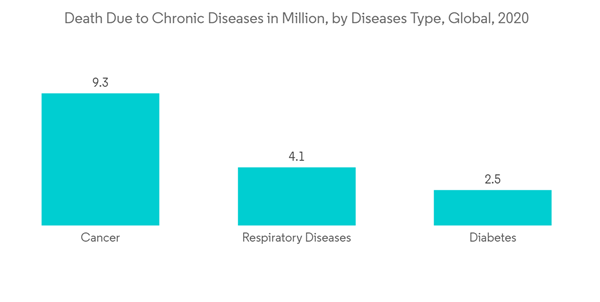
The market was impacted by the COVID-19 pandemic at a moderate growth rate. In the business world, the COVID-19 pandemic has created unprecedented economic uncertainty. While some businesses are relatively insulated due to low exposure, many others have been unable to avoid the pandemic's effects and are experiencing financial hardship. However, the number of surgeries declined during the pandemic, which hampered the market growth. According to a British Journal of Surgery report 2020, approximately 28 million surgeries have been canceled or postponed during COVID-19, substantially impacting the market.
The major factor driving the market is the increasing number of surgical processes. According to a research article by Prashant Bhandarkar et al., published in the World Journal of September 2020, estimated that about 5,000 surgeries are required to meet the surgical burden of diseases of 100,000 people in low- and middle-income countries (LMICs). Additionally, the reported rates of surgery vary from 295 in low- and middle-income countries (LMICs) to 23,000 in high-income countries (HIC) per 100,000 people. Thus, the increasing number of surgical procedures are expected to boost the market.
Additionally, technological advancements in medical devices are boosting the demand for reliable and efficient technologies during surgeries to decrease blood loss and infection rates and reduce operative time, which is the major reason for market growth. For instance, in December 2020, Bolder Surgical, a provider of surgical devices and instruments, introduced the CoolSeal Vessel Sealing platform, which performs minimally invasive surgery in pediatric patients safely and effectively. Rising investments in surgical instruments by hospitals and government initiatives are boosting the market.
However, the high cost of instruments involved and the complications arising after using the devices are the major drawbacks of the market.
Vessel Sealing Devices Market Trends
The Laparoscopic Segment Accounted for a Major Share of the Market
Laparoscopy is a surgical diagnostic method used to analyze the organs inside the abdomen. It is a minimally invasive procedure that requires only small incisions. It uses an instrument called a laparoscope to look at the abdominal organs. Vessel sealing devices are the major part of the laparoscopic surgeries performed.The major factors driving the growth of the segment are the increasing awareness of the advantages of laparoscopic surgeries over open surgeries and an increasing number of laparoscopic surgeries across the world.
However, many studies have described the safety and efficiency of vessel sealing devices in laparoscopic surgeries That will help in fueling the market over the projecting period. For instance, as per the study published in Surgical Innovation in June 2021, the title "Safety and Efficacy of Using Staplers and Vessel Sealing Devices for Laparoscopic Splenectomy: A Randomized Controlled Trial", vessel sealing devices have great safety and effectiveness over endoscopic staplers. Thus, the higher safety and effectiveness of vessel sealing devices boost the market.
Laparoscopy is a minimally invasive surgical procedure that allows a surgeon to view the inside of the abdomen and pelvis through small incisions rather than large skin incisions. Because of their convenience, minimally invasive procedures are becoming increasingly popular. Because minimally invasive surgeries (MIS) cause less post-operative pain, patients are prescribed lower doses of pain relievers, thereby driving the segment growth.
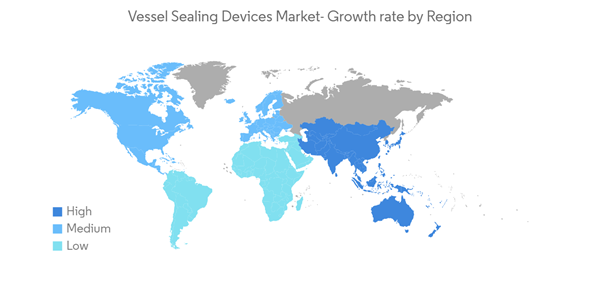
North America Dominates the Market, and It is Expected to do the Same during the Forecast Period
North America is expected to dominate the overall vessel sealing devices market throughout the forecast period. The largest share is mainly due to the presence of key players and an increase in per capita healthcare spending and expansion in the investment and product launches made by the major market players in the region. For instance, in June 2021, Johnson and Johnson's Ethicon launched ENSEAL X1 Curved Jaw Tissue Sealer for use in colorectal, bariatric surgery, gynecological, and thoracic procedures.Furthermore, there is a growing awareness among the population about the advantages of surgeries with such advanced technology. High adoption of surgical procedures with advanced energy in the United States, an increase in the number of approvals for new devices by the Food and Drug Administration, and a rise in the fraction of minimally invasive surgical procedures performed in the United States aid North America's high market share. According to the article published in the JAMA Network in December 2021, by Aviva S. Mattingly, BA1, in the United States, from January 1, 2019, to January 20, 2021, a total of 13,108,567 surgical procedures were performed. In 2019, there were 6,651,921 procedures, and in 2020, there will be 5,973,573 procedures. Thus, the increasing number of surgeries in the country drives the demand for vessel sealing devices, thereby boosting the market.
According to the American Cancer Society, Cancer Treatment and Survivorship Facts and Figures 2019-2021, on January 1, 2019, an estimated 16.9 million people in the United States had a history of cancer, and on January 1, 2030, the number of cancer survivors is expected to rise to more than 22.1 million, owing to population growth and aging. This is intended to drive the market in North America.
Thus, the increasing number of surgeries and technological advancements in medical devices boost the market in the region.
Vessel Sealing Devices Industry Overview
The vessel sealing devices market is moderately competitive, and it consists of several major players. Some of the market players are Medtronic PLC, B Braun Melsungen AG, Erbe Medical India Pvt. Ltd, Olympus Corporation, Ethicon US LLC (Johnson and Johnson), Boston Scientific Corporation, BOWA-electronic GmbH and Co. KG, CONMED Corporation, and Bolder Surgical Holdings Inc.Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Medtronic PLC
- B Braun Melsungen AG
- Erbe Medical India Pvt. Ltd
- Olympus Corporation
- Ethicon US LLC (Johnson & Johnson)
- Boston Scientific Corporation
- BOWA-electronic GmbH & Co. KG
- OmniGuide Holdings Inc.
- CONMED Corporation
- Hologic (Bolder Surgical Holdings Inc.)
- XCELLANCE Medical Technologies
- LAMIDEY NOURY.FR.