs and the low success rate of clinical trials are expected to hinder market growth.
Prostate Cancer Diagnostics & Therapy Market Trends
The Transrectal Ultrasound (TRUS) Segment is Expected to Grow at Rapid Pace Over the Forecast Period
Transrectal ultrasound is expected to hold a significant share of the market over the forecast period. The transrectal ultrasound (TRUS) guided biopsy is also among the common diagnostic procedures performed for prostate cancer, which is still considered a viable option. It is quick and convenient to the provider, with a familiar approach to most urologists and a low risk of urinary retention. It is very useful to observe the abnormal areas of the prostate, size, and shape of the prostate. TRUS is considered a viable option in some cases as an important alternative for the early detection of prostate cancer. For patients who cannot undergo MRI procedures due to claustrophobia, metal implants, or lack of insurance coverage, TRUS is a good option with clinical benefits, thereby contributing to the growth of the studied segment.Furthermore, according to the study published in the Journal of Urology, in February 2022, men who have negative TRUS biopsies have an extremely low prostate cancer-specific death rate, especially if their prostate-specific antigen (PSA) is below 10. This raises major concerns regarding the widespread use of MRI-targeting for the first prostate biopsy and advises that MRI-targeting should be reserved for men whose PSA is greater than 10 ng/ml following a negative biopsy. This will lead to increased adoption of transrectal ultrasound for the detection of prostate cancers, thereby driving this segment's growth.
In addition, the rising product launches are expected to contribute to the growth of the studied segment. For instance, in May 2021, RSIP Vision launched a new prostate MRI-to-ultrasound registration tool that provides MRI-to-ultrasound registration using standard MRI scans and trans-rectal ultrasound images. Such advanced solutions will further drive this segment's growth due to the increased adoption of transrectal ultrasound diagnostics in the detection of prostate cancers.
In several cases, the patients often suffer from associated diseases, where the TRUS biopsy has a considerable edge over other procedures. Thus, it is widely known and accepted. At present, with several new approaches coming up with imaging, the growth of this segment is estimated to be rapid during the forecast period.
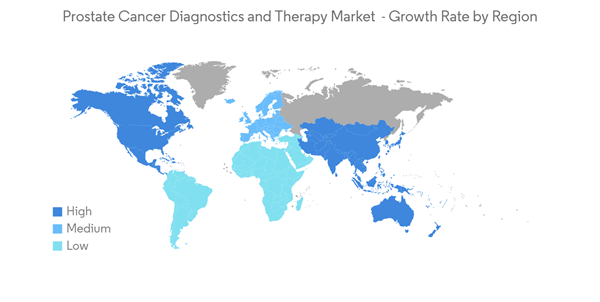
North America is Expected to Retain a Significant Market Share During the Forecast Period
North America is anticipated to hold a significant market share, owing to the factors such as the increasing prevalence of prostate cancer, coupled with the rising awareness and increasing research and development in this region. According to the American Cancer Society, in 2022, an estimated 268,490 new prostate cancer cases was identified were and reported in the United States. In addition according to the same source, about 6 cases in 10 are diagnosed in men who are 65 or older, and it is rare in men under 40. The average age of men at diagnosis is about 66. Such an incidence of prostate cancer is expected to contribute to the growth of the market in this region.The United States Food and Drug Administration is taking steps to enhance the growth of the cancer therapy market by approving drugs, which are in the clinical phase, thus accelerating clinical developments. Companies and research organizations are investing in research and development (R&D), thereby contributing to the growth of the market.
The rising number of prostate cancer cases is creating huge opportunities for market players. The major market players are focusing on R&D activities to bring new and reliable treatments to the market. For instance, in September 2021, the United States Food and Drug Administration authorized the marketing of software to assist medical professionals who examine body tissues (pathologists) in the detection of areas that are suspicious for cancer as an adjunct (supplement) to the review of digitally-scanned slide images from prostate biopsies (tissue removed from the body). Such initiatives will lead to increased adoption of prostate cancer diagnosis and treatment in the United States, thereby driving market growth.
Market players in North America are focusing on various growth strategies such as new product launches, partnerships, mergers, and acquisitions. For instance, in September 2021, Paige received USFDA approval for the de novo marketing of PaigeProsate, a clinical-grade AI solution for prostate cancer detection. In addition, in January 2021, Myovant partnered with Pfizer to commercialize its prostate cancer drug Orgovyx (relugolix), approved in December 2020.
Therefore, owing to the above-mentioned factors, the market is expected to drive in this region over the forecast period.
Prostate Cancer Diagnostics & Therapy Market Competitor Analysis
The market for prostate cancer diagnostics and therapy is highly competitive due to the presence of a number of market players. Several large drug and equipment manufacturers exist within the global prostate cancer diagnostic and therapy market, and their status as major players lies primarily within specific product markets. Some of the key players in the market include Abbott Laboratories, Astellas Inc., AstraZeneca, Bayer HealthCare AG, Cellanyx Diagnostics Inc., Dendreon Corporation, Ferring Pharmaceuticals, Ipsen Group, Sanofi-Aventis, Tolmar Inc, GE Healthcare, Siemens Healthineers, and F. Hoffmann-La Roche AG.Additional benefits of purchasing the report:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Abbvie Inc.
- Astellas Pharma Inc.
- AstraZeneca PLC
- Bayer AG
- F. Hoffmann-La Roche Ltd
- Ipsen SA
- Johnson & Johnson Services Inc.
- Koninklijke Philips NV
- Merck & Co. Inc.
- Pfizer Inc.
- Sanofi-Aventis
- Thermo Fisher Scientific