The COVID-19 pandemic impacted the growth of the sterility testing market. Sterility testing was a key tool for most pharmaceutical and biotechnological companies that started focusing on their research and development for the identification of leads that can diagnose and treat the SARS-CoV-2 infection. The increase in COVID-19 cases drove the major companies and research institutions to look outside of conventional medicine or medicines, which resulted in significant investment in research and development efforts by both private and public entities worldwide. These activities have significantly boosted the sterility testing market. Moreover, the severity and high infection rate of infections and the growing number of drugs and vaccines led to high demand for sterility testing in the pharmaceutical and biopharmaceutical production industry. This has increased the demand for strict sterility testing of the products. With the rising adoption and demand for sterility testing, the studied market is expected to regain its full potential in the next two to three years.
Factors such as the increasing research and development in life sciences, rising number of drug launches, and rise in demand for sterilized products are boosting the market growth. In addition, the rising demand for sterilized products globally and a few potential safety issues connected to the products, which typically result from manufacturing processes and complex biological and structural characteristics of the products, are anticipated to drive the market during the forecast period.
The rising burden of chronic and infectious diseases among the population raises the demand for strict sterility testing of drugs and medical devices to ensure the safety of the products and devices. This is anticipated to fuel the market growth over the forecast period. For instance, according to the British Heart Foundation England Factsheet, published in January 2022, about 6.4 million people were living with cardiovascular diseases in England in 2021. In addition, as per the same source, around 7.4 million people were living with heart and circulatory diseases in the United Kingdom in 2021. Also, as per the data published by WHO, in October 2022, the incidence of tuberculosis was estimated to be around 10.6 million people globally in 2021, of which 1.2 million were children.
Additionally, the increasing number of chronic diseases further increases the demand for effective drugs, which is also expected to propel the need for sterility tests of the drugs before launching them into the market, thereby fuelling the market growth. For instance, in September 2022, the Government of India launched a vaccine against cervical cancer Quadrivalent Human Papillomavirus vaccine (qHPV), which has been developed by the Serum Institute of India (SII) and the Department of Biotechnology (DBT). Also, in September 2022, Bayer launched vericiguat in India to reduce the risk of cardiovascular deaths and repeated hospitalizations.
Furthermore, the increasing research and development in the field of life sciences, coupled with huge public and private funding in the sector, is also contributing to the market growth. For instance, as per the Union Budget for the year 2021-2022, the Indian Ministry of Science and Technology allocated a budget of INR 35 billion, a 25% hike from the 2020-21 budget, for the Department of Biotechnology (DBT) of India, mainly for biotechnology research and development and industrial and entrepreneurship development in the country which will boost the studied market.
Moreover, the growing company activities in adopting various business strategies such as expansion, collaboration, and others are also contributing to the market growth. For instance, in September 2021, Nelson Labs and Sterigenics Germany GmbH, global leaders in comprehensive laboratory testing and sterilization services, announced the opening of a newly expanded center of excellence for microbiological laboratory testing as well as increased sterilization capacity in their Wiesbaden, Germany, facilities.
Therefore, owing to the aforementioned factors, the studied market is expected to grow over the forecast period. However, the stringent regulatory framework is expected to hinder the growth of the sterility testing market over the forecast period.
Sterility Testing Market Trends
Pharmaceutical and Biological Manufacturing Segment Expected to Grow Over the Forecast Period
The pharmaceutical and biological manufacturing segment is expected to witness significant growth in the sterility testing market over the forecast period. The factors attributing to the segment growth are the rising demand for drugs, vaccines, and high-growth pharmaceutical and biological industries.Sterility testing is carried out at all levels of manufacturing pharmaceutical and biological products to minimize the risk of product contamination. As the demand for pharmaceutical and medical products for different health conditions is increasing, the frequency of sterility testing in the industries is also increasing, which is expected to boost the market.
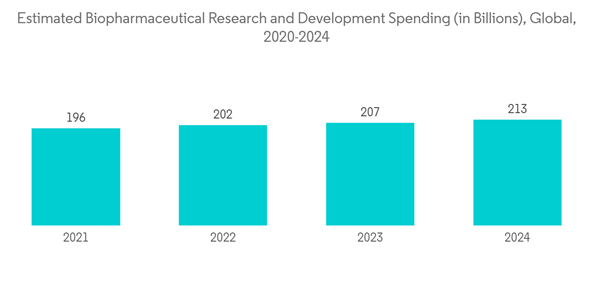
Furthermore, the increase in R&D spending in the biopharmaceutical sector aided in the growth of the market. For instance, according to the data published by IFPMA, in December 2021, the annual R&D spending of the biopharmaceutical industry has been 7.3 times greater than that of the aerospace and defense industries, 6.5 times more than that of the chemicals industry, and 1.5 times more than that of the software and computer services industry. As per the report, in 2022, over 202 billion was estimated to be spent around the world in biopharmaceutical research and development. The large investments in the market are considered a result of the rising acceptance of sterility testing and its related services over the years. This is also expected to increase segment growth over the forecast period.
Therefore, these increasing R&D activities and expenditures in the segment are expected to propel the market growth over the forecast period.
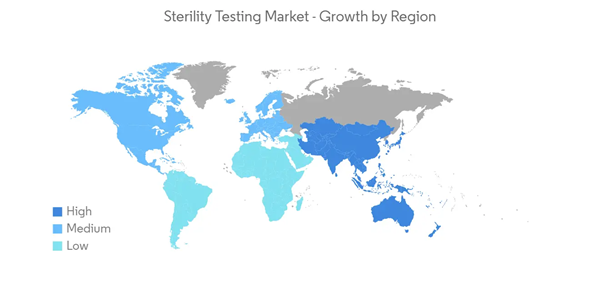
North America Expected to Have the Significant Market Share Over the Forecast Period
North America is expected to hold a significant share in the sterility testing market over the forecast period, mainly due to the growing research and innovations in the biotechnology and pharmaceutical industries in the region, the increasing investment in research and development activities in life sciences and medical technology, and favorable government initiatives.In the North American region, the United States holds the largest market for sterility testing, and it is expected to dominate over the forecast period as well. The high share of the United States in the sterility testing market is attributed to the presence of several global pharmaceutical and biopharmaceutical companies and huge funding for research and development by the government and private entities in the country. For instance,
In addition, the rising number of drug launches and increasing company focus on conducting various clinical trials in the region for treating various chronic, infectious, and other diseases are also contributing to the market growth. For instance, in March 2022, VBI Vaccines Inc. launched PreHevbrioin in the United States for the prevention of infection caused by all known subtypes of hepatitis B virus (HBV) in adults of age 18 years and older. This vaccine was approved by the FDA as the only approved 3-antigen HBV vaccine for adults in November 2021.
The increasing incidence of diabetes in compliance with the rising investment from the Canadian government for diabetes research and development is propelling the growth of the market. For instance, Canada's Budget 2021 proposed new investments in diabetes research, surveillance, prevention, and the development of a national framework for diabetes. As part of this, the spokesperson of the government of Canada announced that the Canadian government, through the Canadian Institutes of Health Research (CIHR), was planning to recommit to the JDRF-CIHR partnership to Defeat Diabetes and announced the investment of up to USD 15 million for a total research impact of USD 30 million. Such government investments are anticipated to drive market growth over the forecast period.
Furthermore, according to the data published by the CBO in February 2022, the PhRMA members spent USD 102 billion on R&D activities in the United States in 2021. Also, as per the data published by Statistics Canada from June 2022, the in-house spending of Canada on its pharmaceutical R&D activities was USD 23.4 billion in 2021, and this number was expected to increase by 1.7% to USD 23.8 billion in 2022. Thus, the expected increase in pharmaceutical and medicine manufacturing in the region is anticipated to fuel the adoption of sterility testing, which in turn is expected to propel the market growth over the forecast period.
Therefore, owing to the factors such as high R&D spending, increasing investments by the government, and rising drug launches, the studied market is expected to grow over the forecast period.
Sterility Testing Industry Overview
The sterility testing market is moderately competitive and consists of several major players. With growing opportunities in the biopharmaceutical segment, other new players are also expected to enter the market. Some of the major players in the market include BioMérieux, Charles River Laboratories Inc., Merck KGaA, Sartorius AG, SGS SA, Sotera Health LLC, and Thermo Fisher Scientific Inc.Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- bioMerieux SA
- Charles River Laboratories
- Merck KGaA
- Sartorius AG
- SGS SA
- Sotera Health (Nelson Laboratories LLC)
- STERIS
- Thermo Fisher Scientific
- Laboratory Corporation of America Holdings (TOXIKON)
- WuXi AppTec
- Rapid Micro Biosystems Inc.
- Pace Analytical