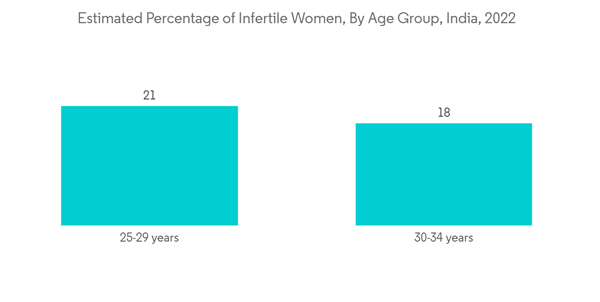The Women's Health Diagnostics Market size is estimated at USD 28.88 billion in 2024, and is expected to reach USD 39.40 billion by 2029, growing at a CAGR of 6.41% during the forecast period (2024-2029).
This product will be delivered within 2 business days.
Key Highlights
- During the early days of the COVID-19 pandemic, the women's health diagnostics market experienced a setback due to unfavorable changes in guidelines and regulations, as well as postponed and limited screening, operations, and diagnostic tests at hospitals and breast cancer treatment facilities. However, in the later stages of the pandemic, there was an increase in demand for diagnostic tests to identify menstrual abnormalities due to the post-COVID impacts on the menstrual cycle.
- Research published in the Journal of Education and Health Promotion in June 2022 showed that SARS-CoV-2 occurrences caused ovarian, uterine, and vaginal infections due to the widespread expression of angiotensin-converting enzyme 2 (ACE2), disrupting the female reproductive system's operations, resulting in infertility and various menstrual illnesses. Thus, COVID-19 patients were advised to complete their fertility assessments after recovery. These factors have created a demand for various diagnostic tests. They are expected to stabilize the loss incurred during the initial pandemic days, fueling the growth of the women's health diagnostics market during the forecast period.
- The market is expected to experience rapid growth due to the increasing incidence of chronic and lifestyle-related disorders, the rising number of imaging and diagnostic centers, and the increased adoption of point-of-care and rapid diagnostic tests, along with collaborations and partnerships by players. The increasing burden of diseases such as breast cancer, ovarian and cervical cancer, autoimmune diseases, and infertility among women creates the need for proper diagnosis. Additionally, the increasing stress rate among women is driving the burden of various conditions such as obesity, the risk of drug abuse, and increasing alcohol use, further leading to infertility among them.
- For example, data updated by the Census Bureau in April 2022 showed that fertility rates of women ages 20-24 declined by 43% in recent years, with only 66.59 births to every 1,000 women in that age range, indicating a 42.79% decline in fertility from past decades. Furthermore, as per a study published and revised by NIH in January 2023, nearly 11% of women of reproductive age, i.e., 18 to 35 years in the United States, have experienced fertility problems in current years. Thus, with the increasing rate of infertility among women, the demand for fertility testing and diagnosis is increasing in developing countries, which is anticipated to fuel market growth during the forecast period.
- Moreover, with the increasing awareness among women about their health and the rising burden of diseases, there is a constant need for imaging and diagnostic centers worldwide. Since most diagnostic and imaging tests are performed at diagnostic centers, it is expected to drive the market's growth. The Food and Drug Administration's (FDA's) Mammography Quality Standards Act and Program (MQSA) update in March 2023 showed that nearly 24,710 accredited units for mammography were present in about 8,821 certified facilities in the United States as of March 2023. Thus, the availability of such facilities in developing countries worldwide creates several opportunities for women's health diagnostics and is expected to drive the market's growth.
- Furthermore, the various strategies adopted by key market players are expected to boost the market. For instance, in June 2021, OWN (Oprah Winfrey Network) and its first-ever health initiative, Own Your Health, announced an exclusive partnership with Hologic's Project Health Equality public health initiative. The collaboration aims to raise awareness about disparities in healthcare quality received by Black women in the United States for breast, cervical, and uterine fibroids. Therefore, the rise in demand for the development of diagnostic devices, a rise in healthcare infrastructure, and new marketing initiatives by key vendors in emerging countries are expected to provide lucrative opportunities for market expansion during the forecast period.
Women's Health Diagnostics Market Trends
Breast Cancer Segment is Expected to Show Significant Growth During the Forecast Period
- Breast cancer is a serious health concern that affects a significant number of women worldwide. Detecting the disease early can lead to better results, such as a higher survival rate, more treatment options, and an improved quality of life. Due to the rising incidence of breast cancer, the demand for breast cancer tests is increasing, both in developing and developed countries, which is expected to drive segment growth over the forecast period.
- Breast cancer is the most commonly occurring cancer in women and the second most common cancer worldwide. The increasing patient pool of breast cancer is expected to accelerate the demand for breast cancer diagnostics. As per the data updated by Cancer Australia in January 2023, nearly 20,030 new breast cancer cases were diagnosed in the country in 2021, with 164 cases in males and 19,866 cases in females.
- The same source also reported that in 2021, nearly 1 in 15 (or 6.7%) females were at risk of being diagnosed with breast cancer by age 85. Hence, the high burden of breast cancer is increasing the demand for breast cancer diagnostics and generating the need for diagnostic devices and tests, leading to the growth of the segment over the forecast period.
- Key companies operating in the women's health diagnostics market are adopting various strategies to gain a competitive edge in the market. They are focusing on organic growth strategies, such as product launches, advanced product development, and product approvals. Furthermore, they are adopting inorganic strategies, such as mergers and acquisitions and collaborations. For instance, in January 2021, Hologic Inc. acquired Biotheranostics, a privately held, commercial-stage firm that develops molecular diagnostic tests for metastatic and breast cancers.
- Product launches are another key factor driving the growth of the market. For example, in January 2021, Roche launched its CE-IVD-approved automated digital pathology algorithms, uPath HER2 (4B5) image analysis and uPath HER2 Dual ISH image analysis for breast cancer. These algorithms help determine the best treatment strategy for each patient. Therefore, the abovementioned factors are likely to boost the growth of the segment over the forecast period.
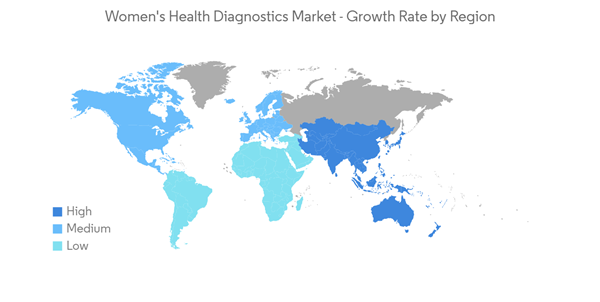
North America Expected to Hold a Significant Market Share During the Forecast Period
- North America is projected to dominate the women's health diagnostics market due to several factors, such as the high prevalence of chronic diseases among women, including cancer, lifestyle-related disorders, high adoption of technologically advanced products, product launches, and government initiatives. The rising incidence of breast and ovarian cancer and other chronic diseases is contributing to the increasing demand for women's health diagnostics in the region. Furthermore, the presence of key market players in the region is boosting market growth.
- For instance, in 2022, nearly 287,850 new cases of invasive breast cancer and 51,400 new cases of non-invasive (in situ) breast cancer were diagnosed among women in the United States, according to the Breast Cancer Organization. Breast cancer accounts for about 30% of all new cancer cases in women in the United States annually. Thus, the high prevalence of breast cancer is expected to drive the demand for women's health diagnoses and boost the market's growth over the forecast period.
- In addition, the strong foothold of key market players offering technologically advanced products for women's health diagnostics is expected to further fuel regional growth. For instance, Lia launched the world's first flushable and biodegradable pregnancy test in March 2021. Lia's pregnancy tests are made from plant fibers and are 100% plastic-free, which means they biodegrade in just ten weeks.
- Moreover, regulatory approvals from authorities such as the United States Food and Drug Administration (USFDA) are also contributing to the market growth in the region. For example, in January 2021, the Center for Devices and Radiological Health (CDRH) of the USFDA granted premarket approval (PMA) to Seno Medical Instruments Inc. for its Imagio Breast Imaging System. The diagnostic breast cancer imaging technology will help physicians better differentiate between benign and malignant breast lesions using non-invasive optoacoustic/ultrasound (OA/US) technology to provide information on suspicious breast lesions. Such approvals are fueling the growth of the market.
- Therefore, considering the above factors, the North American women's health diagnostics market is anticipated to exhibit significant growth over the forecast period.
Women's Health Diagnostics Industry Overview
The women's health diagnostics market is experiencing a significant contribution from numerous companies. Major players in the market include Siemens Healthcare Private Limited, Hologic Inc., GE Healthcare, F.Hoffmann-La Roche Ltd, and Koninklijke Philips NV. These companies are implementing various strategies such as new product development, mergers, and acquisitions to strengthen their market presence and drive growth in the market.Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
1 INTRODUCTION
4 MARKET DYNAMICS
5 MARKET SEGMENTATION (Market Size by Value - USD million)
6 COMPETITIVE LANDSCAPE
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Abbott Laboratories
- Becton, Dickinson and Company
- Biomerieux SA
- Carestream Health
- F. Hoffmann-La Roche AG
- GE Healthcare
- Hologic Inc.
- Koninklijke Philips NV
- Mankind Pharma
- Myriad Genetics
- Perkinelmer Inc.
- Procter & Gamble Co.
- Quest Diagnostics Inc.
- Siemens Healthineers AG