COVID-19 has significantly impacted the Japanese cardiovascular devices market due to the decrease in cardiovascular surgeries during the pandemic. According to the NCBI study in January 2021, many departments prohibited cardiological procedures, and the rate of cardiovascular procedures varied according to the pandemic scenario. The decline in procedures during the pandemic restricted the growth of the cardiovascular devices market due to less adoption of these devices. Hence, as the pandemic has subsided, the market is expected to gain traction in the forecast period due to the resumption of cardiovascular surgical procedures after the relaxation of strict regulation on elective surgeries, which will lead to market growth in the post-pandemic era.
The major factors driving the market growth in Japan are the increasing burden of cardiovascular diseases and the increased adoption of minimally invasive procedures.
According to the National Cerebral and Cardiovascular Center, Japan, in December 2021, the number of heart failure patients in Japan was estimated to be approximately 1.2 million. According to the same source, the prevalence rate of cardiovascular diseases increases with age, and the number of heart failure patients is expected to increase further in Japan, where people live to be 100 years old. The high prevalence of heart failure patients in Japan is anticipated to increase the adoption of cardiovascular devices, driving the market growth.
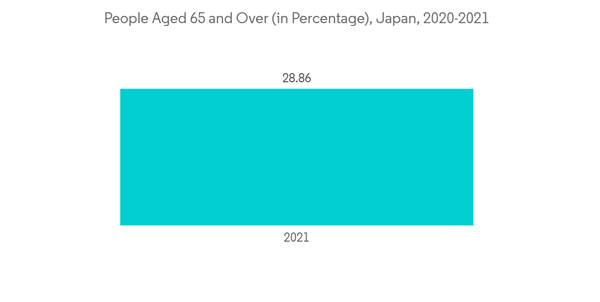
Furthermore, Japan has the world's fastest-aging population. According to World Data 2022, around 29% of Japan's population was 65 and above in 2021. As the geriatric population is more prone to chronic cardiovascular diseases, the rise in the geriatric population is expected to drive market growth in Japan.
In addition, rising initiatives from the key market players are also expected to drive market growth in Japan due to the rise in the sale of such products. For instance, in March 2022, Rampart IC signed an exclusive distribution agreement with Japan Lifeline (JLL), securing distribution to the Japanese market. The Rampart M1128 is a device developed for interventional cardiologists (ICs) but has found its application in several interventional specialties. Such distribution partnerships will lead to increased adoption of cardiovascular devices in Japan, driving the market growth.
Thus, the rising geriatric population and a high prevalence of heart diseases are expected to show significant growth over the forecast period. However, stringent regulatory policies and the high cost of instruments and procedures are expected to hinder the market growth.
Japan Cardiovascular Devices Market Trends
Electrocardiogram (ECG) Expected to Hold Significant Market Share
An electrocardiogram is expected to hold a significant market growth share over the forecast period. An electrocardiogram (ECG) records the electrical signals from the heart. It is a standard and painless test used to detect heart problems and monitor the heart's health. Such diagnostic features are helpful in cardiovascular procedures and are expected to drive the growth of the studied segment.Portable and compact electrocardiograms (ECGs) are witnessing greater adoption, especially in the home healthcare segment. Moreover, there has been an increase in the incidence of cardiovascular diseases (CVD), wherein long-term ECG monitoring is necessary. With the introduction of wireless ECG, doctors can monitor and diagnose patients remotely on time, further fueling the market growth.
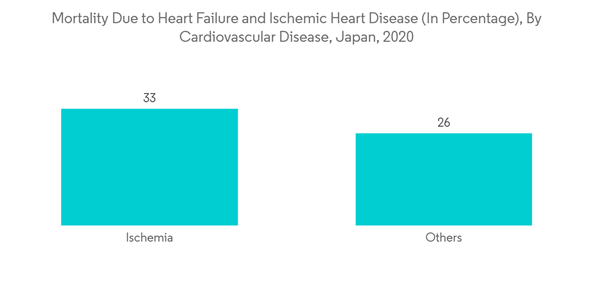
The factors driving the growth of this segment include the rise in heart diseases, such as heart failure, ischemic heart disease, and cardiac arrhythmias, among others. According to the National Cerebral and Cardiovascular Center, Japan, in December 2021, heart disease was a significant contributor to deaths in Japan, accounting for 15% of deaths. In addition, according to the same source, within heart diseases, heart failure contributes to 41%, and ischemia contributes to 33% of deaths in Japan. Therefore, such a high prevalence of heart failure in Japan will lead to increased adoption of ECG for diagnosing heart failure and ischemia, driving this segment's growth.
The rising initiatives from the key market players, such as the launch of ECG services in Japan, will lead to increased adoption of ECG devices in this region, driving this segment's growth. For instance, in September 2021, Nitto Denko Corporation, Astellas Pharma Inc., and M. Heart Co., Ltd. entered a memorandum of understanding concerning an ECG testing service. With this, Nitto will develop and manufacture the EG Holter, a novel disposable Holter ECG device. Astellas exclusively sells it in Japan.
Thus the above-mentioned factors are expected to contribute to the growth of the studied segment over the forecast period.
Remote Cardiac Monitoring Expected to Hold Notable Market Share
Remote cardiac monitoring is a method by which information from a patient's implantable rhythm management device can be communicated directly to a physician's office. Remote patient monitoring lets providers manage acute and chronic conditions and helps avoid patient travel costs and infection risks.The increasing geriatric population and the rising product launch approvals by various market players are expected to contribute to the growth of the studied segment. For instance, the Nippon article published in October 2022 mentioned that the number of seniors in Japan aged 65 years and older increased by 60,000 for a record total of 36.27 million. Such an increase in the geriatric population who are prone to cardiovascular diseases and require continuous monitoring of their heart condition is expected to contribute to the growth of the studied segment.
Additionally, the increasing product approvals are expected to contribute to the growth of the studied segment. For instance, in January 2022, MicroPort CRM received PMDA Japanese regulation for its Bluetooth-enabled Alizea pacemakers. Paired with the company's smart view connect home monitor, Alizea pacemakers can be remotely monitored.
Thus, the rising geriatric population and frequent product approvals are expected to contribute to the growth of the studied segment.
Japan Cardiovascular Devices Market Competitor Analysis
The Japanese cardiovascular devices market is moderately fragmented. The factors responsible for the market growth include the rise in technological advancements in the products and increasing advancements from the key market players, among others. Some players currently dominating the market are Abbott Laboratories, Boston Scientific Corporation, Edwards Lifesciences, Medtronic PLC, Cardinal Health Inc., Biotronik, Siemens Healthineers AG, and Canon Medical Systems Corporation.Additional benefits of purchasing the report:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Abbott Laboratories
- Boston Scientific Corporation
- Cardinal Health Inc.
- Medtronic PLC
- Edwards Lifesciences
- Biotronik
- Siemens Healthineers AG
- Canon Medical Systems Corporation