The COVID-19 pandemic affected the market studied significantly. The global lockdown measures impacted the diagnostic and surgical industries, which have also decreased public mobility. Owing to the fear of the risk of contamination, several surgeries which were deemed non-urgent were postponed in France during the initial phase of the pandemic. The IFSO in April 2020 recommended that all elective surgical and endoscopic cases for metabolic and bariatric surgery should be postponed during the pandemic period. Thus, the bariatric surgery market was impacted severely during the COVID-19 pandemic. However, with the restrictions lifted when the severity reduced, there was a resumption of bariatric surgeries in France. This considerably impacted the market growth. Hence, owing to the above-mentioned factors, the market studied is recovering from the damage caused by the pandemic and is expected to witness growth over the analysis period.
The key factors propelling the growth of the market studied are the increase in obese patients and the prevalence rate of type-2 diabetes and heart diseases.
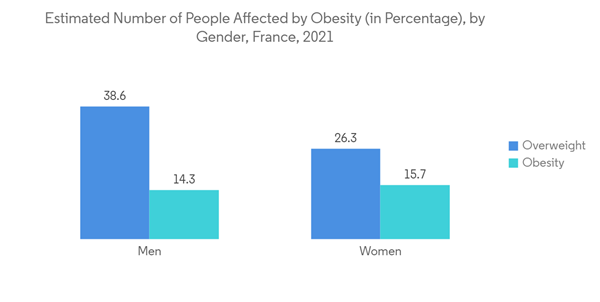
Obesity is considered a metabolic dysfunction and is often associated with a wide range of chronic illnesses that create a major healthcare burden. The major diseases caused by obesity include hypertension, Type-2 diabetes dyslipidemia, and other diseases. Therefore, obesity is undoubtedly one of the most common causes of diabetes. As bariatric surgery is one of the most efficient methods to control both, the demand for surgery is increasing in France. For instance, according to an article published by C3 Collaborating for Health in July 2021, around 1 in 7 people in France were obese, accounting for 17% of the total population. More than 8.57 million people in the country were obese in 2020. Thus, the large population affected by obesity in the nation is expected to boost the demand for bariatric surgeries. This is further expected to contribute to the growth of the market studied.
In addition, new technologies in bariatric surgery and the growing demand for minimally invasive surgical procedures are expected to act as key drivers for market growth during the forecast period. Thus, bariatric procedures are expected to increase in France, which may accelerate the market studied. Therefore, owing to an increase in obesity among the target population and the emergence of new technology, among other factors, the market studied is anticipated to witness growth over the forecast period. However, the high cost of the surgeries is predicted to hinder the growth of the market studied.
France Bariatric Surgery Market Trends
Stapling Devices Segment is Expected to Grow Significantly over the Forecast Period
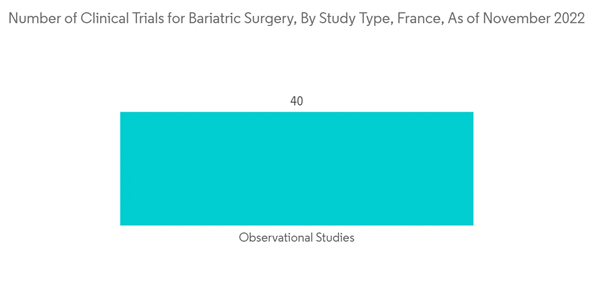
The stapling devices segment is expected to show a comparatively higher growth over the forecast period than other devices used in bariatric surgeries. These devices are used to remove a part of an organ, cut through and seal organs and tissues, and create connections between structures. Thus, most bariatric procedures are currently being performed with mechanical stapling devices, thereby contributing to the overall growth.It is one of the most used devices, as it tends to result in more significant and more consistent weight loss surgery results among patients and a more significant reduction in obesity-related health problems. Several clinical trials are being performed in France, focusing on bariatric surgeries. For instance, in April 2022, an interventional clinical trial was completed, which was conducted by Pr Fabienne d'Arripe-Longueville in collaboration with the Centre Hospitalier Universitaire de Nice and Association Azur Sport Santé, Nice, France since July 2020. Thus, such studies and instances focusing on bariatric surgeries are expected to create opportunities for bariatric surgery devices available in the country, fuelling segment growth.
Moreover, the recent product launches concerning surgical staplers in the country are expected to boost segment growth. For instance, in August 2021, Panther Healthcare launched their smart powered stapler platform that offers intelligent and automatic control over tissue compression, transection, and resection in Europe, including France. Such product launches are expected to add to the growth of the segment. Hence, the factors such as increasing pipeline studies and product launches are propelling the segment growth, and it is expected to continue its strong hold over the analysis period.
Gastric Balloons is Anticipated to be the Fastest Growing Segment Over the Forecast Period
A gastric balloon is an implantable device known as an intragastric balloon or a stomach balloon. This inflatable medical device is placed temporarily into the stomach to help the patients reduce weight. These are generally non-invasive weight-loss procedures.For a few of the over-obese population, diet and exercise programs do not help them lose a significant amount of weight. According to the September 2021 update by MFMER, gastric balloons are a better option for people concerned about weight loss without surgery, where a silicone balloon is placed in the patient’s stomach. This helps manage a proper diet by limiting food intake. Thus, owing to its advantages, the demand for gastric balloon procedures is growing among the obese population in France. This is expected to fuel the segment growth faster in France.
Furthermore, several service providers in France focus on providing better customer service. Most of the procedures are successful, with a small number of unsuccessful ones. According to the 2022 update by MyMediTravel, women typically lose around 2 to 3 kilograms, while men lose nearly 4 to 7 kilograms of their body weight in the first week. Most often, patients lose approximately 7% to 15% of their excess body weight during the period (almost 6 months) the gastric balloon is inside their stomach. Additionally, as per the source, the success rate of the procedure in France is moderate to high, where the total weight loss rate ranges from 30% to 47% of total procedures after a gastric balloon. Therefore, due to its benefits and increasing adoption rate in France, the segment is predicted to witness growth over the forecast period.
However, gastric balloons are not covered under any national health service in France. According to the Millennium Obesity Montpellier - Obesity treatment center 2022 update, the cost of the procedures is around EUR 1,500, and the patient needs to pay out-of-pocket for gastric balloon procedures. This is limiting the growth of the segment in the country.
France Bariatric Surgery Market Competitor Analysis
The French bariatric surgery market is moderately competitive. The manufacturers are innovating new technologies, which are non-invasive, to reduce patients’ time in the hospitals, decreasing the chances of infection and saving costs. Additionally, smaller players are implementing strategies, such as acquisitions, to expand their product portfolios and gain roots in the industry. Some of the major players in the nation include Apollo Endosurgery Inc., Ethicon Inc. (Johnson & Johnson), Conmed Corporation, COVIDien PLC (Medtronic PLC), and TransEnterix Inc.Additional benefits of purchasing the report:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Apollo Endosurgery Inc.
- Johnson & Johnson (Ethicon Inc.)
- Conmed Corporation
- Medtronic PLC (Covidien)
- TransEnterix Inc.
- B. Braun SE
- Olympus Corporation
- Stryker
- Peters Surgical
- Sterifil