COVID-19 had a significant impact on the growth of the market during the pandemic period. Chronic obstructive pulmonary disease (COPD) patients had an increased risk of severe pneumonia and poor outcomes when they develop COVID-19. This may be related to poor underlying lung reserves or increased expression of ACE-2 receptors in small airways. An NCNI article published in March 2022, mentioned that COPD is associated with worse clinical outcomes from COVID-19. Also, according to a research study published in the Nature Journal in February 2021, in South Korea, among the COVID-19 infected patients who are 40 years and older, 3.1% had COPD; of these 7.1% received ICU care, and 5.7% received mechanical ventilation. Thus, the COVID-19 pandemic had a significant impact on the asthma and COPD drugs market.
Some of the factors that are driving the market growth include an increase in the incidence and prevalence of asthma and COPD, advanced therapeutic drugs, and a growing geriatric population.
Chronic obstructive pulmonary disease (COPD) is a common, preventable, and treatable chronic lung disease that affects men and women worldwide. According to the NCBI article published in March 2022, the estimated pooled prevalence of COPD was 11.1% in South Asian countries in 2021. The report also mentioned that the prevalence of COPD was highest in North India (19.4%) and Bangladesh (13.5%). Moreover, the increasing geriatric population globally is boosting the market growth. According to the WHO data published in October 2022, the population aged 60 years and over will increase to 1.4 billion in 2030 and is expected to reach 2.1 billion by 2050. The UN report in 2022 mentioned that the share of the population aged 65 years or over is projected to rise from 10% in 2022 to 16% in 2050. As, the geriatric population is most prone to developing asthma conditions, the increase in the geriatric population is expected to contribute to the growth of the market.
In addition, technological advancements are anticipated to provide the market with lucrative opportunities. Limitations of traditional treatments, such as the late onset of action and low efficacy, are projected to encourage the development of novel targeted drugs, thereby boosting the market. For instance, in February 2021, Sanofi, in collaboration with Regeneron Pharmaceuticals, initiated a Phase III clinical trial to assess the efficacy and safety of itepekimab on the annualized rate of acute moderate-or-severe COPD exacerbations in former smokers with moderate-to-severe chronic obstructive pulmonary disease (COPD). The trial was expected to be completed in July 2024.
The market players are adopting various strategies such as product launches, developments, collaborations, acquisitions, and expansions to increase their market shares. For instance, in June 2021, Valeo Pharma Inc. commercially launched the Enerzair Breezhaler and Atectura Breezhaler for the treatment of asthma patients in Canada. These products have demonstrated that they improve the level of control of asthma symptoms and better prevent related complications.
Thus, the factors such as an increase in the incidence and prevalence of asthma and COPD, advanced therapeutic drugs, and increasing product developments are expected to contribute to the growth of the market. However, factors such as stringent government regulations for product approvals and side effects associated with drugs are expected to impede market growth over the forecast period.
Asthma & COPD Drugs Market Trends
The Asthma Segment is Expected to Hold a Significant Market Share Over the Forecast Period
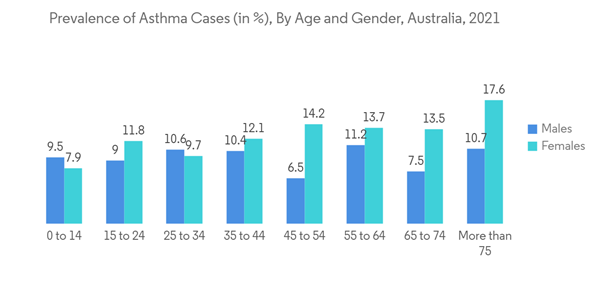
Asthma is considered one of the world's most common diseases affecting the population and a life-threatening condition affecting patients' regular breathing. It is a persistent condition that stretches and narrows the air passages of the lungs, causing constant assaults of breathlessness, bronchospasm, and reversible obstruction of airflow. Moreover, there is a large rise in the consumption of cigarettes, which contributes to asthma symptoms. In addition, the rise in industrialization has also contributed to an increase in the number of cases of asthma, which is expected to drive the growth of the studied segment.The substantial increase in the prevalence and incidence of asthma has increased the patient population, increasing the demand for these drugs. According to the Australian Bureau of Statistics data 2021, over 2.7 million Australians had asthma in 2021. The data also mentioned that females were more likely to have asthma (12% compared to 9.4%). Enormous investments in the study of asthma drugs accompanied by strategic collaborations such as mergers and acquisitions are the business strategies followed by the major companies in the market.
For instance, in March 2021, GlaxoSmithKline PC, in collaboration with Iqvia Pty Ltd, initiated a Phase III clinical trial to evaluate the efficacy and safety of GSK3511294 (Depemokimab) in adult and adolescent patients with severe uncontrolled asthma. The trial was expected to be completed in October 2023. Similarly, in February 2021, AstraZeneca PLC, in collaboration with Amgen Inc., initiated a Phase I clinical trial to evaluate the pharmacokinetic (PK) profile of a single subcutaneous (SC) dose of tezepelumab in children aged ≥ 5 to 11 years with asthma. The trial was expected to be completed in September 2021. Thus, the aforementioned factors such as an increase in the prevalence and incidence of asthma and rising product developments are expected to drive segment growth over the forecast period.
North America is Expected to Hold a Significant Market Share Over the Forecast Period
The North American region is expected to dominate the asthma and COPD drugs market during the forecast period. This growth can be attributed to the rising incidence of diseases such as COPD and asthma, the aging population, increasing research and development activities, and the presence of key market players.The rising prevalence of Asthma and COPD in North American Countries is expected to fuel the growth of the market. For instance, the AAFA article published in September 2022, mentioned that more than 25 million people in the United States have asthma. And an NIH article published in March 2022 mentioned that in the United States, COPD affected more than 15 million adults. Similarly, asthma is one of the major public health concerns in Canada which is expected to contribute to the growth of the studied market. According to the Statistics Canada data published in August 2022, over 2,802,700 cases of asthma were recorded in Canada. The high number of patients affected by asthma increases the demand for drugs for the treatment of the target disease, thereby boosting the growth of this region.
Moreover, the rise in the number of fundings undertaken by government and non-government organizations is expected to boost market growth in this region. For instance, in March 2022, United States federal health programs issued a bill that includes USD 8.5 billion to continue support for CDC asthma programs. This includes USD 30 million for the National Asthma Control program. Such funding is likely to increase awareness about asthma, which will increase the adoption of drugs in the treatment of asthma, leading to verticle growth.
In addition, the local presence of key pharmaceutical players, advanced healthcare infrastructure, and increasing research and development activities in the development of novel therapies are boosting the market growth. For instance, in March 2021, AstraZeneca PLC started a Phase III clinical trial in Mexico and the United States, to assess the efficacy and safety of budesonide/glycopyrronium/formoterol inhalers in adults and adolescents with severe asthma inadequately controlled with the standard of care. The trial was expected to be completed in September 2024. Also, in July 2021, AstraZeneca’s Biologics License Application (BLA) for tezepelumab was accepted and granted Priority Review for the treatment of asthma by the United States Food and Drug Administration (FDA). Tezepelumab is being developed by AstraZeneca in collaboration with Amgen Inc. Thus, the factors such as a rise in the number of fundings, increasing research and development activities, and increasing prevalence of Asthma and COPD are expected to drive market growth over the forecast period.
Asthma & COPD Drugs Market Competitor Analysis
Asthma and COPD drugs market is fragmented and competitive. The industry is competitive in nature, with the key players involved in mergers and acquisitions, new product developments, collaborations and partnerships, and expansion strategies to augment market penetration. Companies like GlaxoSmithKline PLC, Merck & Co. Inc., AstraZeneca PLC, Boehringer Ingelheim GmbH, and Novartis AG, among others, hold a substantial market share.Additional benefits of purchasing the report:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- AstraZeneca PLC
- Boehringer Ingelheim GmbH
- GlaxoSmithKline PLC
- Novartis AG
- F. Hoffmann-La Roche Ltd
- Pfizer Inc.
- Grifols SA
- Merck & Co. Inc.
- Sanofi SA
- Sumitomo Dainippon Pharma Co. Ltd
- Teva Pharmaceutical Industries Ltd
- Chiesi Farmaceutici SpA