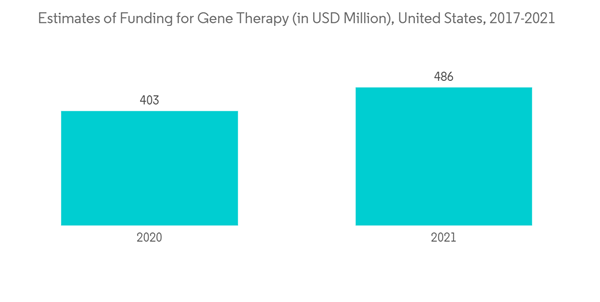Amid the COVID-19 pandemic, the health systems of various countries rapidly invested in research and development to combat the virus. Potential compounds were screened from the CHEMBL, ZINC, FDA-approved drugs, and molecules under clinical trials. Research groups worldwide have identified drugs for the treatment of COVID-19 by screening both novel and existing drugs for their ability to alleviate symptoms and stop viral replication. Thus, the drug discovery market was positively impacted due to the urge to find a cure for COVID-19. Furthermore, post the pandemic, there is a rise in investments in different modalities of drug development, such as gene and cell therapies for both COVID-19 diseases and other novel diseases, which is expected to impact the market's growth as per the analysis positively. Furthermore, the research for developing antiviral drugs for COVID-19 is still in progress. For instance, in May 2022, the National Institute of Allergy and Infectious Diseases (NIAID) awarded approximately USD 577 million to establish nine Antiviral Drug Discovery (AViDD) Centers for Pathogens of Pandemic Concern. The AViDD centers will carry out creative, interdisciplinary research to create candidate COVID-19 antivirals, particularly those that can be used outside of a hospital setting, as well as antivirals that target particular viral families with a high potential to trigger a pandemic in the future. Such grants are expected to drive the drug discovery market's growth post-pandemic.
The propelling factors for the growth of the drug discovery market include the rising burden of a wide range of diseases (such as cardiovascular and CNS-related disorders), rising healthcare expenditure, and the upcoming patent expiration of blockbuster drugs.
Cardiovascular diseases, infectious diseases, diabetes, and kidney-related complications are highly prevalent worldwide. According to key facts about cardiovascular diseases (CVDs) published by the WHO in June 2021, CVDs are the leading cause of death globally, representing around 32% of all global deaths. CVD remains the leading cause of death and disability and remains highly complicated, costly, and burdensome. Moreover, new strategies are required to streamline and reduce the costs of clinical trials while placing greater importance on the patient experience and voice. Thus, the increasing burden of a diverse range of diseases among all age groups and its burden globally drive the demand for drug discovery and its market.
Furthermore, there has been an upsurge in the usage of advanced technologies, such as high-throughput process development, bioinformatics, and combinatorial chemistry, for better drug candidate identification. Drug discovery has evolved significantly with emerging technologies, helping the process become more refined, accurate, and time-efficient. The adoption of artificial intelligence systems in disease diagnostics and treatment design is growing, and this is expected to witness robust growth in the coming years. For instance, in March 2020, Existencia, an AI-based drug discovery company, entered a collaborative agreement with Diamond Light Source and Scripps Research to identify COVID-19 antiviral therapies. Thus, these factors are anticipated to contribute to the market's growth over the forecast period.
However, the huge capital investment, low-profit margins, and stringent drug discovery regulations are expected to hinder the market growth.
Drug Discovery Market Trends
Biologics Drug Segment is Expected to Show a Significant Growth Over the Forecast Period.
During the pandemic, the rising adoption of biologics due to their specific advantages, especially target specificity, bodes well for the market studied. As a result, many major biotech firms are in the middle of a race to investigate the SARS-CoV-2 genome and prepare therapeutics. Furthermore, COVID-19 has significantly impacted the growth of biologics due to the rise in the development of biologics for SARS-CoV-2. For instance, in February 2022, AbCellera launched bebtelovimab (LY-CoV1404), the second antibody developed through AbCellera's collaboration with Eli Lilly and Company (Lilly), which has received Emergency Use Authorization from the FDA for the treatment of mild to moderate COVID-19.Biologic drugs offer several benefits, such as highly effective and potent action, fewer side effects, and the potential to cure diseases rather than merely treat the symptoms, significantly increasing the demand for these products.
The efficacy and safety of biologics have led to their rapid adoption by people, driving the growth of the biologics segment. This has led to an increase in partnerships among drug discovery companies for the development of biologics. For instance, in August 2022, Sanofi and Innovent Biologics entered a strategic collaboration to accelerate the development of oncology medicines and expand their presence in China. In addition, in February 2022, Abzena, Alira Health, and Oncodesign launched DRIVE biologics. These companies are entering a consortium to offer their partners synergy and expertise in biologics drug discovery and development programs. Such partnerships are expected to drive the growth of this segment due to the rise in the development and adoption of biologics for various diseases over the forecast period.
Moreover, the rise in approvals for biologics from international regulatory agencies is further expected to propel the growth of this segment. For instance, in February 2022, the FDA accepted for review the Prior Approval Supplement (PAS) to the Biologics License Application (BLA) for ABRILADA (adalimumab-afzb) as an interchangeable biosimilar to Humira (adalimumab). Such initiatives are expected to drive the growth of the biologics segment.
Therefore, owing to the above-mentioned factors, the biologics segment is expected to experience tremendous growth.
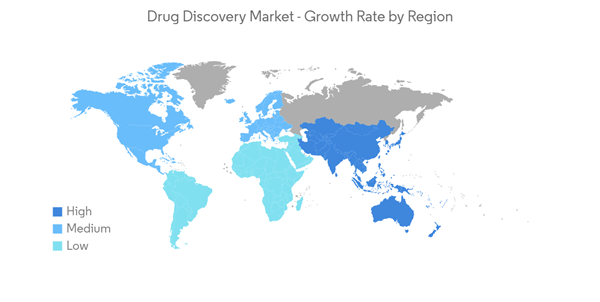
North America is Expected to Hold a Significant Market Share Over the Forecast Period
North America has a significant share of the world's R&D spending on pharmaceuticals and biologics. The factors contributing to the growth of this region include a rise in investments by drug development companies, huge grants from the government of the United States, a strong foothold of major drug development companies, well-developed healthcare infrastructure, and a rise in chronic diseases, among others.For instance, according to the ACS "Cancer Facts & Figures 2022," around 1,918,030 new cancer cases and 609,360 deaths due to cancer are estimated to be reported in the United States by the end of 2022. Such a high incidence of cancer in this region is anticipated to drive drug discovery for cancer, driving the market growth.
Furthermore, the rise in technological advancements in drug discovery in the United States is expected to drive the drug discovery market in this region. For instance, in September 2022, CytoReason entered a multi-year partnership with Pfizer to use CytoReason's artificial intelligence technology for Pfizer's drug development program. Such collaborations among key market players in the United States to implement AI technology in drug discovery are expected to drive the growth of this market studied in this region.
Therefore, owing to the above-mentioned factors, the drug discovery market is expected to increase over the forecast period.
Drug Discovery Industry Overview
The drug discovery market is highly competitive and consists of a number of major players. The market players are implementing innovative strategies and adopting artificial intelligence and machine learning in drug discovery to speed up the process. Furthermore, the key market players are entering into strategic partnerships to develop novel drug molecules. Some of the companies in this market include Eli Lilly and Company, Novartis AG, Bayer AG, AbbVie Inc., AstraZeneca PLC, F. Hoffmann-La Roche Ltd., Pfizer Inc., and Sanofi.Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- AbbVie Inc.
- Bristol-Myers Squibb Company
- AstraZeneca PLC
- Bayer AG
- Eli Lilly and Company
- F. Hoffmann-La Roche Ltd
- GlaxoSmithKline PLC
- Merck & Co. Inc.
- Novartis
- Pfizer Inc.
- Takeda Pharmaceuticals
- Sanofi