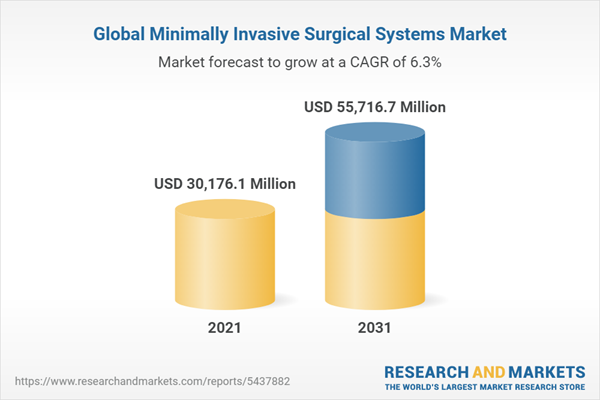
The global minimally invasive surgical systems market is estimated to be valued at $27,882.8 million in 2020 and is anticipated to reach $55,716.7 million by the end of 2031, growing at a CAGR of 6.3% during 2021-2031.
Market Report Coverage - Minimally Invasive Surgical Systems
Market Segmentation
- Product - Conventional Technologies (Endoscopes and Endoscopic Systems, Video and Visualization Systems, Capsule Endoscopy, Instruments and Accessories, Others), Surgical Robotics (Robotic Systems, Instruments and Accessories, Services)
- Application - General Surgery, Urology Surgery, Gynecology Surgery, Orthopedic Surgery, Cardiovascular Surgery, Neurosurgery, Others
- End User - Hospitals, Ambulatory Surgical Centers (ASCs), Specialty Clinics
- Region - North America, Europe, Asia-Pacific, Latin America, Middle East and Africa
Regional Segmentation
- North America - U.S., Canada
- Europe - Germany, France, Spain, U.K., Italy, Netherlands, Belgium, Switzerland, Sweden, Denmark, Rest-of-Europe
- Asia-Pacific - Japan, South Korea, India, China, Australia and New Zealand, Malaysia, Singapore, Rest-of-Asia-Pacific
- Latin America - Brazil, Mexico, Argentina, Rest-of-Latin America
- Middle East and Africa - Kingdom of Saudi Arabia (K.S.A.), United Arab Emirates (U.A.E.), Israel, Rest-of-Middle East and Africa
Market Growth Drivers
- Rising Incidence of Chronic Diseases
- Advantages Over Open Surgeries
- Increase in Global Geriatric Population
- Increasing Healthcare Costs
- Technological Advancement in Minimally Invasive Surgical Procedures and Technologies
Market Challenges
- High Cost of Minimally Invasive Surgical Robotic Systems
- Shortage of Skilled Professionals
- Restrictive Reimbursement Landscape
Market Opportunities
- Engage in Mergers and Acquisitions to Diversify Endoscopic Portfolio
- Development of Surgical Simulators for Training Professionals
- Development of Low-Cost Surgical Robotic Systems
Key Companies Profiled
Arthrex Inc., Asensus Surgical, Inc., Boston Scientific Corporation, Conmed Corporation, Fujifilm Holdings Corporation, Intuitive Surgical, Inc., Karl Storz SE & Co. KG, Medtronic plc, Olympus Corporation, Hoya Corporation (Pentax Medical), Siemens Healthineers AG, Smith & Nephew plc, Sony Corporation, Stryker Corporation, Zimmer Biomet Holdings, Inc.
How This Report Can Add Value
Who should buy this report?
- Surgical Instruments and Accessories (both Conventional MISS and Surgical Robotics) OEMs to gain a holistic view about the market potential of various systems and the developing economies for business expansions.
- Surgical Robotics Manufacturers
- Established healthcare companies that are integrating software, machine learning, and AI into the surgical robotic platform
- Endoscopes, Endoscopic Systems, Capsule Endoscopy Manufacturers
- Established medical technology companies to gain insights about the market potential, market entry strategies, new technologies in the market, and key competitors.
- Medical Research Institutions
- Healthcare Facilities
- Specialty Clinics
Key questions answered in the Report
- What are the key regulations governing the minimally invasive surgical systems market, across the globe?
- What are the key technological developments on which the current industry leaders are spending their major share of research and development (R&D) investment?
- How is the role of minimally invasive surgery technologies expected to evolve in the future?
- Who are the leading players who hold significant dominance on the global minimally invasive surgical systems market, currently?
- How likely are current business models to persist in the future?
- What are the key market drivers, challenges, growth opportunities, and trends?
- How do minimally invasive procedures function as compared to each other?
- How is the expiration of patents likely to disrupt the market dynamics?
- What are the key strategies incorporated by the leading players in the minimally invasive surgical systems landscape?
- What is the likelihood of new players entering the market in near term?
- What is the current revenue contribution for different endoscopes and endoscopy systems, and what are the expected modifications in the forecast period?
- What is the current revenue contribution for the different types of conventional minimally invasive surgical systems, and what are expected modifications in the same forecast period?
- What is the current revenue contribution for the different types of surgical robotic systems, and what are the expected modifications in the same forecast period?
- Which countries contribute to the major share of current demand and which countries hold significant scope for business expansion activities, by the key players?
Minimally Invasive Surgical Systems Market Overview
As per the National Cancer Institute, minimally invasive surgery is defined as, “surgery that is done using small incisions (cuts) and few stitches. During minimally invasive surgery, one or more small incisions may be made in the body. A laparoscope (thin, tube-like instrument with a light and a lens for viewing) is inserted through one opening to guide the surgery. Tiny surgical instruments are inserted through other openings to do the surgery. Minimally invasive surgery may cause less pain, scarring, and damage to healthy tissue, and the patient may have a faster recovery than with traditional surgery.”
As per the U.S. Food and Drug Administration (FDA), “Different types of computer-assisted surgical systems can be used for pre-operative planning, surgical navigation, and to assist in performing surgical procedures. Robotically assisted surgical (RAS) devices are one type of computer-assisted surgical system. Sometimes referred to as robotic surgery, RAS devices enable the surgeon to use computer and software technology to control and move surgical instruments through one or more tiny incisions in the patient’s body (minimally invasive) for a variety of surgical procedures.”
Market Drivers
Presently, the factors driving the growth of the market as per the minimally invasive surgical systems report include rising incidence of chronic diseases, increase in global geriatric population, advantages of MIS over open surgeries, increasing healthcare costs, technological advancements in surgical procedures and technologies.
Market Challenges
The challenges that are restricting the growth of the market include high cost of surgical robotics systems, shortage of skilled professionals, and restrictive reimbursement landscape.
High cost of the surgical robotic system is among the most challenging factors for the end-user to buy and treatment for patients. It has been observed that many patients prefer international travel for their surgeries to take the advantage of currency rate. Also, due to the shortage of skilled professionals have led a slight increasing the expense of the robotic surgery. Restrictive reimbursement landscape is another factor affecting the growth of the global minimally invasive surgical systems market.
Market Opportunities
Potential opportunities that is likely to boost the growth of the market include development of low-cost surgical robotic systems, development of surgical simulators for the training of professionals, and engagement in mergers and acquisitions to diversify the endoscopic portfolio.
The current radiation therapy systems have a price of around $2 million, therefore many companies are working in developing cost-effective system and even working towards the development of a low-cost surgical robotic platform. And with time, technologically advanced products are being working, researched to get an upgraded product.
Impact of COVID-19
The COVID-19 pandemic had a huge impact on elective surgical procedures in the first half of 2020. The COVID-19 pandemic has drastically impacted the daily hospital routine services on the global level. During the initial phase of the pandemic, hospitals greatly reduced or cancelled conducting elective surgical procedures for patient safety and to prioritize the care treatments for patients with COVID?19.
The reduction in the volume of elective surgical procedures on a large scale had a substantial impact on patients as well as a financial impact on the medical device manufacturers. This resulted in the decreased sales of the minimally invasive surgical systems.
Market Segmentation
Minimally Invasive Surgical Systems Market (by Product Type)
Under this segmentation, the market in divided into conventional technologies and surgical robotics.
The conventional technologies segment dominates the market with a share of 80.4% of global minimally invasive surgical systems market mainly due to the increase in the number of minimally invasive surgical procedures.
Minimally Invasive Surgical Systems Market (by Application)
Under this segmentation, the market in divided into general surgery, urology surgery, orthopedic surgery, cardiovascular surgery, neurosurgery, others (cosmetic and bariatric surgery)
These above-mentioned applications are based on conventional technologies and surgical robotics segment.
Orthopedic surgery under the conventional technology segment is expected to register the highest CAGR of 7.0% during the forecast period.
Minimally Invasive Surgical Systems Market (by End-User)
Under this segmentation, the market in divided into hospitals, ambulatory surgical centers (ABCs), specialty clinics.
The hospitals segment dominates the minimally invasive surgical systems market with a share of 73.3% of global market mainly due to the number of hospitals providing access to the surgical treatments for the patients.
Ambulatory surgical centers (ASCs) segment is expected to register the highest CAGR of 8.0% during the forecast period.
Minimally Invasive Surgical Systems Market (by Region)
The different regions covered under the market report includes North America, Europe, Asia-Pacific, Latin America, and Middle East and Africa.
North America dominates the market in 2020 and is anticipated to uphold its dominance throughout the forecast period. The growth in the market is majorly driven by the increasing research and development activities and many companies being located in the U.S.
Latin America, and Middle East and Africa market is expected to grow with the highest CAGR of 7.8% during the forecast period.
Key Market Players and Competition Synopsis
Some of the key players operating in the market, include Arthrex Inc., Asensus Surgical Inc. (formerly TransEnterix Inc.), Boston Scientific Corporation, Conmed Corporation, Fujifilm Holdings Corporation, Intuitive Surgical Inc., Karl Storz SE & Co. KG, Medtronic plc, Olympus Corporation, Hoya Corporation (Pentax Medical), Siemens Healthineers AG, Smith & Nephew plc, Sony Corporation, Stryker Corporation, Zimmer Biomed Holdings Inc.
The minimally invasive surgical systems market market has witnessed several strategic and technological developments from January 2017- July 2021, undertaken by the different market players to attain their respective market shares in this emerging domain. Some of the strategies covered in this segment are product development and upgradations, partnerships, collaborations, business expansions, funding activities, regulatory approvals, mergers, and acquisitions. The preferred strategy for companies has been partnerships, collaborations, and business expansions.
Table of Contents
1 Market Definition and Scope of Study
1.1 Market Definition
1.2 Scope of Study
1.3 Market Segmentation
1.4 Key Questions Answered
1.5 Forecast Period Selection Criteria
2 Markets
2.1 Global Market Outlook
2.1.1 Key Findings and Opportunity Assessment
2.1.2 Assumptions and Limitations
2.1.3 Key Vendors
2.2 Key Enabling Technologies
2.2.1 Rising Adoption of 4K Imaging Technologies
2.2.2 Haptics in Minimally Invasive Surgical Technologies
2.2.3 Artificial Intelligence in Minimally Invasive Surgical Technologies
2.2.4 The Advent of Miniature Minimally Invasive Surgical Instruments
2.3 Business Model Analysis
2.4 Patent Landscape
2.4.1 Minimally Invasive Conventional Technologies
2.4.2 Minimally Invasive Surgical Robotics
2.4.3 Patent Expiration Impact
2.5 Market Dynamics
2.5.1 Overview
2.5.2 Impact Analysis
2.5.2.1 Short-Term
2.5.2.2 Long-Term
2.5.3 Market Drivers
2.5.3.1 Rising Incidence of Chronic Diseases
2.5.3.2 Advantages Over Open Surgeries
2.5.3.3 Increase in Global Geriatric Population
2.5.3.4 Increasing Healthcare Costs
2.5.3.5 Technological Advancement in Minimally Invasive Surgical Procedures and Technologies
2.5.4 Market Restraints
2.5.4.1 High Cost of Minimally Invasive Surgical Robotic Systems
2.5.4.2 Shortage of Skilled Professionals
2.5.4.3 Restrictive Reimbursement Landscape
2.5.5 Market Opportunities
2.5.5.1 Engage in Mergers and Acquisitions to Diversify Endoscopic Portfolio
2.5.5.2 Development of Surgical Simulators for Training Professionals
2.5.5.3 Development of Low-Cost Surgical Robotic Systems
2.5.6 Market Trends
2.5.6.1 Operating Lease Programs Implemented by Surgical Robotic Manufacturers
2.6 Industry Opportunity Analysis
2.7 Analyst Perspective
3 Impact of COVID-19 Pandemic on Minimally Invasive Surgical Systems
3.1 Pre-COVID-19 Scenario
3.1.1 Market Size and Growth Scenarios
3.1.1.1 Global and Regional Analysis
3.1.2 Procedure Volume
3.1.3 Demand and Supply
3.1.4 Opportunities
3.1.5 Business Model
3.2 During COVID-19 Scenario
3.2.1 Market Size and Growth Scenarios
3.2.1.1 Global and Regional Analysis
3.2.2 Procedure Volume
3.2.2.1 Case Study –Various Phases of Elective Surgeries During COVID-19
3.2.3 Demand and Supply
3.2.4 Opportunities
3.2.5 Business Model
3.3 Future Impact of COVID-19
3.3.1 Market Size and Growth Potential
3.3.1.1 Global and Regional Markets
3.3.2 Impact on Demand and Supply
3.3.3 Impact on Supply Chain and Logistics
3.3.4 Impact on Procedure Volume
3.3.5 Impact on Hospital Procurement
3.3.5.1 Case Study – Navigating Through the Pandemic: An Assessment of Weill Cornell Medicine
3.3.6 Impact on Technology Diffusion
3.3.7 Impact on Export and Import
3.3.8 Impact on Competitive Landscape
3.3.9 Impact on Entry Barriers and Opportunities for New Companies
3.4 Market Scenarios
3.4.1 Optimistic Scenario
3.4.2 Realistic Scenario
3.4.3 Pessimistic Scenario
4 Supply Chain
4.1 Key Entities of the Supply Chain
4.1.1 Manufacturers/Original Equipment Manufacturers (OEMs)
4.1.1.1 Manufacturers’ Disruptions
4.1.1.1.1 Electronic Parts
4.1.1.1.2 Chips
4.1.1.1.3 Sensors
4.1.1.1.4 Other Parts
4.1.2 Contract Manufacturers
4.1.2.1 Contract Manufacturers’ Disruptions
4.1.2.1.1 Electronic Parts
4.1.2.1.2 Chips
4.1.2.1.3 Sensors
4.1.2.1.4 Other Parts
4.2 Distribution Challenges
4.3 End Users’ Access Limitations of Regular Supplies
4.4 Supply Chain Analysis
4.4.1 Pre-COVID
4.4.2 During COVID-19
4.4.3 Post-COVID-19
4.5 Post-COVID-19 Scenarios of Supply Chain
4.5.1 Optimistic
4.5.2 Realistic
4.5.3 Pessimistic
5 Application
5.1 Global Minimally Invasive Surgical Systems Market (by Application)
5.1.1 Overview
5.1.2 Opportunity Assessment
5.1.2.1 During COVID-19
5.1.2.2 Post-COVID-19
5.1.3 Market Attractiveness Analysis
5.1.3.1 During COVID-19
5.1.3.2 Post-COVID-19
5.1.4 Market Size and Forecast
5.1.4.1 Minimally Invasive Conventional Technologies (Optimistic and Realistic Scenarios)
5.1.4.1.1 General Surgery
5.1.4.1.2 Urology Surgery
5.1.4.1.3 Gynecology Surgery
5.1.4.1.4 Orthopedic Surgery
5.1.4.1.5 Cardiovascular Surgery
5.1.4.1.6 Neurosurgery
5.1.4.1.7 Others
5.1.4.2 Minimally Invasive Surgical Robotics
5.1.4.2.1 General Surgery
5.1.4.2.2 Urology Surgery
5.1.4.2.3 Gynecology Surgery
5.1.4.2.4 Orthopedic Surgery
5.1.4.2.5 Cardiovascular Surgery
5.1.4.2.6 Neurosurgery
5.1.4.2.7 Others
5.2 Global Minimally Invasive Surgical Systems Market (by End User)
5.2.1 Overview
5.2.2 Opportunity Assessment
5.2.3 Market Attractiveness Analysis
5.2.4 Market Size and Forecast
5.2.4.1 Minimally Invasive Conventional Technologies
5.2.4.1.1 Hospitals
5.2.4.1.2 Ambulatory Surgical Centers (ASCs)
5.2.4.1.3 Specialty Clinics
5.2.4.2 Minimally Invasive Surgical Robotics
5.2.4.2.1 Hospitals
5.2.4.2.2 Ambulatory Surgical Centers (ASCs)
5.2.4.2.3 Specialty Clinics
6 Product
6.1 Global Minimally Invasive Surgical Systems Market (by Product Type)
6.1.1 Overview
6.1.2 Opportunity Assessment
6.1.2.1 During COVID-19
6.1.2.2 Post-COVID-19
6.1.3 Market Attractiveness Analysis
6.1.3.1 During COVID-19
6.1.3.2 Post-COVID-19
6.1.4 Market Size and Forecast
6.1.4.1 Minimally Invasive Conventional Technologies
6.1.4.1.1 Endoscopes and Endoscopic Systems
6.1.4.1.1.1 Rigid Endoscopes
6.1.4.1.1.1.1 Market Size and Growth Potential
6.1.4.1.1.1.2 Laparoscopes
6.1.4.1.1.1.3 Urology Endoscopes
6.1.4.1.1.1.4 Gynecology Endoscopes
6.1.4.1.1.1.5 ENT Endoscopes
6.1.4.1.1.1.6 Arthroscopes
6.1.4.1.1.1.7 Neuroendoscopes
6.1.4.1.1.1.8 Other Rigid Endoscopes
6.1.4.1.1.2 Flexible Endoscopes
6.1.4.1.1.2.1 Market Size and Growth Potential
6.1.4.1.1.2.2 Gastrointestinal Flexible Endoscopes
6.1.4.1.1.2.3 Urology Flexible Endoscopes
6.1.4.1.1.2.4 Gynecology Flexible Endoscopes
6.1.4.1.1.2.5 Bronchoscopes
6.1.4.1.1.2.6 Laryngoscopes
6.1.4.1.1.2.7 Pharyngoscopes
6.1.4.1.1.2.8 ENT Endoscopes
6.1.4.1.2 Video and Visualization Systems
6.1.4.1.2.1 Displays and Monitors
6.1.4.1.2.1.1 Market Size and Growth Potential
6.1.4.1.2.1.2 Wireless Displays and Monitors
6.1.4.1.2.1.3 Wired Displays and Monitors
6.1.4.1.2.2 Cameras
6.1.4.1.2.2.1 Market Size and Growth Potential
6.1.4.1.2.2.2 Single-Chip Cameras
6.1.4.1.2.2.3 3-Chip Cameras
6.1.4.1.2.3 Light Sources
6.1.4.1.2.3.1 Market Size and Growth Potential
6.1.4.1.2.3.2 Xenon
6.1.4.1.2.3.3 Halogen
6.1.4.1.2.3.4 LED
6.1.4.1.2.3.5 Laser
6.1.4.1.2.4 Video Recorders
6.1.4.1.2.4.1 Market Size and Growth Potential
6.1.4.1.2.4.2 Inbuilt
6.1.4.1.2.4.3 Standalone
6.1.4.1.2.5 Camera Heads
6.1.4.1.2.5.1 Market Size and Growth Potential
6.1.4.1.2.5.2 Charged Couple Device (CCD)
6.1.4.1.2.5.3 Complementary Metal Oxide Semiconductor (CMOS)
6.1.4.1.2.6 Video Processors
6.1.4.1.2.6.1 Optimistic
6.1.4.1.2.6.2 Realistic
6.1.4.1.2.6.3 Pessimistic
6.1.4.1.2.7 Printers
6.1.4.1.2.8 Video Converters
6.1.4.1.3 Instruments and Accessories
6.1.4.1.3.1 Insufflators
6.1.4.1.3.2 Energy Devices
6.1.4.1.3.3 Closure Devices
6.1.4.1.3.4 Irrigation Systems
6.1.4.1.3.5 Access Devices
6.1.4.1.3.6 Hand Instruments
6.1.4.1.3.7 Procedure Kits
6.1.4.1.3.8 Endoscope Tip Protectors, Tubing, and Water Systems
6.1.4.1.3.9 Forceps
6.1.4.1.3.10 Retrieval Kits
6.1.4.1.3.11 Injection Needles
6.1.4.1.3.12 Other Instruments and Accessories
6.1.4.1.4 Others (Reprocessing and Sterilization)
6.1.4.1.5 Capsule Endoscopy
6.1.4.1.5.1 Workstations and Data Recorders
6.1.4.1.5.2 Capsules
6.1.4.1.5.3 Others (Services and Patency System)
6.1.4.2 Minimally Invasive Surgical Robotics
6.1.4.2.1 Robotic Systems
6.1.4.2.1.1 Optimistic Scenario
6.1.4.2.1.2 Realistic Scenario
6.1.4.2.1.3 Pessimistic Scenario
6.1.4.2.2 Instruments and Accessories (Robotic Surgery)
6.1.4.2.2.1 Optimistic Scenario
6.1.4.2.2.2 Realistic Scenario
6.1.4.2.2.3 Pessimistic Scenario
6.1.4.2.3 Services
6.1.4.2.3.1 Optimistic Scenario
6.1.4.2.3.2 Realistic Scenario
6.1.4.2.3.3 Pessimistic Scenario
7 Region
7.1 Overview
7.2 Opportunity Assessment
7.2.1.1 During COVID-19
7.2.1.2 Post-COVID-19
7.3 Market Attractiveness Analysis
7.3.1.1 During COVID-19
7.3.1.2 Post-COVID-19
7.4 Supply Chain Disruption
7.4.1 North America
7.4.2 Europe
7.4.3 Asia-Pacific
7.4.4 Rest-of-the-World
7.5 North America
7.5.1 Overview
7.5.2 Regulatory Framework (U.S.)
7.5.3 Opportunity Assessment
7.5.4 Key Vendors
7.5.5 Market Size and Forecast
7.5.5.1 North America Minimally Invasive Surgical Systems Market (by Product Type)
7.5.5.1.1 North America Minimally Invasive Conventional Technologies Market
7.5.5.1.1.1 Optimistic
7.5.5.1.1.2 Realistic
7.5.5.1.1.3 Pessimistic
7.5.5.1.2 North America Minimally Invasive Surgical Robotics
7.5.5.1.2.1 Optimistic
7.5.5.1.2.2 Realistic
7.5.5.1.2.3 Pessimistic
7.5.5.2 North America Minimally Invasive Surgical Systems Market (by Country)
7.5.5.2.1 U.S.
7.5.5.2.1.1 U.S. Minimally Invasive Surgical Systems Market (by Product Type)
7.5.5.2.2 Canada
7.5.5.2.2.1 Canada Minimally Invasive Surgical Systems Market (by Product Type)
7.6 Europe
7.6.1 Overview
7.6.2 Regulatory Framework (Europe)
7.6.2.1 Impact of the EU’s Medical Device Regulations (MDR)
7.6.3 Opportunity Assessment
7.6.4 Key Vendors
7.6.5 Market Size and Forecast
7.6.5.1 Europe Minimally Invasive Surgical Systems (by Product Type)
7.6.5.1.1 Europe Minimally Invasive Conventional Technologies Market
7.6.5.1.1.1 Optimistic
7.6.5.1.1.2 Realistic
7.6.5.1.1.3 Pessimistic
7.6.5.1.2 Europe Minimally Invasive Surgical Robotics Market
7.6.5.1.2.1 Optimistic
7.6.5.1.2.2 Realistic
7.6.5.1.2.3 Pessimistic
7.6.5.2 Europe Minimally Invasive Surgical Systems Market (by Country)
7.6.5.2.1 Germany
7.6.5.2.1.1 Germany Minimally Invasive Surgical Systems Market (by Product Type)
7.6.5.2.2 France
7.6.5.2.2.1 France Minimally Invasive Surgical Systems Market (by Product Type)
7.6.5.2.3 U.K.
7.6.5.2.3.1 U.K. Minimally Invasive Surgical Systems Market (by Product Type)
7.6.5.2.4 Italy
7.6.5.2.4.1 Italy Minimally Invasive Surgical Systems Market (by Product Type)
7.6.5.2.5 Spain
7.6.5.2.5.1 Spain Minimally Invasive Surgical Systems Market (by Product Type)
7.6.5.2.6 Netherlands
7.6.5.2.6.1 Netherlands Minimally Invasive Surgical Systems Market (by Product Type)
7.6.5.2.7 Belgium
7.6.5.2.7.1 Belgium Minimally Invasive Surgical Systems Market (by Product Type)
7.6.5.2.8 Switzerland
7.6.5.2.8.1 Switzerland Minimally Invasive Surgical Systems Market (by Product Type)
7.6.5.2.9 Sweden
7.6.5.2.9.1 Sweden Minimally Invasive Surgical Systems Market (by Product Type)
7.6.5.2.10 Denmark
7.6.5.2.10.1 Denmark Minimally Invasive Surgical Systems Market (by Product Type)
7.6.5.2.11 Rest-of-Europe
7.6.5.2.11.1 Rest-of-Europe Minimally Invasive Surgical Systems Market (by Product Type)
7.7 Asia-Pacific
7.7.1 Overview
7.7.2 Regulatory Framework
7.7.2.1 China
7.7.2.2 Japan
7.7.3 Opportunity Assessment
7.7.4 Key Vendors
7.7.5 Market Size and Forecast
7.7.5.1 Asia-Pacific Minimally Invasive Surgical Systems Market (by Product Type)
7.7.5.1.1 Asia-Pacific Minimally Invasive Conventional Technologies Market
7.7.5.1.1.1 Optimistic
7.7.5.1.1.2 Realistic
7.7.5.1.1.3 Pessimistic
7.7.5.1.2 Asia-Pacific Minimally Invasive Surgical Robotics Market
7.7.5.1.2.1 Optimistic
7.7.5.1.2.2 Realistic
7.7.5.1.2.3 Pessimistic
7.7.5.2 Asia-Pacific Minimally Invasive Surgical Systems Market (by Country)
7.7.5.2.1 Japan
7.7.5.2.1.1 Japan Minimally Invasive Surgical Systems Market (by Product Type)
7.7.5.2.2 China
7.7.5.2.2.1 China Minimally Invasive Surgical Systems Market (by Product Type)
7.7.5.2.3 India
7.7.5.2.3.1 India Minimally Invasive Surgical Systems Market (by Product Type)
7.7.5.2.4 South Korea
7.7.5.2.4.1 South Korea Minimally Invasive Surgical Systems Market (by Product Type)
7.7.5.2.5 Australia and New Zealand
7.7.5.2.5.1 Australia and New Zealand Minimally Invasive Surgical Systems Market (by Product Type)
7.7.5.2.6 Malaysia
7.7.5.2.6.1 Malaysia Minimally Invasive Surgical Systems Market (by Product Type)
7.7.5.2.7 Singapore
7.7.5.2.7.1 Singapore Minimally Invasive Surgical Systems Market (by Product Type)
7.7.5.2.8 Rest-of-Asia-Pacific
7.7.5.2.8.1 Rest-of-Asia-Pacific Minimally Invasive Surgical Systems Market (by Product Type)
7.8 Latin America
7.8.1 Overview
7.8.2 Opportunity Assessment
7.8.3 Key Vendors
7.8.4 Market Size and Forecast
7.8.4.1 Latin America Minimally Invasive Surgical Systems Market (by Product Type)
7.8.4.1.1 Latin America Minimally Invasive Conventional Technologies Market
7.8.4.1.1.1 Optimistic
7.8.4.1.1.2 Realistic
7.8.4.1.1.3 Pessimistic
7.8.4.1.2 Latin America Minimally Invasive Surgical Robotics Market
7.8.4.1.2.1 Optimistic
7.8.4.1.2.2 Realistic
7.8.4.1.2.3 Pessimistic
7.8.4.2 Latin America Minimally Invasive Surgical Systems Market (by Country)
7.8.4.2.1 Brazil
7.8.4.2.1.1 Brazil Minimally Invasive Surgical Systems Market (by Product Type)
7.8.4.2.2 Mexico
7.8.4.2.2.1 Mexico Minimally Invasive Surgical Systems Market (by Product Type)
7.8.4.2.3 Argentina
7.8.4.2.3.1 Argentina Minimally Invasive Surgical Systems Market (by Product Type)
7.8.4.2.4 Rest-of-Latin America
7.8.4.2.4.1 Rest-of-Latin America Minimally Invasive Surgical Systems Market (by Product Type)
7.9 Middle East and Africa
7.9.1 Overview
7.9.2 Opportunity Assessment
7.9.3 Key Vendors
7.9.4 Market Size and Forecast
7.9.4.1 Middle East and Africa Minimally Invasive Surgical Systems Market (by Product Type)
7.9.4.1.1 Middle East and Africa Minimally Invasive Conventional Technologies Market
7.9.4.1.1.1 Optimistic
7.9.4.1.1.2 Realistic
7.9.4.1.1.3 Pessimistic
7.9.4.1.2 Middle East and Africa Minimally Invasive Surgical Robotics Market
7.9.4.1.2.1 Optimistic
7.9.4.1.2.2 Realistic
7.9.4.1.2.3 Pessimistic
7.9.4.2 By Country
7.9.4.2.1 K.S.A.
7.9.4.2.1.1 K.S.A. Minimally Invasive Surgical Systems Market (by Product Type)
7.9.4.2.2 U.A.E.
7.9.4.2.2.1 U.A.E. Minimally Invasive Surgical Systems Market (by Product Type)
7.9.4.2.3 Israel
7.9.4.2.3.1 Israel Minimally Invasive Surgical Systems Market (by Product Type)
7.9.4.2.4 Rest-of-Middle East and Africa
7.9.4.2.4.1 Rest-of-Middle East and Africa Minimally Invasive Surgical Systems Market (by Product Type)
8 Competitive Benchmarking & Company Profiles
8.1 Competitive Benchmarking
8.1.1 Market Share Analysis
8.1.1.1 Minimally Invasive Conventional Technologies
8.1.1.2 Minimally Invasive Surgical Robotics
8.1.2 Key Strategies and Developments (Deal Tracking)
8.1.2.1 Minimally Invasive Conventional Technologies
8.1.2.1.1 Partnerships, Alliances, and Business Expansion
8.1.2.1.2 New Offerings
8.1.2.1.3 Mergers and Acquisitions
8.1.2.1.4 Regulatory and Legal Activities
8.1.2.1.5 Funding Activities
8.1.2.2 Minimally Invasive Surgical Robotics
8.1.2.2.1 Partnerships, Alliances, and Business Expansions
8.1.2.2.2 New Offerings and Upgradations
8.1.2.2.3 Mergers and Acquisitions
8.1.2.2.4 Regulatory and Legal Activities
8.1.2.2.5 Funding Activities
8.1.3 Competitor Snapshot
8.2 Company Profiles
8.2.1 Overview
8.2.2 Arthrex Inc.
8.2.2.1 Company Outlook
8.2.2.2 Role of the Company
8.2.2.3 Product Portfolio
8.2.2.4 Competitive Analysis
8.2.2.4.1 Strengths of the Company
8.2.2.4.2 Weakness of the Company
8.2.3 Asensus Surgical, Inc.
8.2.3.1 Company Outlook
8.2.3.2 Role of the Company
8.2.3.3 Product Portfolio
8.2.3.4 Financials
8.2.3.5 Competitive Analysis
8.2.3.5.1 Strength of the Company
8.2.3.5.2 Weakness of the Company
8.2.4 Boston Scientific Corporation
8.2.4.1 Company Outlook
8.2.4.2 Role of the Company
8.2.4.3 Product Portfolio
8.2.4.4 Financials
8.2.4.5 Competitive Analysis
8.2.4.5.1 Strengths of the Company
8.2.4.5.2 Weakness of the Company
8.2.5 Conmed Corporation
8.2.5.1 Company Outlook
8.2.5.2 Role of the Company
8.2.5.3 Product Portfolio
8.2.5.4 Financials
8.2.5.5 Competitive Analysis
8.2.5.5.1 Strengths of the Company
8.2.5.5.2 Weaknesses of the Company
8.2.6 Fujifilm Holdings Corporation
8.2.6.1 Company Outlook
8.2.6.2 Role of the Company
8.2.6.3 Product Portfolio
8.2.6.4 Financials
8.2.6.5 Competitive Analysis
8.2.6.5.1 Strengths of the Company
8.2.6.5.2 Weakness of the Company
8.2.7 Intuitive Surgical, Inc.
8.2.7.1 Company Outlook
8.2.7.2 Role of the Company
8.2.7.3 Product Portfolio
8.2.7.4 Financials
8.2.7.5 Competitive Analysis
8.2.7.5.1 Strengths of the Company
8.2.7.5.2 Weaknesses of the Company
8.2.8 Karl Storz SE & Co. KG
8.2.8.1 Company Outlook
8.2.8.2 Role of the Company
8.2.8.3 Product Portfolio
8.2.8.4 Competitive Analysis
8.2.8.4.1 Strength of the Company
8.2.8.4.2 Weaknesses of the Company
8.2.9 Medtronic plc
8.2.9.1 Company Outlook
8.2.9.2 Role of the Company
8.2.9.3 Product Portfolio
8.2.9.4 Financials
8.2.9.5 Competitive Analysis
8.2.9.5.1 Strengths of the Company
8.2.9.5.2 Weakness of the Company
8.2.10 Olympus Corporation
8.2.10.1 Company Outlook
8.2.10.2 Role of the Company
8.2.10.3 Product Portfolio
8.2.10.4 Financials
8.2.10.5 Competitive Analysis
8.2.10.5.1 Strengths of the Company
8.2.10.5.2 Weaknesses of the Company
8.2.11 Hoya Corporation (Pentax Medical)
8.2.11.1 Company Outlook
8.2.11.2 Role of the Company
8.2.11.3 Product Portfolio
8.2.11.4 Financials
8.2.11.5 Competitive Analysis
8.2.11.5.1 Strengths of the Company
8.2.11.5.2 Weakness of the Company
8.2.12 Siemens Healthineers AG
8.2.12.1 Company Outlook
8.2.12.2 Role of the Company
8.2.12.3 Product Portfolio
8.2.12.4 Financials
8.2.12.5 Competitive Analysis
8.2.12.5.1 Strengths of the Company
8.2.12.5.2 Weakness of the Company
8.2.13 Smith & Nephew plc
8.2.13.1 Company Outlook
8.2.13.2 Role of the Company
8.2.13.3 Product Portfolio
8.2.13.4 Financials
8.2.13.5 Competitive Analysis
8.2.13.5.1 Strengths of the Company
8.2.13.5.2 Weakness of the Company
8.2.14 Sony Corporation
8.2.14.1 Company Outlook
8.2.14.2 Role of the Company
8.2.14.3 Product Portfolio
8.2.14.4 Financials
8.2.14.5 Competitive Analysis
8.2.14.5.1 Strengths of the Company
8.2.14.5.2 Weakness of the Company
8.2.15 Stryker Corporation
8.2.15.1 Company Outlook
8.2.15.2 Role of the Company
8.2.15.3 Product Portfolio
8.2.15.4 Financials
8.2.15.5 Competitive Analysis
8.2.15.5.1 Strength of the Company
8.2.15.5.2 Weaknesses of the Company
8.2.16 Zimmer Biomet Holdings, Inc.
8.2.16.1 Company Outlook
8.2.16.2 Role of the Company
8.2.16.3 Product Portfolio
8.2.16.4 Financials
8.2.16.5 Competitive Analysis
8.2.16.5.1 Strengths of the Company
8.2.16.5.2 Weakness of the Company
9 Research Methodology
9.1 Data Collection and Analysis
9.2 Data Sources
9.2.1 Primary Data Sources
9.2.2 Secondary Data Sources
9.3 Data Sources and Categorization
9.4 Data Triangulation
9.5 Estimation and Forecasting Methodology
List of Figures
Figure 1: Evolution of Surgery
Figure 2: Growing Prominence of Minimally Invasive Surgical Procedures, 2018 and 2030
Figure 3: Global Minimally Invasive Surgical Systems Market Growth, 2020-2031
Figure 4: Key Barriers for Start-ups Looking to Enter the Market
Figure 5: Leading Players in the Global Minimally Invasive Surgical Robotics Market (2020)
Figure 6: Leading Players in the Global Conventional Technologies Market (2020)
Figure 7: Global Minimally Invasive Surgical Systems Market Growth (by Region), $Million, 2020-2031
Figure 8: Comparative Analysis: Incision Length for Various Types of Surgery
Figure 9: Global Minimally Invasive Surgical Systems Market Segmentation
Figure 10: Regional Minimally Invasive Surgical Systems Market Segmentation
Figure 11: Global Minimally Invasive Surgical Systems Market Forecast, $Million, 2020-2031
Figure 12: Global Minimally Invasive Surgical Systems Market Size and Forecast (by Product Type), $Million, 2020-2031
Figure 13: Global Minimally Invasive Surgical Systems Market Incremental Revenue Opportunity, 2020-2031
Figure 14: Advantages of 4K Medical Imaging
Figure 15: Artificial Intelligence (AI), Machine Learning (ML), and Deep Learning (DL)
Figure 16: Impact - Convergence of AI and Minimally Invasive Surgical Robotics
Figure 17: Technological Evolution toward Autonomous Surgical Systems
Figure 18: Shortfall of Surgical Specialists and Surgical Procedure Volume in In-Patient and Out-Patient Facilities (U.S.), 2018
Figure 19: Leveraging Technology to Access Hard-to-Reach Organs
Figure 20: Rapidly Increasing Patient Pool of Chronic Conditions (U.S.)
Figure 21: Comparison: Robotic Prostatectomy Vs. Open Prostatectomy
Figure 22: Increasing Prevalence of Diseases (by Age Group), Million, 2008-2020
Figure 23: Average Quoted Prices: da Vinci Surgical Robot Models (2014-2015)
Figure 24: Factors Contributing to Adverse Events in Robotic-Assisted Surgical Procedures
Figure 25: Intuitive Surgical, Inc.: Installed Base and Revenue Under Operating Lease Program, 2018-2020
Figure 26: Global Minimally Invasive Surgical Systems Market, Procedures, Volume, 2018-2030
Figure 27: Global Minimally Invasive Surgical Systems Market Y-o-Y Growth Comparative Analysis (With Vs. Without COVID-19)
Figure 28: Minimally Invasive Surgeries Volume – Pre-COVID-19 Scenario, 2019, 2025, and 2030
Figure 29: Global Minimally Invasive Surgical Systems Market Opportunities Impact Analysis (Pre-COVID-19 Scenario)
Figure 30: Minimally Invasive Surgical Systems Market, Key Business Models (Pre-COVID-19 Scenario)
Figure 31: During COVID-19 Impact Trends and Scenarios (by Region), 2019-2021
Figure 32: Number of Elective Surgical Procedures Canceled due to COVIDâ€19 (by Region)
Figure 33: Average Number of Procedures Performed Per Week (Veterans Affairs Hospitals), 2020
Figure 34: Procedure Recovery (by Organ System), 2020
Figure 35: Global Minimally Invasive Surgical Systems Market Focus Areas (During COVID-19 Scenario)
Figure 36: Minimally Invasive Surgical Systems Market, Business Models (During-COVID-19 Scenario)
Figure 37: COVID-19 Impact Trends and Forecast Scenarios (by Region), 2019-2030
Figure 38: Global Minimally Invasive Surgical Systems Market Future Demand and Supply Impact Analysis
Figure 39: Global Minimally Invasive Surgical Systems Market Supply Chain Disruption
Figure 40: Percentage of U.S. Hospitals with Negative Margins, 2021
Figure 41: Changes in Expense of U.S. Hospitals Compared to Pre-Pandemic Levels
Figure 42: Percent of Hospitals with Negative Operating Margins (by Scenario), 2021
Figure 43: Global Minimally Invasive Surgical Systems Market, Key Technologies
Figure 44: Global Minimally Invasive Surgical Systems Market Growth Scenarios, 2019-2031
Figure 45: Global Minimally Invasive Surgical Systems Market Size and Growth Potential (Optimistic Scenario), 2020-2031
Figure 46: Global Minimally Invasive Surgical Systems Market Size and Growth Potential (Realistic Scenario), 2020-2031
Figure 47: Global Minimally Invasive Surgical Systems Market Size and Growth Potential (Pessimistic Scenario), 2020-2031
Figure 48: Global Minimally Invasive Surgical Systems Market, Supply Chain Disruptions Impact Analysis (Manufacturer), 2020-2021
Figure 49: Global Minimally Invasive Surgical Systems Market, Supply Chain Disruptions Impact Analysis (Contract Manufacturers), 2020-2021
Figure 50: Global Minimally Invasive Surgical Systems Market Supply Chain Impact
Figure 51: Global Minimally Invasive Surgical Systems Market, Key Measures, 2020-2021
Figure 52: COVID-19 Cases Per Million (by Key Countries), 2020-2021
Figure 53: Global Minimally Invasive Surgical Systems Market Future Supply Chain
Figure 54: Global Minimally Invasive Surgical Systems Supply Chain, Future Evolutionary Scenarios, 2021-2030
Figure 55: Global Minimally Invasive Surgical Systems Market Segmentation (by Application)
Figure 56: Global Minimally Invasive Surgical Systems Market Incremental Revenue Opportunity Analysis (by Application), $Million, 2020-2022
Figure 57: Global Minimally Invasive Surgical Systems Market Incremental Revenue Opportunity Analysis (by Application), $Million, 2022-2031
Figure 58: Market Attractiveness Analysis - Global Minimally Invasive Surgical Systems Market (by Application), 2020-2022
Figure 59: Market Attractiveness Analysis - Global Minimally Invasive Surgical Systems Market (by Application), 2022-2031
Figure 60: Global Minimally Invasive Surgical Systems Market (by Application), $Million, 2020 and 2031
Figure 61: Global Minimally Invasive Conventional Technologies Market Share (by Application), $Million, 2020 and 2031
Figure 62: Global Minimally Invasive Surgical Systems Market Optimistic and Realistic Growth Scenario Considerations
Figure 63: Global Minimally Invasive Conventional Technologies Market Optimistic and Realistic Forecast Scenarios (General Surgery), $Million, 2020-2031
Figure 64: Global Minimally Invasive Conventional Technologies Market Optimistic and Realistic Forecast Scenarios (Urology Surgery), $Million, 2020-2031
Figure 65: Global Minimally Invasive Conventional Technologies Market Optimistic and Realistic Forecast Scenarios (Gynecology Surgery), $Million, 2020-2031
Figure 66: Global Minimally Invasive Conventional Technologies Market Optimistic and Realistic Forecast Scenarios (Orthopedic Surgery), $Million, 2020-2031
Figure 67: Global Minimally Invasive Conventional Technologies Market Optimistic and Realistic Forecast Scenarios (Cardiovascular Surgery), $Million, 2020-2031
Figure 68: Global Minimally Invasive Conventional Technologies Market Optimistic and Realistic Forecast Scenarios (Neurosurgery), $Million, 2020-2031
Figure 69: Global Minimally Invasive Conventional Technologies Market Optimistic and Realistic Scenarios (Cosmetic and Bariatric Surgery), $Million, 2020-2031
Figure 70: Global Minimally Invasive Surgical Robotics Market Share (by Application), $Million, 2020 and 2031
Figure 71: Global Minimally Invasive Surgical Robotics Market Optimistic and Realistic Scenarios (General Surgery), $Million, 2020-2031
Figure 72: Global Minimally Invasive Surgical Robotics Market Optimistic and Realistic Forecast Scenarios (Urology Surgery), $Million, 2020-2031
Figure 73: Global Minimally Invasive Surgical Robotics Market Optimistic and Realistic Forecast Scenarios (Gynecology Surgery), $Million, 2020-2031
Figure 74: Global Minimally Invasive Surgical Robotics Market Optimistic and Realistic Forecast Scenarios (Orthopedic Surgery), $Million, 2020-2031
Figure 75: Global Minimally Invasive Surgical Robotics Market Optimistic and Realistic Forecast Scenarios (Cardiovascular Surgery), $Million, 2020-2031
Figure 76: Global Minimally Invasive Surgical Robotics Market Optimistic and Realistic Forecast Scenarios (Neurosurgery), $Million, 2020-2031
Figure 77: Global Minimally Invasive Surgical Robotics Market Optimistic and Realistic Forecast Scenarios (Others), $Million, 2020-2031
Figure 78: Global Minimally Invasive Surgical Systems Market (by End User)
Figure 79: Global Minimally Invasive Surgical Systems Market Incremental Revenue Opportunity (by End User), $Million, 2020-2031
Figure 80: Market Attractiveness Analysis - Global Minimally Invasive Surgical Systems Market (by End User), 2020-2031
Figure 81: Global Minimally Invasive Surgical Systems Market (by End User), $Million, 2020-2031
Figure 82: Global Minimally Invasive Conventional Technologies Market Share (by End User), $Million, 2020 and 2031
Figure 83: Global Minimally Invasive Conventional Technologies Market (Hospitals), $Million, 2020-2031
Figure 84: Global Minimally Invasive Conventional Technologies Market (Ambulatory Surgical Centers), $Million, 2020-2031
Figure 85: Global Minimally Invasive Conventional Technologies Market (Specialty Clinics), $Million, 2020-2031
Figure 86: Global Minimally Invasive Surgical Robotics Market Share (by End User), $Million, 2020 and 2031
Figure 87: Global Minimally Invasive Surgical Robotics Market (Hospitals), $Million, 2020-2031
Figure 88: Global Minimally Invasive Surgical Robotics Market (Ambulatory Surgical Centers), $Million, 2020-2031
Figure 89: Global Minimally Invasive Surgical Robotics Market (Specialty Clinics), $Million, 2020-2031
Figure 90: Global Minimally Invasive Surgical Systems Market Segmentation (by Product Type)
Figure 91: Global Minimally Invasive Surgical Systems Market Incremental Revenue Opportunity Analysis (by Product), $Million, 2020-2022
Figure 92: Global Minimally Invasive Surgical Systems Market Incremental Revenue Opportunity Analysis (by Product), $Million, 2022-2031
Figure 93: Market Attractiveness Analysis - Global Minimally Invasive Surgical Systems Market (by Product Type), 2020-2022
Figure 94: Market Attractiveness Analysis - Global Minimally Invasive Surgical Systems Market (by Product Type), 2022-2031
Figure 95: Global Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Figure 96: Global Minimally Invasive Surgical Systems Market Share (by Product Type), $Million, 2020 and 2031
Figure 97: Global Minimally Invasive Conventional Technologies Market Share (by Product Type), $Million, 2020 and 2031
Figure 98: Market Segmentation - Endoscopes and Endoscopic Systems
Figure 99: Global Minimally Invasive Conventional Technologies Market Share (by Endoscopes and Endoscopic Systems), $Million, 2020 and 2031
Figure 100: Global Minimally Invasive Conventional Technologies Market (Endoscopes and Endoscopic Systems), $Million, 2020-2031
Figure 101: Market Segmentation - Rigid Endoscopes
Figure 102: Global Minimally Invasive Conventional Technologies Market Optimistic Scenario (Rigid Endoscopes), $Million, 2020-2031
Figure 103: Global Minimally Invasive Conventional Technologies Market Realistic Scenario (Rigid Endoscopes), $Million, 2020-2031
Figure 104: Global Minimally Invasive Conventional Technologies Market (by Rigid Endoscopes), $Million, 2020-2031
Figure 105: Global Minimally Invasive Conventional Technologies Market Pessimistic Scenario (Rigid Endoscopes), $Million, 2020-2031
Figure 106: Global Minimally Invasive Conventional Technologies Market (Laparoscopes), $Million, 2020 and 2031
Figure 107: Market Segmentation - Urology Endoscopes
Figure 108: Global Minimally Invasive Conventional Technologies Market (Urology Endoscopes), $Million, 2020-2031
Figure 109: Global Minimally Invasive Conventional Technologies Market Share (by Urology Endoscopes), $Million, 2020 and 2031
Figure 110: Global Minimally Invasive Conventional Technologies Market (Cystoscopes), $Million, 2020-2031
Figure 111: Global Minimally Invasive Conventional Technologies Market (Resectoscopes), $Million, 2020-2031
Figure 112: Global Minimally Invasive Conventional Technologies Market (Nephroscopes), $Million, 2020-2031
Figure 113: Global Minimally Invasive Conventional Technologies Market (Other Urology Endoscopes), $Million, 2020-2031
Figure 114: Global Minimally Invasive Conventional Technologies Market (Gynecology Endoscopes), $Million, 2020-2031
Figure 115: Global Minimally Invasive Conventional Technologies Market (ENT Endoscopes), $Million, 2020-2031
Figure 116: Global Minimally Invasive Conventional Technologies Market (Arthroscopes), $Million, 2020-2031
Figure 117: Global Minimally Invasive Conventional Technologies Market (Neuroendoscopes), $Million, 2020-2031
Figure 118: Global Minimally Invasive Conventional Technologies Market (Other Rigid Endoscopes), $Million, 2020-2031
Figure 119: Market Segmentation - Flexible Endoscopes
Figure 120: Global Minimally Invasive Conventional Technologies Market Optimistic Scenario (Flexible Endoscopes), $Million, 2020-2031
Figure 121: Global Minimally Invasive Conventional Technologies Market Realistic Scenario (Flexible Endoscopes), $Million, 2020-2031
Figure 122: Global Minimally Invasive Conventional Technologies Market Share (by Flexible Endoscopes), $Million, 2020 and 2031
Figure 123: Global Minimally Invasive Conventional Technologies Market Pessimistic Scenario (Flexible Endoscopes), $Million, 2020-2031
Figure 124: Market Segmentation - Gastrointestinal Flexible Endoscopes
Figure 125: Global Minimally Invasive Conventional Technologies Market (Gastrointestinal Flexible Endoscopes), $Million, 2020-2031
Figure 126: Global Minimally Invasive Conventional Technologies Market Share (by Gastrointestinal Flexible Endoscopes), $Million, 2020 and 2031
Figure 127: Global Minimally Invasive Conventional Technologies Market (Gastroscopes), $Million, 2020-2031
Figure 128: Global Minimally Invasive Conventional Technologies Market (Colonoscopes), $Million, 2020-2031
Figure 129: Global Minimally Invasive Conventional Technologies Market (Duodenoscopes), $Million, 2020-2031
Figure 130: Global Minimally Invasive Conventional Technologies Market (Sigmoidoscopes), $Million, 2020-2031
Figure 131: Global Minimally Invasive Conventional Technologies Market (Enteroscopes), $Million, 2020-2031
Figure 132: Global Minimally Invasive Conventional Technologies Market (Other GI Endoscopes), $Million, 2020-2031
Figure 133: Global Minimally Invasive Conventional Technologies Market (Urology Flexible Endoscopes), $Million, 2020-2031
Figure 134: Global Minimally Invasive Conventional Technologies Market (Gynecology Flexible Endoscopes), $Million, 2020-2031
Figure 135: Global Minimally Invasive Conventional Technologies Market (Bronchoscopes), $Million, 2020-2031
Figure 136: Global Minimally Invasive Conventional Technologies Market (Laryngoscopes), $Million, 2020-2031
Figure 137: Global Minimally Invasive Conventional Technologies Market (Pharyngoscopes), $Million, 2020-2031
Figure 138: Global Minimally Invasive Conventional Technologies Market (ENT Endoscopes), $Million, 2020-2031
Figure 139: Market Segmentation - Video and Visualization Systems
Figure 140: Global Minimally Invasive Conventional Technologies Market (Video and Visualization Systems), $Million, 2020-2031
Figure 141: Global Minimally Invasive Conventional Technologies Market Share (by Video and Visualization Systems), $Million, 2020 and 2031
Figure 142: Market Segmentation - Displays and Monitors
Figure 143: Global Minimally Invasive Conventional Technologies Market Optimistic Scenario (Displays and Monitors), $Million, 2020-2031
Figure 144: Global Minimally Invasive Conventional Technologies Market Realistic Scenario (Displays and Monitors), $Million, 2020-2031
Figure 145: Global Minimally Invasive Conventional Technologies Market Pessimistic Scenario (Displays and Monitors), $Million, 2020-2031
Figure 146: Global Minimally Invasive Conventional Technologies Market (Wireless Displays and Monitors), $Million, 2020-2031
Figure 147: Global Minimally Invasive Conventional Technologies Market (Wired Displays and Monitors), $Million, 2020-2031
Figure 148: Market Segmentation - Cameras
Figure 149: Global Minimally Invasive Conventional Technologies Market Optimistic Scenario (Cameras), $Million, 2020-2031
Figure 150: Global Minimally Invasive Conventional Technologies Market Realistic Scenario (Cameras), $Million, 2020-2031
Figure 151: Global Minimally Invasive Conventional Technologies Market Share (by Cameras), $Million, 2020 and 2031
Figure 152: Global Minimally Invasive Conventional Technologies Market Pessimistic Scenario (Cameras), $Million, 2020-2031
Figure 153: Global Minimally Invasive Conventional Technologies Market (Single-Chip Cameras), $Million, 2020-2031
Figure 154: Global Minimally Invasive Conventional Technologies Market (3-Chip Cameras), $Million, 2020-2031
Figure 155: Market Segmentation - Light Sources
Figure 156: Global Minimally Invasive Conventional Technologies Market Optimistic Scenario (Light Sources), $Million, 2020-2031
Figure 157: Global Minimally Invasive Conventional Technologies Market Realistic Scenario (Light Sources), $Million, 2020-2031
Figure 158: Global Minimally Invasive Conventional Technologies Market Share (by Light Sources), $Million, 2020 and 2031
Figure 159: Global Minimally Invasive Conventional Technologies Market Pessimistic Scenario (Light Sources), $Million, 2020-2031
Figure 160: Global Minimally Invasive Conventional Technologies Market (Xenon), $Million, 2020-2031
Figure 161: Global Minimally Invasive Conventional Technologies Market (Halogen), $Million, 2020-2031
Figure 162: Global Minimally Invasive Conventional Technologies Market (LED), $Million, 2020-2031
Figure 163: Global Minimally Invasive Conventional Technologies Market (Laser), $Million, 2020-2031
Figure 164: Market Segmentation - Video Recorders
Figure 165: Global Minimally Invasive Conventional Technologies Market Optimistic Scenario (Video Recorders), $Million, 2020-2031
Figure 166: Global Minimally Invasive Conventional Technologies Market Realistic Scenario (Video Recorders), $Million, 2020-2031
Figure 167: Global Minimally Invasive Conventional Technologies Market Share (by Video Recorders), $Million, 2020 and 2031
Figure 168: Global Minimally Invasive Conventional Technologies Market Pessimistic Scenario (Video Recorders), $Million, 2020-2031
Figure 169: Global Minimally Invasive Conventional Technologies Market (Inbuilt Video Recorders), $Million, 2020-2031
Figure 170: Global Minimally Invasive Conventional Technologies Market (Standalone Video Recorders), $Million, 2020-2031
Figure 171: Market Segmentation - Camera Heads
Figure 172: Global Minimally Invasive Conventional Technologies Market Optimistic Scenario (Camera Heads), $Million, 2020-2031
Figure 173: Global Minimally Invasive Conventional Technologies Market Realistic Scenario (Camera Heads), $Million, 2020-2031
Figure 174: Global Minimally Invasive Conventional Technologies Market Share (by Camera Heads), $Million, 2020 and 2031
Figure 175: Global Minimally Invasive Conventional Technologies Market Pessimistic Scenario (Camera Heads), $Million, 2020-2031
Figure 176: Global Minimally Invasive Conventional Technologies Market (CCD Camera Heads), $Million, 2020-2031
Figure 177: Global Minimally Invasive Conventional Technologies Market (CMOS Camera Heads), $Million, 2020-2031
Figure 178: Global Minimally Invasive Conventional Technologies Market Optimistic Scenario (Video Processors), $Million, 2020-2031
Figure 179: Global Minimally Invasive Conventional Technologies Market Realistic Scenario (Video Processors), $Million, 2020-2031
Figure 180: Global Minimally Invasive Conventional Technologies Market Pessimistic Scenario (Video Processors), $Million, 2020-2031
Figure 181: Global Minimally Invasive Conventional Technologies Market (Printers), $Million, 2020-2031
Figure 182: Global Minimally Invasive Conventional Technologies Market (Video Converters), $Million, 2020-2031
Figure 183: Market Segmentation - Instruments and Accessories
Figure 184: Global Minimally Invasive Conventional Technologies Market (Instruments and Accessories), $Million, 2020-2031
Figure 185: Global Minimally Invasive Conventional Technologies Market Share (by Instruments and Accessories), $Million, 2020 and 2031
Figure 186: Global Minimally Invasive Conventional Technologies Market (Insufflators), $Million, 2020-2031
Figure 187: Global Minimally Invasive Conventional Technologies Market (Energy Devices), $Million, 2020-2031
Figure 188: Global Minimally Invasive Conventional Technologies Market (Closure Devices), $Million, 2020-2031
Figure 189: Global Minimally Invasive Conventional Technologies Market (Irrigation Systems), $Million, 2020-2031
Figure 190: Global Minimally Invasive Conventional Technologies Market (Access Devices), $Million, 2020-2031
Figure 191: Global Minimally Invasive Conventional Technologies Market (Hand Instruments), $Million, 2020-2031
Figure 192: Global Minimally Invasive Conventional Technologies Market (Procedure Kits), $Million, 2020-2031
Figure 193: Global Minimally Invasive Conventional Technologies Market (Endoscope Tip Protectors, Tubing, and Water Systems), $Million, 2020-2031
Figure 194: Global Minimally Invasive Conventional Technologies Market (Forceps), $Million, 2020-2031
Figure 195: Global Minimally Invasive Conventional Technologies Market (Retrieval Kits), $Million, 2020-2031
Figure 196: Global Minimally Invasive Conventional Technologies Market (Injection Needles), $Million, 2020-2031
Figure 197: Global Minimally Invasive Conventional Technologies Market (Other Instruments and Accessories), $Million, 2020-2031
Figure 198: Global Minimally Invasive Conventional Technologies Market (Others), $Million, 2020-2031
Figure 199: Market Segmentation - Capsule Endoscopy
Figure 200: Global Minimally Invasive Conventional Technologies Market (Capsule Endoscopy), $Million, 2020-2031
Figure 201: Global Minimally Invasive Conventional Technologies Market Share (by Capsule Endoscopy), $Million, 2020 and 2031
Figure 202: Global Minimally Invasive Conventional Technologies Market (Workstations and Data Recorders), $Million, 2020-2031
Figure 203: Global Minimally Invasive Conventional Technologies Market (Capsules), $Million, 2020-2031
Figure 204: Global Minimally Invasive Conventional Technologies Market (Services and Patency System), $Million, 2020-2031
Figure 205: Market Segmentation - Surgical Robotics
Figure 206: Global Minimally Invasive Surgical Robotics Market Share (by Product Type), $Million, 2020 and 2031
Figure 207: Global Minimally Invasive Surgical Robotics Market Optimistic Scenario (Robotics Systems), $Million, 2020-2031
Figure 208: Global Minimally Invasive Surgical Robotics Market Realistic Scenario (Robotics Systems), $Million, 2020-2031
Figure 209: Global Minimally Invasive Surgical Robotics Market Pessimistic Scenario (Robotics Systems), $Million, 2020-2031
Figure 210: Global Minimally Invasive Surgical Robotics Market Optimistic Scenario (Instruments and Accessories), $Million, 2020-2031
Figure 211: Global Minimally Invasive Surgical Robotics Market Realistic Scenario (Instruments and Accessories), $Million, 2020-2031
Figure 212: Global Minimally Invasive Surgical Robotics Market Optimistic Scenario (Instruments and Accessories), $Million, 2020-2031
Figure 213: Global Minimally Invasive Surgical Robotics Market Optimistic Scenario (Services), $Million, 2020-2031
Figure 214: Global Minimally Invasive Surgical Robotics Market Realistic Scenario (Services), $Million, 2020-2031
Figure 215: Global Minimally Invasive Surgical Robotics Market Pessimistic Scenario (Services), $Million, 2020-2031
Figure 216: Global Minimally Invasive Surgical Systems Market (by Region), 2020 and 2031
Figure 217: Global Minimally Invasive Surgical Systems Market Incremental Revenue Opportunity Analysis (by Region), $Million, 2020-2022
Figure 218: Global Minimally Invasive Surgical Systems Market Incremental Revenue Opportunity Analysis (by Region), $Million, 2022-2031
Figure 219: Market Attractiveness Analysis - Global Minimally Invasive Surgical Systems Market (by Region), 2020-2022
Figure 220: Market Attractiveness Analysis - Global Minimally Invasive Surgical Systems Market (by Region), 2022-2031
Figure 221: North America Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 222: Regulatory Process for Medical Devices in the U.S.
Figure 223: North America Minimally Invasive Surgical Systems Market Incremental Revenue Opportunity, $Million, 2020-2031
Figure 224: North America Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Figure 225: North America Minimally Invasive Conventional Technologies Market Optimistic Scenario, $Million, 2020-2031
Figure 226: North America Minimally Invasive Conventional Technologies Market Pessimistic Scenario, $Million, 2020-2031
Figure 227: North America Minimally Invasive Surgical Robotics Market Optimistic Scenario, $Million, 2020-2031
Figure 228: North America Minimally Invasive Surgical Robotics Market Pessimistic Scenario, $Million, 2020-2031
Figure 229: North America Minimally Invasive Surgical Systems Market Share (by Country), $Million, 2020 and 2031
Figure 230: U.S. Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 231: Canada Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 232: Europe Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 233: MDR Transitional Provisions (EU)
Figure 234: Regulatory Process for Medical Devices in the European Union
Figure 235: Europe Minimally Invasive Surgical Systems Market Incremental Revenue Opportunity, $Million, 2020-2031
Figure 236: Europe Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Figure 237: Europe Minimally Invasive Conventional Technologies Market Optimistic Scenario, $Million, 2020-2031
Figure 238: Europe Minimally Invasive Conventional Technologies Market Pessimistic Scenario, $Million, 2020-2031
Figure 239: Europe Minimally Invasive Surgical Robotics Market Optimistic Scenario, $Million, 2020-2031
Figure 240: Europe Minimally Invasive Surgical Robotics Market Pessimistic Scenario, $Million, 2020-2031
Figure 241: Europe Minimally Invasive Surgical Systems Market (by Country), $Million, 2020 and 2031
Figure 242: Germany Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 243: France Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 244: U.K. Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 245: Italy Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 246: Spain Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 247: Netherlands Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 248: Belgium Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 249: Switzerland Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 250: Sweden Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 251: Denmark Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 252: Rest-of-Europe Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 253: Asia-Pacific Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 254: Regulatory Process for Medical Devices in China
Figure 255: Regulatory Process for Medical Devices in Japan
Figure 256: Asia-Pacific Minimally Invasive Surgical Systems Market Incremental Revenue Opportunity, $Million, 2020-2031
Figure 257: Asia-Pacific Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Figure 258: Asia-Pacific Minimally Invasive Conventional Technologies Market Optimistic Scenario, $Million, 2020-2031
Figure 259: Asia-Pacific Minimally Invasive Conventional Technologies Market Pessimistic Scenario, $Million, 2020-2031
Figure 260: Asia-Pacific Minimally Invasive Surgical Robotics Market Optimistic Scenario, $Million, 2020-2031
Figure 261: Asia-Pacific Minimally Invasive Surgical Robotics Market Pessimistic Scenario, $Million, 2020-2031
Figure 262: Asia-Pacific Minimally Invasive Surgical Systems Market (by Country), $Million, 2020 and 2031
Figure 263: Japan Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 264: China Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 265: India Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 266: South Korea Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 267: Australia and New Zealand Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 268: Malaysia Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 269: Singapore Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 270: Rest-of-Asia-Pacific Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 271: Latin America Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 272: Latin America Minimally Invasive Surgical Systems Market Incremental Revenue Opportunity, $Million, 2020-2031
Figure 273: Latin America Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Figure 274: Latin America Minimally Invasive Conventional Technologies Market Optimistic Scenario, $Million, 2020-2031
Figure 275: Latin America Minimally Invasive Conventional Technologies Market Pessimistic Scenario, $Million, 2020-2031
Figure 276: Latin America Minimally Invasive Surgical Robotics Market Optimistic Scenario, $Million, 2020-2031
Figure 277: Latin America Minimally Invasive Surgical Robotics Market Pessimistic Scenario, $Million, 2020-2031
Figure 278: Latin America Minimally Invasive Surgical Systems Market (by Country), $Million, 2020 and 2031
Figure 279: Brazil Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 280: Mexico Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 281: Cost Comparison for Laparoscopic Weight Loss Surgery (U.S. and Mexico)
Figure 282: Argentina Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 283: Rest-of-Latin America Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 284: Middle East and Africa Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 285: Middle East and Africa Minimally Invasive Surgical Systems Market Incremental Revenue Opportunity, $Million, 2020-2031
Figure 286: Middle East and Africa Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Figure 287: Middle East and Africa Minimally Invasive Conventional Technologies Market Optimistic Scenario, $Million, 2020-2031
Figure 288: Middle East and Africa Minimally Invasive Conventional Technologies Market Pessimistic Scenario, $Million, 2020-2031
Figure 289: Middle East and Africa Minimally Invasive Surgical Robotics Market Optimistic Scenario, $Million, 2020-2031
Figure 290: Middle East and Africa Minimally Invasive Surgical Robotics Market Pessimistic Scenario, $Million, 2020-2031
Figure 291: Middle East and Africa Minimally Invasive Surgical Systems Market (by Country), $Million, 2020 and 2031
Figure 292: K.S.A. Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 293: U.A.E. Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 294: Israel Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 295: Rest-of-Middle East and Africa Minimally Invasive Surgical Systems Market, $Million, 2020-2031
Figure 296: Market Share Analysis - Minimally Invasive Conventional Technologies, 2020
Figure 297: Market Share Analysis – Minimally Invasive Surgical Robotics, 2020
Figure 298: Share of Key Developments and Strategies in the Global Minimally Invasive Surgical Systems Market (by Category), January 2017-November 2021
Figure 299: Share of Key Developments and Strategies - Minimally Invasive Conventional Technologies (by Category), January 2017-November 2021
Figure 300: Key Developments and Strategies - Minimally Invasive Conventional Technologies (by Year), January 2017-November 2021
Figure 301: Partnerships, Alliances, and Business Expansions - Minimally Invasive Conventional Technologies, January 2017-November 2021
Figure 302: New Offerings - Minimally Invasive Conventional Technologies, January 2017-November 2021
Figure 303: Mergers and Acquisitions - Minimally Invasive Conventional Technologies, January 2017-November 2021
Figure 304: Regulatory and Legal Activities - Minimally Invasive Conventional Technologies, January 2017-November 2021
Figure 305: Funding Activities - Minimally Invasive Conventional Technologies, January 2017-November 2021
Figure 306: Share of Key Developments and Strategies – Minimally Invasive Surgical Robotics (by Category), January 2017-November 2021
Figure 307: Key Developments and Strategies - Minimally Invasive Surgical Robotics (by Year), January 2017-November 2021
Figure 308: Partnerships, Alliances, and Business Expansions - Minimally Invasive Surgical Robotics, January 2017-November 2021
Figure 309: New Offerings and Upgradations - Minimally Invasive Surgical Robotics, January 2017-November 2021
Figure 310: Mergers and Acquisitions - Minimally Invasive Surgical Robotics, January 2017-November 2021
Figure 311: Regulatory and Legal Activities - Minimally Invasive Surgical Robotics, January 2017-November 2021
Figure 312: Funding Activities - Minimally Invasive Surgical Robotics, January 2017-November 2021
Figure 313: Arthrex Inc.: Product Portfolio
Figure 314: Asensus Surgical, Inc.: Product Portfolio
Figure 315: Asensus Surgical, Inc.: Overall Financials, $Million, 2018-2020
Figure 316: Asensus Surgical, Inc.: Net Revenue (by Product), $Million, 2018-2020
Figure 317: Asensus Surgical, Inc.: Net Revenue (by Region), $Million, 2018-2020
Figure 318: Boston Scientific Corporation: Product Portfolio
Figure 319: Boston Scientific Corporation: Overall Financials, $Million, 2018-2020
Figure 320: Boston Scientific Corporation: Net Revenue (by Segment), $Million, 2018-2020
Figure 321: Boston Scientific Corporation: Net Revenue (by Region), $Million, 2018-2020
Figure 322: Conmed Corporation: Product Portfolio
Figure 323: Conmed Corporation: Overall Financials, $Million, 2018-2020
Figure 324: Conmed Corporation: Net Revenue (by Segment), $Million, 2018-2020
Figure 325: Conmed Corporation: Net Revenue (by Region), $Million, 2018-2020
Figure 326: Fujifilm Holdings Corporation: Product Portfolio
Figure 327: Fujifilm Holdings Corporation: Overall Financials $Million, 2018-2020
Figure 328: Fujifilm Holdings Corporation: Net Revenue (by Segment), $Million, 2018-2020
Figure 329: Fujifilm Holdings Corporation: Net Revenue (by Region), $Million, 2018-2020
Figure 330: Intuitive Surgical, Inc.: Product Portfolio
Figure 331: Intuitive Surgical, Inc.: Overall Financials, $Million, 2018-2020
Figure 332: Intuitive Surgical, Inc.: Net Revenue (by Product), $Million, 2018-2020
Figure 333: Intuitive Surgical, Inc.: Net Revenue (by Region), $Million, 2018-2020
Figure 334: Karl Storz SE &Co. KG: Product Portfolio
Figure 335: Medtronic plc: Product Portfolio
Figure 336: Medtronic plc: Overall Financials, $Million, 2019-2021
Figure 337: Medtronic plc: Net Revenue (by Segment), $Million, 2019-2021
Figure 338: Medtronic plc: Net Revenue (by Region), $Million, 2019-2021
Figure 339: Olympus Corporation: Product Portfolio
Figure 340: Olympus Corporation: Overall Financials, $Million, 2019-2021
Figure 341: Olympus Corporation: Net Revenue (by Segment), $Million, 2019-2021
Figure 342: Olympus Corporation: Net Revenue (by Region), $Million, 2019-2021
Figure 343: Hoya Corporation (Pentax Medical): Product Portfolio
Figure 344: Hoya Corporation: Overall Financials, $Million, 2019-2021
Figure 345: Hoya Corporation: Net Revenue (by Segment), $Million, 2019-2021
Figure 346: Hoya Corporation: Net Revenue (by Region), $Million, 2019-2021
Figure 347: Siemens Healthineers AG: Product Portfolio
Figure 348: Siemens Healthineers AG: Overall Financials, $Million, 2018-2020
Figure 349: Siemens Healthineers AG: Net Revenue (by Product), $Million, 2018-2020
Figure 350: Siemens Healthineers AG: Net Revenue (by Region), $Million, 2018-2020
Figure 351: Smith & Nephew plc: Product Portfolio
Figure 352: Smith & Nephew plc: Overall Financials, $Million, 2018-2020
Figure 353: Smith & Nephew plc: Net Revenue (by Product), $Million, 2018-2020
Figure 354: Smith & Nephew plc: Net Revenue (by Region), $Million, 2018-2020
Figure 355: Sony Corporation: Product Portfolio
Figure 356: Sony Corporation: Overall Financials, $Million, 2019-2021
Figure 357: Sony Corporation: Net Revenue (by Segment), $Million, 2019-2021
Figure 358: Sony Corporation: Net Revenue (by Region), $Million, 2019-2021
Figure 359: Stryker Corporation: Product Portfolio
Figure 360: Stryker Corporation: Overall Financials, $Million, 2018-2020
Figure 361: Stryker Corporation: Net Revenue (by Segment), $Million, 2018-2020
Figure 362: Stryker Corporation: Net Revenue (by Region), $Million, 2018-2020
Figure 363: Zimmer Biomet Holdings, Inc.: Product Portfolio
Figure 364: Zimmer Biomet Holdings, Inc.: Overall Financials, $Million, 2018-2020
Figure 365: Zimmer Biomet Holdings, Inc.: Net Revenue (by Product), $Million, 2018-2020
Figure 366: Zimmer Biomet Holdings, Inc.: Net Revenue (by Region), $Million, 2018-2020
Figure 367: Research Methodology
Figure 368: Primary Research
Figure 369: Secondary Research
Figure 370: Data Triangulation
Figure 371: Overview of Market Estimation Process
List of Tables
Table 1: Impact Analysis for Key Market Drivers and Restraints, 2021-2031
Table 2: List of Surgical Robotic Systems with Haptic Feedback Capabilities
Table 3: Patent Landscape – Minimally Invasive Conventional Technologies, January 2018-December 2020
Table 4: Patent Landscape – Minimally Invasive Surgical Robotics, January 2018-December 2020
Table 5: Impact Analysis
Table 6: Global Minimally Invasive Surgical Systems Market, Hospital Procurement Future Impact Analysis
Table 7: Global Minimally Invasive Surgical Systems Market, Key Barriers to Entry and Possible Solutions
Table 8: Global Minimally Invasive Surgical Systems Market Opportunities (Future Impact)
Table 9: Global Minimally Invasive Surgical Systems Market Supply Chain Distribution Challenges
Table 10: Global Minimally Invasive Surgical Systems Market Access Limitations Impact Analysis
Table 11: Global Minimally Invasive Surgical Systems Market, Post-COVID-19 Optimistic Scenario
Table 12: Global Minimally Invasive Surgical Systems Market, Post-COVID-19 Realistic Scenario
Table 13: Global Minimally Invasive Surgical Systems Market, Post-COVID-19 Pessimistic Scenario
Table 14: North America Conventional Technologies Market, (by Product Type), $Million, 2020-2031
Table 15: North America Surgical Robotics Market, $Million, 2020-2031
Table 16: U.S. Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 17: Canada Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 18: Europe Conventional Technologies Market, $Million, 2020-2031
Table 19: Europe Surgical Robotics Market, $Million, 2020-2031
Table 20: Germany Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 21: France Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 22: U.K. Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 23: Italy Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 24: Spain Minimally Invasive Surgical Systems Market (Product Type), $Million, 2020-2031
Table 25: Netherlands Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 26: Belgium Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 27: Switzerland Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 28: Sweden Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 29: Denmark Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 30: Rest-of-Europe Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 31: Asia-Pacific Conventional Technologies Market, $Million, 2020-2031
Table 32: Asia-Pacific Surgical Robotics Market, $Million, 2020-2031
Table 33: Japan Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 34: China Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 35: India Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 36: Major Minimally Invasive Surgeries Performed with the Help of Laparoscopy in India
Table 37: South Korea Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 38: Australia and New Zealand Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 39: Leading Hospitals in Australia Using the da Vinci Robotic Surgical System
Table 40: Malaysia Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 41: Singapore Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 42: Rest-of-Asia-Pacific Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 43: Latin America Conventional Technologies Market, $Million, 2020-2031
Table 44: Latin America Surgical Robotics Market, $Million, 2020-2031
Table 45: Brazil Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 46: Mexico Minimally Invasive Surgical Systems Market (Product Type), $Million, 2020-2031
Table 47: Argentina Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 48: Rest-of-Latin America Surgical Robotics Market (by Product Type), $Million, 2020-2031
Table 49: Middle East and Africa Conventional Technologies Market, $Million, 2020-2031
Table 50: Middle East and Africa Surgical Robotics Market, $Million, 2020-2031
Table 51: K.S.A. Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 52: U.A.E. Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 53: Israel Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 54: Rest-of-Middle East and Africa Minimally Invasive Surgical Systems Market (by Product Type), $Million, 2020-2031
Table 55: Competitor Snapshot (Key Players)
Table 56: Asensus Surgical, Inc.: Net Revenue (U.S. and International), $Million, 2018-2020
Table 57: Intuitive Surgical, Inc.: Unit Sold, Installed Base, and Procedure Volume, 2014-2020
Table 58: Intuitive Surgical, Inc.: Net Revenue (U.S. and International), $Million, 2018-2020
Companies Mentioned
- Arthrex Inc.
- Asensus Surgical, Inc.
- Boston Scientific Corporation
- Conmed Corporation
- Fujifilm Holdings Corporation
- Intuitive Surgical, Inc.
- Karl Storz SE & Co. KG
- Medtronic plc
- Olympus Corporation
- Hoya Corporation (Pentax Medical)
- Siemens Healthineers AG
- Smith & Nephew plc
- Sony Corporation
- Stryker Corporation
- Zimmer Biomet Holdings, Inc.
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 450 |
| Published | February 2022 |
| Forecast Period | 2021 - 2031 |
| Estimated Market Value ( USD | $ 30176.1 Million |
| Forecasted Market Value ( USD | $ 55716.7 Million |
| Compound Annual Growth Rate | 6.3% |
| Regions Covered | Global |
| No. of Companies Mentioned | 15 |