Hepatocellular carcinoma (HCC) is the most common type of chronic liver cancer in adults and the most common cause of death in patients with cirrhosis. Hepatocellular carcinoma (HCC) is the third most common cause of cancer-related death and is usually detected at an intermediate to late stage, with a poor overall patient prognosis.
In China, the incidence of primary liver cancer ranked fifth among that of malignant tumors in 2020, with 410,000 new cases, including 303,000 cases in men; the mortality rate of the cancer ranked second with 391,000 deaths in 2020, a mortality rate of 17.2/100,000 of the total population. From 2016 to 2020, global average annual incidence of primary liver cancer was 995,000 cases, with Asia 732,000 cases, accounting for 73.6%, and China 423,000 cases, accounting for 42.5%. The high incidence of liver cancer in China is highly correlated with the fact that China is a country with high prevalence of hepatitis B, environmental pollution, and some spirits-addicted residents.
Liver cancer is a common malignant tumor in China. Liver cancer starts insidiously but progresses rapidly, and by the time it is detected, it is often at an advanced stage, making the treatment of surgery ineffective. Moreover, radiotherapy is of little use in treating liver cancer. Therefore, targeted drug therapy becomes the standard treatment for patients with advanced liver cancer. Sorafenib is the world's first multi-targeted, multi-kinase inhibitor for the treatment of liver cancer. It can directly suppress tumor growth by inhibiting RAF/MEK/ERK signaling pathway, and indirectly suppress tumor growth by blocking the formation of tumor neovascularization through inhibiting VEGFR and PDGFR. Therefore, it is the first-line drug used for the treatment of advanced primary liver cancer.
Sorafenib is a multi-kinase inhibitor originally developed by Bayer AG with the trade name of Nexavar®. Sorafenib was approved for marketing by the FDA of U.S. in December 2005 and approved by China in August 2006. In 2017, Bayer’s sorafenib was included in the category B of China's national medical insurance catalog after a negotiated price reduction of about 70%, and the price after inclusion in medical insurance is about CNY200 (approximately USD30.8) per tablet. From 2018 to 2021, Bayer AG continues to reduce the price of sorafenib in the Chinese market.
According to the publisher’s market research, the sales value of China’s sorafenib market continued to grow from 2016 to 2019. In 2020, the sales value declined to approximately USD406 million due to COVID-19, but the CAGR from 2016 to 2020 was still up to 22.3%.
As of August 2021, besides Bayer's sorafenib product, only generic versions of Jiangxi Shanxiang Pharmaceutical and Chongqing Yaoyou Pharmaceutical have been officially marketed in the Chinese market. Some other companies such as Yabao Pharmaceutical Group and CSPC Ouyi Pharmaceutical have filed applications for the marketing of generic sorafenib drugs. In September 2020, The China National Intellectual Property Administration issued an invalidation request review decision (No. 46292), declaring that Bayer's patent CN200680007187.1 for sorafenib mesylate in China was invalid. The invalidation petitioner was Chongqing Yaoyou Pharmaceutical. With the invalidation of Bayer's patent applications related to sorafenib, more generic sorafenib drugs from local Chinese pharmaceutical companies will be launched in China.
For Bayer, as more generic drugs are approved and marketed, the market share of Nexavar in China will gradually decline. Bayer is expected to stop the decline of Nexavar's market share through measures such as price cuts.
The publisher expects that from 2021 to 2025, the average sales price of sorafenib in China will decline due to fierce competition while both sales volume and value will continue to rise.
Topics Covered:
- Impact of COVID-19 on China’s Sorafenib Market
- Development Environment of Sorafenib in China
- Sales Volume of Sorafenib in China
- Sales Volume and Value of Sorafenib in China by Region
- Major Sorafenib Manufacturers in China and Their Market Shares
- Sales Price of Sorafenib in China
- Prospects of China’s Sorafenib Market, 2021-2025
Table of Contents
1 Relevant Concept of Sorafenib
1.1 Indications of Sorafenib1.2 Development of China’s Sorafenib Market
1.3 Governmental Approval of Sorafenib in China
1.4 The Impact of COVID-19 on China’s Sorafenib Market
2 Sales of Sorafenib in China, 2016-2020
2.1 Sales Value2.1.1 Sales Value in China
2.1.2 Sales Value in China by Region
2.2 Sales Volume
2.2.1 Sales Volume in China
2.2.2 Sales Volume in China by Region
2.3 Sales of Sorafenib in China by Dosage Form, 2016-2020
2.3.1 Tablets
2.3.2 Other Dosage Forms
3 Analysis of Major Sorafenib Manufacturers in China, 2020
3.1 Analysis of Market Share3.1.1 Market Share of Manufacturers by Sales Value
3.1.2 Market Share of Manufacturers by Sales Volume
3.2 Bayer AG
3.2.1 Company Profile
3.2.2 Sales of Sorafenib in China
3.3 Jiangxi Shanxiang Pharmaceutical Co., Ltd.
3.3.1 Company Profile
3.3.2 Sales of Sorafenib in China
4 Sales Price of Sorafenib of Different Companies in China, 2020-2021
4.1 Bayer AG (Nexavar®)4.2 Jiangxi Shanxiang Pharmaceutical Co., Ltd. (Ligesitai®)
5 Prospects of China’s Sorafenib Market, 2021-2025
5.1 Influencing Factors for the Market Development5.1.1 The Impact of COVID-19 on the Market
5.1.2 Market Drivers and Opportunities
5.1.3 Market Threats and Challenges
5.2 Forecast on Market Size
5.3 Forecast on Market Trend
List of Charts
- Chart Patent Information of Sorafenib in China
- Chart Sales Value of Sorafenib in China, 2016-2020
- Chart Sales Volume of Sorafenib in China, 2016-2020
- Chart Sales Volume of Sorafenib in China by Region, 2016-2020
- Chart Sales Value of Sorafenib in China by Region, 2016-2020
- Chart Market Share of Sorafenib Manufacturers in China by Sales Value, 2016-2020
- Chart Sales Value and Volume of Sorafenib (of Bayer AG) in China, 2016-2020
- Chart Sales Value and Volume of Sorafenib (of Jiangxi Shanxiang Pharmaceutical Co., Ltd.) in China, 2016-2020
- Chart Sales Price of Sorafenib (of Bayer AG) in China, 2016-2020
- Chart Sales Price of Sorafenib (of Jiangxi Shanxiang Pharmaceutical Co., Ltd.) in China, 2016-2020
- Chart Forecast on Sales Value of Sorafenib in China, 2021-2025
- Chart Forecast on Sales Volume of Sorafenib in China, 2021-2025
Companies Mentioned
- Bayer AG (Nexavar®)
- Jiangxi Shanxiang Pharmaceutical Co., Ltd. (Ligesitai®)
Methodology
Background research defines the range of products and industries, which proposes the key points of the research. Proper classification will help clients understand the industry and products in the report.
Secondhand material research is a necessary way to push the project into fast progress. The analyst always chooses the data source carefully. Most secondhand data they quote is sourced from an authority in a specific industry or public data source from governments, industrial associations, etc. For some new or niche fields, they also "double-check" data sources and logics before they show them to clients.
Primary research is the key to solve questions, which largely influence the research outputs. The analyst may use methods like mathematics, logical reasoning, scenario thinking, to confirm key data and make the data credible.
The data model is an important analysis method. Calculating through data models with different factors weights can guarantee the outputs objective.
The analyst optimizes the following methods and steps in executing research projects and also forms many special information gathering and processing methods.
1. Analyze the life cycle of the industry to understand the development phase and space.
2. Grasp the key indexes evaluating the market to position clients in the market and formulate development plans
3. Economic, political, social and cultural factors
4. Competitors like a mirror that reflects the overall market and also market differences.
5. Inside and outside the industry, upstream and downstream of the industry chain, show inner competitions
6. Proper estimation of the future is good guidance for strategic planning.

LOADING...
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 40 |
| Published | September 2021 |
| Forecast Period | 2016 - 2025 |
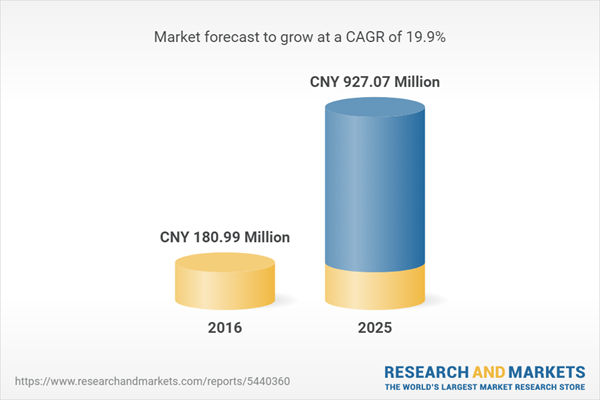
| Estimated Market Value ( CNY | CNY 180.99 Million |
| Forecasted Market Value ( CNY | CNY 927.07 Million |
| Compound Annual Growth Rate | 19.9% |
| Regions Covered | China |
| No. of Companies Mentioned | 2 |