The COVID-19 pandemic had a significant impact on the actinic keratosis treatment market. There was a decrease in the number of aesthetic procedures performed worldwide during the COVID-19 pandemic to reduce the risk of viral transmission. For instance, according to an article published by Elsevier Public Health Emergency Collection, in February 2021, during the crisis, dermatologists were involved in managing cutaneous manifestations related to COVID-19 and occupational disease caused by personal protective equipment. The reduction in the number of procedures was attributed to the fear and risk of infection from COVID-19. However, with the adoption of telemedicine and teleconsultations during the pandemic, the demand for dermatological treatment increased, which led to a rise in the demand for actinic keratosis treatment. However, as COVID-19 cases have declined and the pandemic accelerated the use of teledermatology, the studied market is expected to project stable growth over the studied period.
The increasing prevalence of actinic keratosis, growing demand for minimally invasive procedures, and increasing research and development for developing novel therapies are the key driving factors in the market. For instance, according to an article published by the Indian Journal of Dermatology, in April 2022, actinic keratosis is one of the most frequently seen signs in the outpatient setting and was diagnosed at more than 10% of dermatology visits. In the UK, 15%-23% of individuals have actinic keratosis lesions, and this reaches 37.5% among people older than 50 years of age. This is likely to increase the adoption of the available treatment and early diagnosis of the disease. Furthermore, according to an article published by MDPI, in June 2022, photodynamic therapy (PDT) using 5-aminolevulinic acid (5-ALA) is an emerging treatment option in the care of actinic keratosis. This increase in the prevalence of actinic keratosis leading to a surge in minimally invasive procedures such as photodynamic therapy is expected to aid the overall market growth in the studied period.
The recent product launches by the key market players are also aiding the market growth. For instance, in November 2022, Biofrontera Inc. launched a randomized, double-blind, vehicle-controlled, multicenter Phase 3 clinical study to evaluate the safety and efficacy of Ameluz and BF-RhodoLED XL in the field-directed treatment of actinic keratosis (AK) on the extremities, neck, and trunk. Furthermore, in September 2021, Almirall, S.A. launched Klisyri (tirbanibulin) in Germany and the U.K. The product was launched upon approval from the U.K.’s Medicines & Healthcare Products Regulatory Agency (MHRA) and the European Commission for treating actinic keratosis of the scalp and face in adults.
Risk factors associated with available treatment options may restrain the market growth over the forecast period.
Actinic Keratosis Treatment Market Trends
Photodynamic Therapy Segment is Expected to Hold a Significant Market Share in the Actinic Keratosis Treatment Market
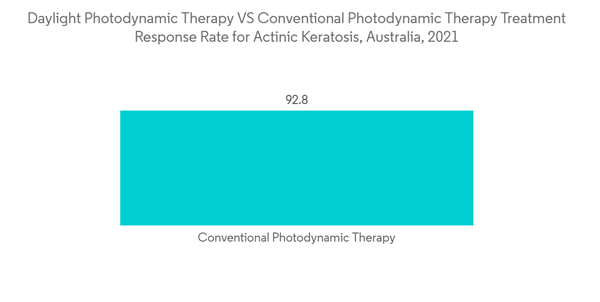
The photodynamic therapy segment holds a significant share of the actinic keratosis treatment market. It is anticipated to show a similar trend over the forecast period due to the noninvasive nature of the treatment, high target specificity, and a lower risk of scarring. The rising incidence of baldness, skin wrinkling, and sunburn patches have led to increased actinic keratosis cases. According to an article published by the National Library of Medicine in May 2022, countries closer to the equator demonstrate higher rates of actinic keratosis. The prevalence rates of actinic keratosis in Australia are close to 60% due to its proximity to the equator.The recent initiatives, launches, and agreements by the market players are also aiding the market growth. For instance, in December 2022, Biofrontera AG's Scandinavian licensing partner Galenica AB, Malmö, Sweden, started the commercialization of Ameluz and BF-RhodoLED in Finland after Galenica had already successfully launched the Ameluz-photodynamic therapy in Sweden, Norway, and Denmark in mid-2021 under a licensing agreement at the end of 2020. Also, in December 2021, Biofrontera Inc. enrolled in a Phase 1 study to evaluate the safety and tolerability of photodynamic therapy (PDT) for the treatment of actinic keratosis (AK) using three tubes of Ameluz together with the company’s BF-RhodoLED XL lamp. Such product approvals are expected to drive the market.
North America is Expected to Hold a Significant Share in the Market and Expected to do Same Over the Forecast Period
North America is expected to hold a significant share of the global actinic keratosis treatment market due to the growing prevalence of actinic keratosis coupled with a higher proportion of older individuals in this region. For instance, as per the 2021 America's Health Rankings data, over 54 million people over the age of 65 are living in the United States, comprising 16.5% of the total population. It was projected that the nation's total number of adults aged 65 and older will rise to an estimated 85.7 million by the year 2050. Furthermore, according to a Statistics Canada update, in April 2022, 1 in 5 Canadians (19%) were aged 65 and older (7 million), and approximately 25% were baby boomers (9.2 million) in 2021. Additionally, according to a World Bank update in January 2023, the population ages 65 and above in Mexico was reported at 8.13% in 2021.The recent initiatives, launches, and agreements by the market players are also aiding the market growth. For instance, in February 2021, Almirall launched its novel microtubule inhibitor, tirbanibulin (Klisyri, Almirall), for the topical treatment of actinic keratosis (AK) on the face and scalp in the United States. Such development is expected to drive the growth of the market.
Actinic Keratosis Treatment Market Competitor Analysis
The actinic keratosis treatment market is moderately concentrated in nature due to the presence of companies operating globally as well as regionally, including Bausch Health Companies Inc, LEO Pharma A/S, Almirall, S.A., GlaxoSmithKline plc, Cipher Pharmaceuticals Inc., Biofrontera AG, Sun Pharmaceutical Industries Limited, 3M Company, Galderma SA, Alma Lasers, and Novartis AG and Hill Dermaceuticals, Inc.Additional benefits of purchasing the report:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Bausch Health Companies Inc
- Almirall, S.A.
- Biofrontera AG
- Sun Pharmaceutical Industries Limited
- Galderma SA
- Alma Lasers
- Hill Dermaceuticals, Inc
- GlaxoSmithKline plc
- Cipher Pharmaceuticals Inc.