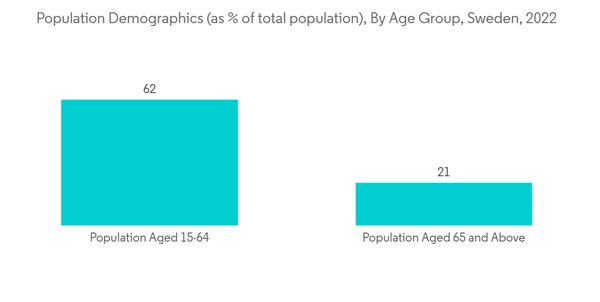Key Highlights
- The COVID-19 pandemic significantly impacted neurology care in Sweden during the early phase, which also impacted the usage of neurology devices. For instance, according to an article published by PubMed in January 2023, a study was conducted in Southern Sweden, which showed that 74% of the patients admitted due to COVID-19 had one or more neurological complications. Thus, the COVID-19 pandemic significantly impacted the market initially; however, as the pandemic subsided, people are having fewer neurological complications associated with COVID-19. Hence, the studied market is expected to have stable growth during the forecast period of the study.
- The key factors propelling the growth of this market are an increase in the incidence of neurological disorders, huge investments by private players in neurology devices, and an increase in R&D expenditure in the field of neuro therapies.
- The rising prevalence of neurological disorders, such as Parkinson's disease, Alzheimer's disease, epilepsy, and brain diseases, among others, is the major factor driving the market growth. For instance, according to an article published by PubMed in September 2022, a study was conducted in Sweden that showed that hospital-treated infections were associated with an increased risk of Alzheimer's diseases and Parkinson's diseases in Sweden, primarily among Alzheimer's disease and Parkinson's diseases cases diagnosed before 60 years. Such studies suggest that infectious events can be a trigger or amplifier of a pre-existing disease process, leading to the clinical onset of neurodegenerative disease at a relatively early age. Thus, the high burden of these neurodegenerative diseases in the country is expected to boost market growth.
- Moreover, according to an article published by PubMed Central in October 2022, a study was conducted in Sweden that showed an increased risk of epilepsy following both bacterial and viral central nervous system infections, most prominently after herpes simplex encephalitis and brain abscesses which can both cause loss of brain tissue and cortical scarring. Thus, the increased risk of epilepsy is expected to increase the disease burden, which is further expected to boost the usage of neurology devices in Sweden.
- Thus, the aforementioned factors, such as the rising prevalence of neurological disorders and the increasing risk factors, are expected to boost the market growth during the forecast period of the study. However, the high cost of equipment and stringent regulations and guidelines for new devices are expected to impede market growth.
Sweden Neurology Devices Market Trends
Neurostimulation Devices Segment is Expected to Have a Significant Growth During the Forecast Period
- Neurostimulation devices are considered implantable and programmable medical devices that deliver electrical stimulation effectively to specific parts of the patient's brain, spinal cord, or peripheral nervous system to help treat various conditions, including chronic pain and movement disorders, among others. Hence, with increasing incidences of various diseases such as overactive bladder and urinary incontinence, the neurostimulation devices segment is expected to grow during the forecast period.
- According to an article published by PubMed in May 2022, a study was conducted in Sweden, which showed that all the types of incontinence, except severe isolated gas incontinence, increased with age up to 64 years. The estimated probability of fecal incontinence in the study population was 8.8% at age 25 years and 17.6% at age 64. Thus, the high prevalence of fecal incontinence is expected to boost segment growth during the forecast period of the study, as neurostimulation procedures are widely used in the treatment of such diseases.
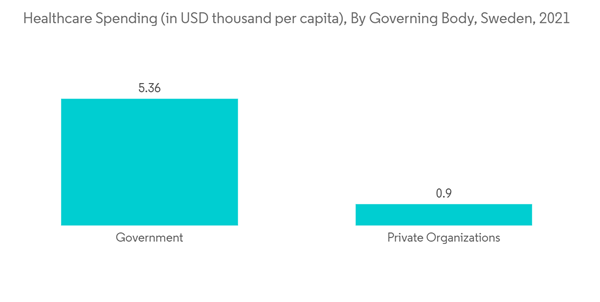
- Furthermore, according to an article published by SpringerLink in March 2022, a study was conducted in Sweden that mentioned that urinary incontinence (UI) is defined as the complaint of involuntary loss of urine and has an estimated prevalence of about 25 to 45% among adult women. Thus, the high burden of urinary incontinence in the country is expected to boost the adoption of neurostimulation devices. Moreover, the rising healthcare spending in the country is also expected to boost segment growth, as it will lead to increased access to advanced neurostimulation devices in the country.
- Hence, the aforementioned factors such as the rising prevalence of fecal incontinence and urinary incontinence and the rising healthcare expenditure, are expected to boost the segment growth during the forecast period of the study.
Neurosurgery Devices Segment is Expected to Have a Significant Growth During the Forecast Period
- Neurosurgery devices are devices that are used for the surgical treatment of various brain and spinal cord diseases. These devices are mainly used for the treatment of various neurological diseases such as dystonia, Alzheimer's disease, Parkinson's disease, and epilepsy, among others. The rising prevalence of such neurological disorders in the UAE and the rising geriatric population is expected to boost segment growth.
- For instance, according to an article published by the International Parkinson and Movement Disorder Society in July 2022, a study was conducted in Sweden, which showed that the pooled prevalence of cervical dystonia was 11.75 per 100,000 people in Sweden. Thus, the high prevalence of dystonia in the country is expected to boost segment growth.
- Furthermore, according to an article published by the Journal of Scientific Reports in October 2022, Parkinson's disease (PD) is often associated with dementia. The affected individual loses their ability to function independently and faces a reduced survival and health-related quality of life. The article also reported that the cumulative prevalence of Parkinson's disease in the last 10 years in Sweden was 54%. Thus, the high burden of Parkinson's disease in the country is expected to boost the usage of neurosurgery devices.
- Hence, the aforementioned factors, such as the rising prevalence of dystonia and Parkinson's disease and the rising geriatric population, are expected to boost the segment growth during the forecast period of the study.
Sweden Neurology Devices Industry Overview
The Sweden neurology devices market is moderately consolidated in nature. There are market participants involved in R&D for the development of advanced stimulation and neuromodulation techniques with minimally invasive procedures that are preferable to the patients. Furthermore, many private companies are showing great interest in developing novel neurological devices to earn huge profits. Some of the key market players are B. Braun SE, Medtronic PLC, Abbott Laboratories, Boston Scientific Corporation, and Stryker Corporation, among others.Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- B. Braun SE
- Stryker Corporation
- Boston Scientific Corporation
- Medtronic PLC
- Abbott Laboratories
- Nihon Kohden Corporation
- Smith & Nephew
- Integra LifeSciences Corporation