Medical device connectivity is one of the industries that had a positive impact due to the COVID-19 pandemic owing to the extensive patient pool getting admitted to the hospital due to the infection. For instance, according to an article published by De Gruyter, in August 2021, Advances in the internet of medical things (IoMT) technology allow to connect of all medical tools, medical databases, and devices via the internet in one collaborative network, which conveys real-time data integration and analysis. IoMT framework-driven COVID-19 self-assessment tool captured signs and symptoms through multiple probing questions, storing the data in the COVID-19 patient database, then analyzing the data to determine whether a person needs to be tested for COVID-19 or other actions may require to be taken. Medical device connectivity system has demonstrated the ability to enhance health outcomes and reduce chaos during the pandemic, which made hospitals realize the advantage of medical device connectivity system over manual systems. Hence, the demand for medical device connectivity system systems increased, eventually driving the market growth.
The major factors for the growth of the market include the rising implementation of electronic health records and health information exchange systems, an increase in the demand for telehealth and telemedicine technologies, and technological advancements in wireless technologies and Bluetooth-enabled medical devices. For instance, according to an article published by Cox Communications, Inc., in May 2022, Bluetooth Low Energy (BLE) was developed and used Bluetooth technology to track. BLE makes use of software and gateways via cell phones and other devices. It enables low-power wireless technology, extending battery life while allowing devices to communicate with one another. The vast majority of smartphones and tablets are BLE compatible, making them perfect for closer contact. Furthermore, in September 2021, the Faculty of Sexual and Reproductive Healthcare (FSRH) and the Royal College of Obstetricians and Gynecologists (RCOG) joined forces to launch a new campaign to disseminate information about telemedicine for early medical abortion (EMA). Thus, the increasing adoption rate of telemedicine services is directly affecting the growth of the medical device connectivity market.
The companies are actively involved in new product launch developments and collaborations to expand their footprint. For instance, in October 2021, Royal Philips launched device drivers that enable integration and interoperability, Philips Capsule Medical Device Information Platform (MDIP). Philips Capsule MDIP integrated into the Philips HealthSuite Platform, installed in more than 3,000 healthcare facilities globally. Philips Capsule MDIP captures streaming clinical data and transforms it into actionable insights for patient care management, aiming to improve collaboration between care teams, streamline clinical workflows, and increase productivity. Such development is expected to drive the growth of the market over the forecast period.
Therefore, the factors mentioned above are attributed collectively to the studied market growth over the forecast period. However high cost of deployment and complete installation limiting the adoption of advanced solutions are expected to hinder market growth over the forecast period.
Medical Device Connectivity Market Trends
Wireless Technologies Segment by Technology is Expected to Witness a Significant Growth over the Forecast Period
Wireless technology includes controlling and programming a medical device, monitoring patients remotely, or transferring patient data from the medical device to another platform, such as a cell phone. The major factors driving the growth of the market include rising technological advancements and an increase in the adoption of wireless medical device connectivity in healthcare facilities. For instance, the Radio Frequency Wireless Technology and Bluetooth Low Energy (BLE) in Medical Devices provide recommendations to develop and support safe and effective wireless medical devices. Medical device manufacturers are encouraged to read this guide and review related recognized standards and guidance documents to help in the development, testing, regulatory submission, and use of wirelessly enabled medical devices. For instance, according to the RFiD Discovery 2022, a unique device identification system for medical devices is mandated by the U.S. FDA's Unique Device Identification (UDI) Rule. This means that every medical device used in the US must be marked with a Unique Device Identifier (UDI) that can be read by both humans and machines. The demand for advanced technologies is fueling the wireless technology segment of the market.Furthermore, the companies are actively involved in new product launch developments and collaborations to expand their footprint in the wireless technology segment. For instance, in September 2021, CareVention HealthCare launched a remote patient monitoring service that uses biosensors and artificial intelligence to monitor patients' vital signs and alert healthcare providers if necessary.
Therefore, the above-mentioned factors are expected to drive segmental growth in the market during the forecast period.
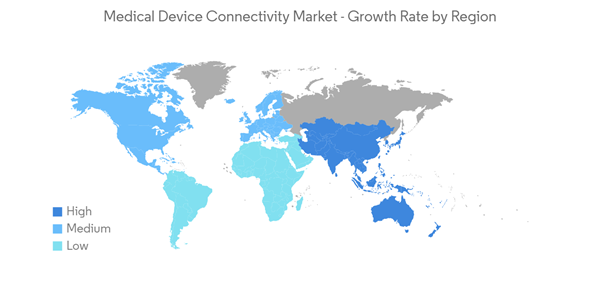
North America is Expected to hold a Significant Market Share over the Forecast Period
Medical device connectivity is a rapidly growing component of healthcare in North America due to the growing burden of chronic diseases and the high adoption of advanced healthcare technologies. The adoption of telemedicine has improved care management, patients' quality of life, and reduced healthcare spending. For instance, according to the 2021 report from the Canadian Institute of Health Information, Canadian health expenditure increased to USD 308.1 billion in 2021 compared to USD 301.5 billion in 2020. Similarly, as per the 2022 report of the Centers for Medicare and Medicaid Services (CMS), the health expenditure of the United States was about USD 4.3 trillion, an increase of about USD 173 billion from the previous year. Thus, the increasing healthcare spending is anticipated to create opportunities for telemedicine, thereby propelling market growth.Additionally, governments and companies are engaged in innovation and initiative in the medical device connectivity market, driving growth in the forecast period. For instance, in October 2021, the FDA's Digital Health Center of Excellence (DHCoE) held a public workshop on the transparency of artificial intelligence/machine learning-enabled medical devices. The workshop followed the recently published list of nearly 350 AI/ML-enabled medical devices. The workshop was aimed at moving forward the objectives of FDA's DHCoE to 'empower stakeholders to advance healthcare by fostering responsible and high-quality digital health innovation.'
Thus, given the factors above, the telemedicine market is expected to grow significantly in North America over the forecast period.
Medical Device Connectivity Industry Overview
The medical device connectivity market is consolidated in nature due to the presence of several companies operating globally as well as regionally. The competitive landscape includes an analysis of a few international as well as local companies which hold the market shares and are well known, including Capsule Technologies, Inc., Cerner Corporation, Cisco Systems, GE Healthcare, Ihealth Lab, Infosys, Koninklijke Philips N.V., Lantronix, Inc., and NantHealth, Inc., among others.Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Capsule Technologies, Inc.
- Cerner Corporation
- Cisco Systems
- GE Healthcare
- Ihealth Lab
- Infosys Limited
- Koninklijke Philips N.V.
- Lantronix, Inc.
- NantHealth, Inc.