The COVID-19 pandemic had a positive impact on the market. Instead of sending patient samples to the laboratory, the usage of lateral flow test kits can be convenient as they are smaller, portable, and can be controlled with minimal training. This makes them attractive for deployment outside of healthcare facilities, in low- and middle-income countries, or for countries rolling out mass population screening programs. Even though they are less accurate than PCR tests, their low cost, speed, and ease of use make lateral flow tests particularly attractive to countries that do not have extensive laboratory facilities or trained health workers to easily conduct PCR tests for COVID-19 screening. Some countries, such as Slovakia and the United Kingdom, are extensively using lateral flow tests as a means of mass screening programs. For example, the British government has been monitoring the use of the Innova SARS-CoV-2 Antigen Rapid Qualitative Test in Liverpool, which currently has high COVID-19 rates, and it plans to roll it out nationwide.
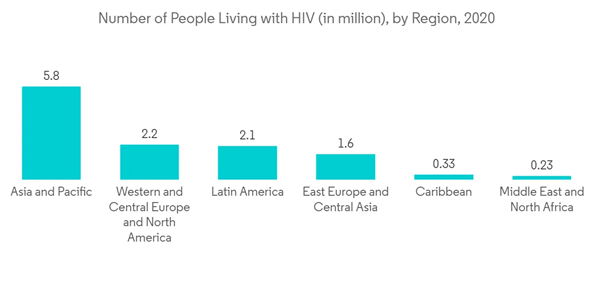
The major factor driving the growth of the lateral flow assay market is the growing global rate of infectious diseases, HIV, and cancer around the world, which require new diagnostic methods of effective treatment to end the growing mortality rate. For instance, according to the November 2021 report of the World Health Organization (WHO) on HIV/AIDS, about 37.7 million people around the world were living with HIV in 2020, of which 25.4 million were residing in the WHO African region. As per the same report, in 2020, about 1.5 million people were diagnosed with HIV around the world. Thus, the high prevalence of HIV is expected to drive the demand for effective and precise diagnostics tests for the efficient and early diagnostics of HIV, which is anticipated to drive the market growth as they provide accurate diagnostics.
Further, technological advancements in the field of lateral flow assays (LFA) have also boosted the market growth. In recent years, major advances in LFA development have included novel signal enhancement techniques, the use of new labels, improved quantification systems, and simultaneous detection. Some new techniques have been used to enhance the signal from gold nanoparticles (colloidal nanoparticles). With the advancement in technology, GNPs have adopted silver enhancement technology or combinations of GNPs with an enzyme (such as horseradish peroxidase), which results in catalytic amplification of the signal. For instance, in June 2021, Novacyt launched two COVID-19 antigen lateral flow tests called PathFlow COVID-19 Rapid Antigen Pro and PathFlow COVID-19 Rapid Antigen.
Additionally, increased participation of the government to control the outbreaks of infectious diseases and the increasing awareness programs about the spread of infections, diagnosis, and prevention, along with the launch of diagnosis systems at home care settings, are expected to contribute to the growth of the global market during the forecast period. However, factors such as delays in product approvals due to stringent regulatory policies and false results from the test are expected to impede the growth of the studied market over the forecast period.
Lateral Flow Assays Market Trends
The Clinical Testing Segment is Expected to Hold the Largest Market Share in the Lateral Flow Assay Market
The clinical testing segment is anticipated to hold the largest market share in the lateral flow assay market due to its importance in the identification and detection of diseases. With the rise in the incidence of infectious diseases and other medical conditions, the demand for these assays is expected to increase, driving the segment's growth. For instance, according to the World Health Organization, Global Tuberculosis Report 2020, approximately 1.4 million people died from tuberculosis-related illnesses in 2019, and nearly 10 million individuals developed tuberculosis in 2019. Hence, the growing burden of tuberculosis is expected to drive the need for rapid and affordable diagnostics, especially in developing countries, which aids in the growth of the studied market.The increased demand for the segment is due to an increase in the new approvals of self-test devices, such as Atomo HIV Self-Test, TRUSTline HIV-Ab/Ag 4th Gen Rapid Test, Dr Trust Hiv Sure, and others, developed in recent years. These self-test devices and kits help the patients in assurance, earlier diagnosis, enhanced convenience, and ease of use, which are likely to fuel the market.
Further, the COVID-19 pandemic has drastically increased launches and research in the field of clinical testing, bolstering the segment growth. For instance, in September 2020, Roche launched a SARS-CoV-2 Rapid Antigen Test for markets accepting the CE Mark. The SARS-CoV-2 Rapid Antigen Test is for use in point-of-care settings for both symptomatic and asymptomatic people.
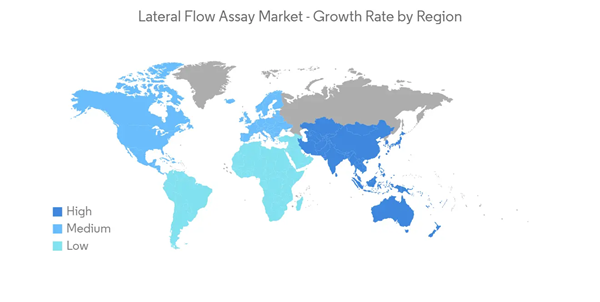
North America Dominates the Market, and It is Expected to do the Same During the Forecast Period
North America is expected to dominate the overall lateral flow assay market throughout the forecast period, owing to a rise in the incidence of various infectious and chronic diseases, such as Lyme disease and tuberculosis, and an increased mortality rate associated with HIV/AIDS in the region. In North America, the United States is anticipated to hold the largest market share due to the presence of a high patient pool and affordability with increasing disposable income.As per the United States Department of Health & Human Services report, published in January 2020, about 37,832 people in the United States were diagnosed with HIV in 2018, and 38,000 new HIV infections still occur in the United States. It has also been reported that there is a rapid spread of various infectious diseases such as meningitis and urinary tract infections, necessitating immediate diagnosis, thereby increasing the demand for fast diagnostic test devices and fueling the market growth. Furthermore, the increased adoption rate of advanced infrastructure, the volume of diagnostic centers, and the rise in awareness among people are estimated to boost the market in the region, contributing to its outstanding share of the global market revenue during the forecast period.
Furthermore, the pandemic hit the region very hard and forced the government to implement mass screening programs, boosting the market growth in the region. In 2020, Abbott Laboratories ramped up its production of coronavirus test kits, including a new tool that could enable mass COVID-19 screening. The company has announced that it is developing the fourth diagnostic test for the coronavirus: A 'lateral flow' blood test that could provide mass testing to the general population, which is further expected to positively impact the market in the region.
Lateral Flow Assays Industry Overview
The lateral flow assay market is moderately competitive and consists of several major players. Some of the companies are expanding their market positions by adopting various strategies, such as mergers and acquisitions, while others are developing new test methods for diagnosis and introducing new products to retain their market shares. For instance, in February 2019, Abbott announced the availability of its latest rapid influenza diagnostic test (RIDT), BinaxNOW Influenza A & B Card 2, which was granted under the Clinical Laboratory Improvements Amendments (CLIA) by the United States Food and Drug Administration (FDA) for rapid detection of influenza virus, positively impacting the market. Some of the companies currently dominating the market are Abbott Laboratories, Hologic Inc., Quidel Corporation, PerkinElmer Inc., and bioMerieux SA.Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- PerkinElmer Inc.
- Merck KGaA
- QIAGEN NV
- Abbott Laboratories (Alere Inc.)
- Quidel Corporation
- Hologic Inc.
- Thermo Fisher Scientific Inc.
- bioMerieux SA
- Becton, Dickinson and Company
- Bio-Rad Laboratories Inc.
- Abcam PLC
- Danaher Corporation
- F. Hoffmann-La Roche AG
- Siemens AG
- Chembio Diagnostic Systems Inc.