The COVID-19 pandemic had a significant impact on the market. Adding appropriate adjuvants to COVID-19 vaccines substantially reduced the number of required doses and improved efficacy or cross-neutralizing protection. For instance, according to an article published by Viruses in February 2022, various vaccine launches for COVID-19 had shown decreased protection against variants of concern (VOCs) of SARS-CoV-2, and there were increasing numbers of breakthrough infections with VOCs in the vaccinated population. This called for the stepped-up development of adjuvanted vaccines that induce effective and durable protective immunity against SARS-CoV-2 and its emerging variants.
Thus, COVID-19 impacted the market positively. As per the above source, adjuvants used with SARS-CoV-2 vaccine candidates were aluminum-based adjuvants, STING agonist-based adjuvants, oil-in-water emulsion adjuvants, TLR agonist adjuvants, and a few more. Therefore, due to the advantage and development of new vaccine adjuvants, the demand also increased during the COVID-19 pandemic and is expected to grow in the coming period.
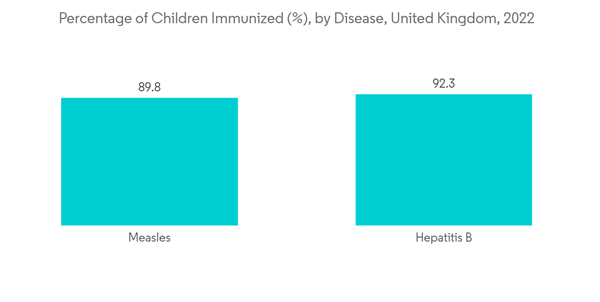
The factors that promote the growth of the vaccine adjuvants market include unmet demand for specific vaccines, increasing government vaccination recommendations, and increasing use of recombinant subunit and synthetic vaccines. For instance, according to the data published by the WHO in the Global Tuberculosis Report 2021, most of the tuberculosis cases were found in the WHO regions of Southeast Asia (43%), Africa (25%), and Western Pacific (18%), and lesser numbers of tuberculosis cases were found in Eastern Mediterranean (8.3%), the Americas (3.0%) and Europe (2.3%). Additionally, according to the WHO data published in 2021, Sub-Saharan Africa likely continues to endure the burden of malaria, accounting for approximately 95% of all cases and 96% of all deaths. Thus, rising cases of infectious diseases are expected to increase the demand for vaccines, resulting in the high growth of the vaccine adjuvants market.
Furthermore, according to the CDC update in January 2021, in order to prevent occupational exposure to the Ebola virus species Zaire ebolavirus, the Advisory Committee on Immunization Practices (ACIP) recommends preexposure vaccination with Ervebo for adults in the U.S. population older than 18 years of age who are at more risk. This is because they are responding to the Ebola virus disease outbreak (EVD). Therefore, increasing government vaccination recommendations is expected to drive market growth.
Companies are introducing new, more sophisticated vaccine adjuvants, which are anticipated to significantly support the market's growth. For instance, in December 2022, Evonik launched PhytoSquene, a non-animal-derived squalene suitable for vaccines and other pharmaceutical applications. PhytoSquene is an amaranth oil-derived squalene on the market for use in adjuvants in parenteral dosage forms. It meets the demand for a secure commercial supply of non-animal-derived squalene. Such instances are expected to drive the market's growth over the forecast period.
Thus, these factors mentioned above, such as unmet demand for specific vaccines, increasing government vaccination recommendations, and increasing use of recombinant subunit and synthetic vaccines, are anticipated to boost the market's growth over the forecast period. However, side effects, the high toxicity of adjuvants, and the high R&D cost of developing a new adjuvant are expected to hinder the market's growth.
Vaccine Adjuvants Market Trends
Carbohydrate Adjuvants Segment Expected to Witness a Significant CAGR Over The Forecast Period
Carbohydrate adjuvants are safe and biocompatible compounds usable as sustained delivery systems and stimulants of ongoing humoral and cellular immune responses, being especially suitable for the development of vaccines against intracellular pathogens where alum is useless.The carbohydrate adjuvants segment is expected to grow significantly over the forecast period due to its wide range and use in human vaccines. For instance, as per an article published by Nature Reviews Chemistry in January 2021, carbohydrates play crucial roles in the immune system function and the stimulation of the immune response that can be exploited by the chemistry community. Carbohydrates possess many beneficial properties, such as high biocompatibility and tolerability and a strong safety profile. A variety of natural carbohydrate structures, particularly MPLA and QS-21, have been clinically evaluated as adjuvants and are part of licensed Adjuvant Systems (AS) in human vaccines against HPV (AS04), herpes zoster, and malaria (AS01). Due to these characteristics, the segmental market is expected to grow.
Additionally, in October 2022, the phase 1 test of the novel low-dose influenza vaccine was developed under the INDIGO project, directed by AIGHD. This vaccine combines a new adjuvant, a component used in several vaccines to induce a stronger immune response, with a licensed influenza vaccine at 1/5th of a dose. Such an initiative is expected to drive segmental growth.
Furthermore, a few key market players are developing novel products and technologies to compete with existing products, while others are acquiring and partnering with other companies trending in the market. For instance, in March 2021, Oragenics, Inc. entered into a material transfer agreement with Biodextris Inc. for the use of three intranasal mucosal adjuvants in the company's Terra CoV-2 vaccine against COVID-19. Adjuvants are added to vaccines to enhance their immunogenicity. BDX100, BDX300, and BDX301 are proteosome-based adjuvants comprised of proteins and lipopolysaccharides with improved attributes, including enhanced immune response, manufacturing efficiency, and the benefits of intranasal vaccine administration. Such development is expected to drive the segmental growth.
Thus, owing to the aforementioned factors, such as a wide variety of vaccines, initiatives in the development of vaccines, and strategic development by key players, the carbohydrate adjuvants segment is expected to register healthy growth over the forecast period.
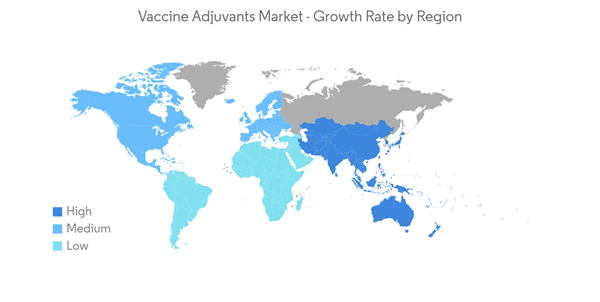
North America Expected to Hold a Significant Market Share Over the Forecast Period
North America is expected to observe significant growth in the market studied during the forecast period. The main factors driving the growth of the vaccine adjuvants market in the North American region are the presence of key market players, the availability of advanced technology, and the growing government initiatives to launch various awareness campaigns to increase vaccination. For instance, in January 2021, the United States Department of Health and Human Services (HHS) launched the HPV VAX NOW campaign to increase human papillomavirus (HPV) vaccination rates among young adults ages 18-26. Also, in August 2022, the Government of Canada published the Canadian Immunization Guide Chapter on Influenza and Statement on Seasonal Influenza Vaccine for 2022-2023, which provides updated recommendations regarding the use of seasonal influenza vaccines in Canada. Hence, such an initiative from the government is expected to drive market growth.Additionally, various strategies such as product approval, launches, agreements, and developments help the market to grow over the study period. For instance, in November 2022, GSK plc reported that the US FDA accepted a Biologics License Application (BLA) and granted priority review for its respiratory syncytial virus (RSV) older adult vaccine candidate. The US FDA grants priority review to applications for vaccines that, if approved, would offer significant improvements in the safety or effectiveness of the treatment or prevention of serious conditions when compared to standard applications.
Therefore, owing to the aforementioned factors, such as several immunization programs, government and non-government initiatives, and increased demand for combined vaccines, the vaccine adjuvants market is expected to augment growth over the analysis period.
Vaccine Adjuvants Industry Overview
The vaccine adjuvants market is consolidated in nature due to the presence of a few companies operating globally and regionally. The strategic initiatives taken by various governments to improve immunization and research and development for vaccine development by market players are propelling the market growth. The companies in the market include Adjuvatis, GlaxoSmithKline PLC, Croda International PLC, Merck KGaA, and Novavax Inc., among others.Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Associated British Foods Plc (spi Pharma Inc.)
- Adjuvatis
- Agenus Inc.
- Aphios Corporation
- Vertellus
- Croda International PLC
- CSL Limited
- Dynavax Technologies Corporation
- GlaxoSmithKline PLC
- InvivoGen
- Merck KGA
- Novavax Inc.
- OZ Biosciences
- Pacific GeneTech Limited
- Air Liquide (seppic)
- Thermo Fisher Scientific Inc.