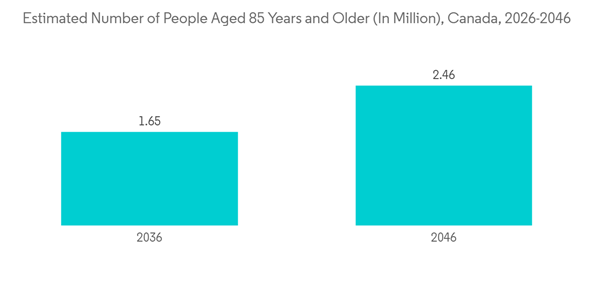The market for cardiovascular medical devices was negatively impacted, particularly in the initial period of the pandemic, owing to the cancellation of elective surgeries during COVID-19-related lockdowns in Canada. Even though some cardiovascular treatments were treated as urgent, others were put off or delayed during the pandemic. However, postmarket vaccine-safety surveillance systems in multiple countries identified a likely association of myocarditis or pericarditis with mRNA COVID-19 vaccination. Hence, to identify the outcome, the country's government is funding the diagnosis of myocarditis or pericarditis. For instance, in October 2022, the Canadian Cardiovascular Society (CCS) received USD 1.6 million from the Canadian government to conduct the study of the clinical and functional outcomes of adults and children who experienced myocarditis and/or pericarditis after receiving an mRNA COVID-19 vaccination. Thus, new funding for identifying cardiovascular diseases after the COVID-19 vaccination is increasing the demand for cardiovascular diseases (CVD).
The major factors driving the growth of the market in Canada are the increasing burden of cardiovascular diseases, the high prevalence of CVD risk factors like diabetes, hypertension, etc., and the increased adoption of minimally invasive procedures. Cardiovascular diseases are the most prevalent across the country. For instance, as per the data published by the Heart and Stroke Foundation Canada in February 2022, 750,000 people have a heart failure and 100,000 people are diagnosed with this incurable condition each year in Canada. As per the same source, one in three Canadians is affected by heart failure, either personally or through a family member or close acquaintance. Hence, the high burden of cardiovascular diseases in the country increases the demand for cardiovascular diagnostics, which is expected to boost the market over the forecast period.
Technologies used in the development of cardiology devices have improved significantly over the past decade, and indications for these devices have expanded. This trend has led to an increasing number of patients being managed with cardiology devices, resulting in exponential therapeutical and monitoring outcomes. For instance, in July 2022, Abbott launched EnSite X EP System with EnSite Omnipolar Technology (OT), a new cardiac mapping platform designed to help physicians better treat abnormal heart rhythms in Canada. The new platform allows for the creation of a 3D model of the patient's cardiac anatomy in real-time and helps in treating areas of the heart.
However, the stringent regulatory policies and high cost of instruments and procedures are the factors restraining the growth of the cardiovascular devices market.
Key Market Trends
Electrocardiogram (ECG) Under Diagnostic and Monitoring Devices Segment is Expected to Record Significant Growth in the Market During the Forecast Period
An electrocardiogram (ECG) records the electrical signals from the heart. It is a common and painless test used to detect heart problems and monitor the heart's health. Portable and compact electrocardiograms (ECGs) are driving the segment's growth due to their rising adoption, especially in home healthcare.A high prevalence of cardiovascular diseases (CVD) in Canada wherein long-term ECG monitoring is necessary and with the introduction of wireless ECG, doctors can monitor and diagnose the patient remotely on time, further fueling the market's growth. For instance, in April 2022, Samsung Canada encouraged users to reach their wellness goals with advanced health monitoring tools, made available in Canada through the Samsung Health Monitor app, which included electrocardiogram (ECG) features.
Among the new technologies in cardiac devices, wearable smart devices equipped with heart-monitoring capabilities, such as bands, watches, and rings, have become the norm for many people in Canada. For instance, in February 2021, Fitbit Sense received an ECG feature that can assess heart rhythm for atrial fibrillation (AFib). With the rapid pace of technological advancements in cardiac monitoring, the segment is expected to see a surge over the forecast period.
Cardiac Rhythm Management Device Under Therapeutic and Surgical Devices Segment is Expected to Record the Highest CAGR in the Market During the Forecast Period
Cardiac rhythm management is a part of electrophysiology, which is used to manage the functioning of the heart. Cardiac rhythm management devices are small battery-powered devices that are placed inside the body and connected to the heart. There are two common types of cardiac rhythm management devices, pacemakers, and implantable cardioverter defibrillators. The factors influencing the segment's growth include the rising burden of cardiac rhythm disorders and the rising elderly population in the country.For instance, as per the Census of Population by Statistics Canada report published in April 2022, there were over 861,000 people aged 85 and older in 2021, more than twice the number compared to the past years. As atrial fibrillation is the most common arrhythmia in the elderly over the age of 80 years, the rising elderly population is likely to increase the demand for cardiac rhythm management devices and boost the market through this segment.
New investments by the government in cardiac rhythm management devices help expand the services in the country and boost the market. For instance, in March 2021, the Rankin government invested USD 700,000 to equip all public schools in Nova Scotia, Canada, with automated external defibrillators used to help those in cardiac arrest. Hence, new investments and initiatives by the government are helping expand the cardiac rhythm management services and drive the market through this segment.
Competitive Landscape
The Canadian cardiovascular devices market is competitive, with the presence of several market players. The major players have established themselves in specific segments of the market. Some of the players currently dominating the market are Abbott Laboratories, Boston Scientific Corporation, Medtronic, Cardinal Health, Biotronik, and Siemens Healthcare GmbH.Additional benefits of purchasing the report:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Abbott Laboratories
- Boston Scientific Corporation
- Cardinal Health
- Biotronik
- Medtronic
- Edwards Lifesciences
- Siemens Healthcare GmbH
- General Electric (GE Healthcare)
- W. L. Gore & Associates Inc.
- Canon Medical Systems Corporation