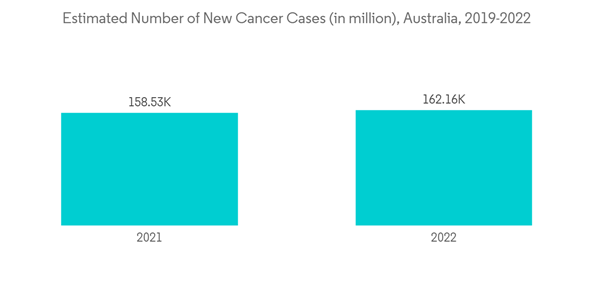The COVID-19 pandemic impacted the studied market significantly. Due to restricted hospital visits and delayed surgeries, the demand for MIS devices declined and hampered the market growth during the initial phase of the pandemic. For example, as per an article published in IOVS journal in June 2022, during the initial phases of the pandemic, the rate of ophthalmic surgical services decreased with restrictions, largely for non-critical conditions. The study also showed that cataract surgery with minimally invasive glaucoma surgical device insertion decreased by 71%. Thus, initially, the pandemic had an adverse impact on the studied market. Currently, the surgeries are operating normally, like in the pre-pandemic days in Australia. As MIS decreased hospital stays, it decreased the chances of COVID-19 co-infection during the limited hospital stays. Hence, the demand for MIS increased, further contributing to the MIS devices demand generation in the country. In addition, the market growth is stabilizing in the current scenario after COVID-19 as the worldwide restrictions have eased down and surgeries have been resumed.
Factors such as an increasing prevalence of lifestyle-related and chronic disorders and a rising preference for minimally invasive procedures are the major contributors to the market's growth.
Lifestyle-related diseases have become common in Australia, and arthritis, asthma, back pain, cancer, cardiovascular disease, chronic obstructive pulmonary disease, diabetes, and mental health conditions are the major conditions that are commonly seen among the target population. According to a report published by AIHW in October 2022, approximately 162,163 new cancer cases were diagnosed in 2022, which increased from 158,530 new cancer cases that were diagnosed in 2021. As MIS provides benefits to patients such as less postoperative pain, fewer operative and postoperative complications, shortened hospital stay, faster recovery times, minimal stress on the immune system, and smaller incisions, the demand for MIS surgeries increases for the treatment of cancer and other lifestyle-related chronic diseases, driving the demand for MIS devices.
Furthermore, global and regional market players are developing and innovating advanced products in the country. For instance, in April 2022, Nova Eye Medical Limited, a medical technology company headquartered in Australia and committed to advanced ophthalmic treatment technologies and devices, launched its next-generation canaloplasty device, iTrack Advance, in selected markets. Dr. Nathan Kerr, an authority on minimally invasive glaucoma surgery and head of glaucoma surgical trials at the Centre for Eye Research Australia, performed the world's first surgical case with the iTrack Advance following the launch of the product. Thus, such activities are anticipated to create opportunities for MIS device innovation and development in Australia, thereby driving market growth over the analysis period.
Therefore, owing to the abovementioned factors, the market studied is expected to witness growth. However, the lack of skilled professionals and the high cost of MIS surgeries and devices are expected to hinder market growth over the studied period.
Australia Minimally Invasive Surgery Devices Market Trends
Endoscopic Devices Segment Expected to Grow Significantly
Endoscopic devices are minimally invasive and can be inserted into natural openings of the human body to observe an internal organ or a tissue in detail. The surgeries performed using endoscopic devices are generally imaging procedures and minor surgeries. Endoscopy can be indicated for various types of conditions, such as GIT disorders, pancreatitis, gastric/stomach cancer, respiratory tract disorders, urinary tract disorders, etc. Due to the clinical significance of endoscopic devices, these devices are gaining market demand and are expected to continue their growth at a faster rate over the next few years.Endoscopic devices pose various advantages for the detection of various diseases. For instance, as per an article published in December 2021 in Cancers Journal, the detection and diagnosis of stomach lesions have improved due to the ongoing advancements in endoscopic technologies over time. Endoscopic technologies have been continuously advancing throughout the years to facilitate improvement in the detection and diagnosis of gastric lesions. Thus, owing to the burden of cancers in Australia, the demand for endoscopic devices is likely to rise, propelling the endoscopic devices market toward growth.
The market for an endoscope is expected to be driven by favorable reimbursement policies, technological advancements, and an increasing number of new players in the endoscopy devices market. As per the September 2022 update by KARL STORZ Endoscopy Australia Pty Ltd, the company manufactures best-in-class endoscopes in Sydney, Australia. In addition to the Sydney location, regional offices are also available in New South Wales, Victoria, Queensland, South Australia, and Western Australia to ensure a reliable infrastructure. Thus, the presence of such global and regional manufacturers that are focused on developing innovative endoscopes is anticipated to garner the segment's growth during the forecast period.
Orthopedic Segment Expected to Hold Significant Market Share
Minimally invasive surgery (MIS), also known as keyhole surgery, is carried out through several small incisions rather than a single large opening to achieve surgical results. With less stress to the nearby soft tissues, the MIS is performed. MIS offers to employ small, focused incisions to solely treat the problematic area during orthopedic surgery. Compared with traditional surgery, smaller incisions during MIS possess less danger, less pain, and quicker recovery for patients. Thus, the demand for MIS devices in orthopedic applications is increasing, driving the segment’s growth.Musculoskeletal conditions are becoming a burden for the Australian target population. Due to this, the demand for MIS surgeries is increasing in the country. For instance, as per data updated by AIHW in July 2022, chronic musculoskeletal conditions affect nearly 3 in 10 Australians. In addition, as per the Australian Bureau of Statistics 2020-21, nearly 6.9 (27% of all Australians) million people with chronic musculoskeletal conditions were self-reported, among which 3.9 million (16%) had back problems, 3.1 million (12%) had arthritis, and 889,000 (3.6%) had osteoporosis.
In addition, as per the data updated by The University of Adelaide in June 2022, arthritis affected nearly 17% of the population, and back disorders affected approximately 15% of the target population. Furthermore, 1 in 2 women and 1 in 4 men will be diagnosed with osteoporosis in their lifetime, with a high risk of fracture in Australia. This shows that the target population in Australia has a burden of musculoskeletal conditions that are further expected to create demand for MIS surgeries, accelerating the demand for the availability of MIS devices for orthopedic treatment. Therefore, the aforementioned factors are expected to drive the segment’s growth over the forecast period.
Australia Minimally Invasive Surgery Devices Market Competitor Analysis
The Australian minimally invasive surgery devices market is moderately competitive, with several international as well as local companies present. The market players have been providing minimally invasive surgery (MIS) solutions and focusing on product developments continuously. The major players in the Australian minimally invasive surgery devices market are Boston Scientific Corporation, Koninklijke Philips NV, Medtronic PLC, Olympus Corporation, Stryker Corporation, and Zimmer Biomet, among others.Additional benefits of purchasing the report:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Boston Scientific Corporation
- GE Healthcare
- Intuitive Surgical Inc.
- Koninklijke Philips NV
- Medtronic PLC
- Olympus Corporation
- Siemens Healthineers
- Smith & Nephew
- Stryker Corporation
- Zimmer Biomet