The COVID-19 pandemic had a notable impact on the market which resulted in the cancellation of spinal surgeries to manage, and many hospitals are facing a shortage of professionals to assist with these surgeries. For instance, the NCBI article published in December 2021 reported that over one-third of elective spine surgeries were canceled due to COVID-19. Additionally, the Cureus article published in March 2022 reported that relative search volume for lumbar fusion, lumbar fusion surgery, and lumbar spinal fusion declined during the COVID period by 23%, 15.6%, and 5.4%. Such a decline in spinal surgeries during the outbreak of the pandemic had a significant impact on the growth of the market. However, the resumption of all elective surgeries, including spinal surgeries, during the post-pandemic period is expected to drive the market's growth during the pandemic period.
The spinal implants and surgical devices market is primarily driven by the increasing burden of spinal disorders, rising demand for minimally-invasive surgeries, and technological advancements in bone grafting products.
For instance, the article published in NCBI in October 2021 mentioned that the global incidence of traumatic spinal fractures was 10.5 per 100,000 persons, with an estimated 768,473 new cases of spinal injury annually worldwide. Also, another NCBI article published in November 2022 mentioned that in younger patients, about 50% of spine fractures were due to motor vehicle collisions, with another 25% being due to falls. The report also mentioned that the annual incidence of vertebral compression fracture was 10.7 per 1000 women and 5.7 per 1000 men globally. Similarly, according to Spinal Injury Association United Kingdom 2021, about 2500 people are injured or diagnosed with spinal cord injuries every year, and an estimated 50,000 people are living with spinal cord injuries in the United Kingdom. Such a high incidence of spinal injuries, disorders, and fractures among the global population is expected to drive the demand for spinal implants and surgical devices, thereby contributing to the market's growth over the forecast period.
On the other hand, the rising key players' strategies, such as acquisitions and mergers to develop advanced products, are expected to contribute to the growth of the market. For instance, in February 2021, NuVasive, Inc. acquired Simplify Medical, a privately held company and developer of the Simplify Cervical Artificial Disc (Simplify Disc) for cervical total disc replacement (cTDR).
Thus, the increasing incidence of spinal injuries, fractures, and disorders, along with increasing demand for suitable spinal implants and advanced devices, are expected to drive the market's growth over the forecast period. However, the high cost of the procedures and stringent regulatory approval procedures are likely to hinder market growth.
Spinal Implants & Surgical Devices Market Trends
Minimally Invasive Surgery is Expected to Hold the Significant Market Growth Over the Forecast Period
Minimally Invasive Spine (MIS) surgeries are performed to stabilize the vertebral bones and spinal joints and/or relieve the pressure being applied to the spinal nerves, often a result of conditions such as spinal instability, bone spurs, herniated discs, scoliosis, or spinal tumors. Factors such as the high incidence of spinal disorders, along with the highly increasing demand for effective surgical options and rising adoption of minimally invasive spine surgeries, are expected to contribute to the growth of the studied segment. For instance, the NCBI article published in January 2022 mentioned that the incidence of herniated discs globally was about 5 to 20 cases per 1000 adults annually, and the estimated prevalence of symptomatic herniated discs of the lumbar spine was about 1%-3% of patients. Such incidences of spinal disorders are expected to drive the demand for minimally invasive spinal surgeries, thereby contributing to segment growth.Additionally, the increasing development of advanced minimally invasive spinal surgeries by various market players is also expected to drive segment growth. For instance, in October 2021, SeaApine Holdings Corporation announced the completion of the first cases in both the United States and Australia for the 7D surgical percutaneous spine module. The release of the percutaneous module represents an application and increased functionality for its 7D flash navigation system.
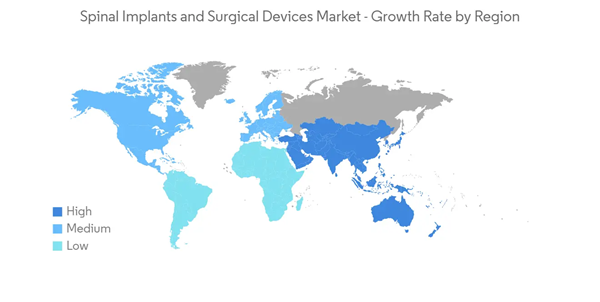
North America is Expected to Hold Major Market Share in the Studied Market Over the Forecast Period
The North American spinal implants and surgical devices market is expected to hold a significant share and is expected to show a similar trend over the forecast period. The growth in the region is driven by technological advancement, increasing incidence of spinal injuries, fractures, and disorders, and reimbursement models that incentivize complex fusion surgery.For instance, the NCBI article published in November 2022 mentioned that approximately 1 to 1.5 million vertebral compression fractures occur annually in the United States alone. The report also stated that annually 40% to 50% of patients over age 80 had a sustained vertebral compression fracture in the United States. Also, the Elsevier article published in May 2022, the prevalence of vertebral fractures in Canada was estimated to be 23.5% in females and 21.5% in males in 2021. Additionally, the article published in Frontiers Journal in September 2022, mentioned that in Canada, more than 86,000 people were affected by spinal cord injuries from either a traumatic or non-traumatic cause. Such a high incidence of various spinal injuries and disorders in North America is expected to drive the demand for spinal implants and surgical devices, thereby boosting the regional market growth.
On the other, the increasing funding from various organizations to treat spinal injuries in this region is also expected to contribute to the market growth. For instance, in January 2022, the University of British Columbia received a USD 24 million grant to treat spinal cord injury and to develop personalized, less invasive treatments. Moreover, the increasing product approvals in this region are also expected to contribute to the market's growth. For instance, in March 2022, Evoke Spinal Cord Stimulation (SCS) System was approved by the Food and Drug Administration (FDA). Evoke System is an implanted, rechargeable spinal cord stimulation system intended to treat long-term (chronic) pain in the trunk or limbs that is difficult to manage. Also, in January 2022, Senza Spinal Cord Stimulation (SCS) system was approved by FDA. It includes an implanted signal generator that is connected to one or two implanted leads and remote control that can turn the stimulator ON or OFF to allow adjustments of therapy settings.
Thus, owing to the abovementioned factors, such as increasing spinal injuries along with the increasing technological advancements and rising product developments, the market is expected to have significant growth in the North American region over the forecast period.
Spinal Implants & Surgical Devices Market Competitor Analysis
The Spinal Implants and Surgery Devices Market studied is moderately competitive, owing to a few major and small players in the market. The market players are focusing on technological advancements and reducing the cost of the products. Some of the major players in the market are Alphatec Spine Inc., Depuy Synthes (Johnson & Johnson), Globus Medical Inc., NuVasive Inc., Orthofix Holdings Inc., RTI Surgical Inc., Stryker Corporation, and Zimmer Biomet.Additional benefits of purchasing the report:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Alphatec Spine Inc.
- Depuy Synthes (Johnson & Johnson)
- Globus Medical Inc.
- Spineart
- Medtronic Plc
- NuVasive Inc.
- Orthofix Holdings Inc.
- RTI Surgical Inc
- Stryker Corporation
- Zimmer Biomet
- Advin Healthcare
- DeGen Medical Inc.
- Osseus