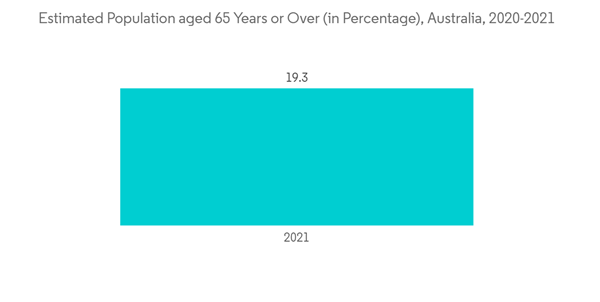The COVID-19 pandemic led to a substantial reduction in gastrointestinal, ENT Surgical, and other endoscopic procedures, which caused a backlog of elective procedures in the country. For instance, a research article published in the Medical Journal of Australia in June 2022 stated that during the pandemic, elective medical procedures, and population cancer screening programs, including colonoscopy and other public measures were suspended and a 42% reduction was reported in the endoscopic procedures in 2020 compared to the previous year. Such a scenario caused a decline in endoscopic procedure volumes in the country. Furthermore, the study stated that in Melbourne, the pandemic restricted non-urgent endoscopy during 2020. The patients were scheduled for elective endoscopy and were re-triaged for priority by the Gastroenterological Society of Australia's COVID-19 guidelines. However, the studied market has been recovering well over the last two years since the restrictions were lifted.
The increasing preference for minimally invasive surgeries and the rise in the prevalence of diseases that require endoscopy procedures are the major drivers for the market growth. For instance, as per an article published in the Australian Journal of Otolaryngology, endoscopic ear surgery gained popularity in recent years in Australia. It also conducted a survey to access the popularity of endoscopic ear surgery in Australia which demonstrated positive results toward the adoption of endoscopic ear surgery in the country in 2021. Also, a research study published in January 2022, indicated a rising incidence of gastrointestinal diseases among the young population (18 to 50 years) in Australia. Such instances are anticipated to positively impact the market, thereby propelling the market growth over the forecast period.
Furthermore, the strategic initiatives by key market players such as collaborations, product launches, and research and development activities are anticipated to propel market growth. For instance, in October 2021, Stryker established a research and development lab in Queensland, Australia. The new R&D Lab will also partner with Stryker's CMF, Endoscopy, Instruments, Joint Replacement, Medical, Spine, and Trauma & Extremities divisions to focus on research that creates technological platforms that can be used across service lines. These factors are projected to increase the supply of endoscopy devices in Australia, thereby boosting the market growth.
However, the high cost of sophisticated endoscopy devices is likely to hinder market growth over the forecast period.
Australia Endoscopy Devices Market Trends
Cardiology Segment is Expected to Witness a Significant Market Share Over the Forecast Period
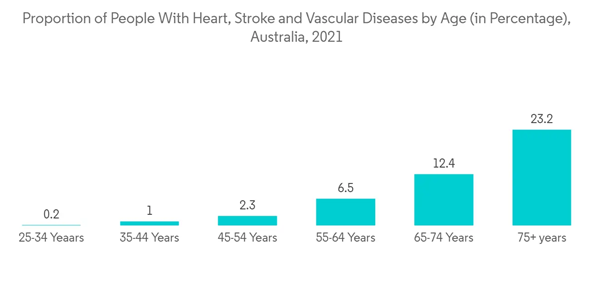
Cardiology endoscopy is used for performing minimally invasive cardiac surgeries and examining the chambers of the heart.The increasing number of cardiovascular diseases in Australia is projected to increase the adoption of endoscopy procedures for diagnostic purposes, thereby driving the segment's growth. For instance, according to the Australian Bureau of Statistics, an estimated 571,000 Australians aged 18 and over (2.9% of the adult population) had chronic heart diseases in 2021. The high burden of cardiovascular diseases in the country is projected to boost segment growth.
Furthermore, in April 2021, the Australian Government invested around USD 12.0 million to support and expand the Heart of Australia program, including the delivery of mobile medical clinic services across northern Queensland. The mobile medical clinic provides access to cardiology, cardiac monitoring, and other services. The government investment in providing easy and effective cardiovascular services is projected to boost the demand for cardiac endoscopy, thereby propelling the segment growth over the forecast period.
Flexible Endoscope is Expected to Hold a Significant Market Share Over The Forecast Period
Flexible endoscopes are minimally invasive devices inserted into natural openings of the body, to observe the internal organ or tissue in detail. The flexible endoscope segment is expected to rise during the forecast period owing to the enormous burden of chronic diseases, the increasing geriatric population, technological advancements, and the increasing preference for minimally invasive surgical treatment.Besides, the technological advancements in products and launches by key players are anticipated to boost the segment's growth in the country. For instance, in December 2021, PENTAX Medical expanded its offering by launching ONE Pulmo single-use bronchoscope, a flexible endoscope. The company also planned to launch Slim DEC Duodenoscope (ED32-i10; OE-A65) across Europe in the upcoming year. The Slim DEC is a flexible duodenoscope that provides consistent therapeutic performance, and outstanding visualization reduces the risk of cross-contamination and helps pediatric patients and adults with narrow anatomies. Such launches are expected to propel the segment's growth during the forecast period.
Australia Endoscopy Devices Industry Overview
The Australian endoscopy devices market is consolidated in nature due to the presence of a few companies operating at the country level. The competitive landscape includes an analysis of a few international and local companies that hold market shares and are well known. Some key market players are Fujifilm Holdings Corporation, Hoya Group (PENTAX Medical), Stryker Corporation, Medtronic PLC, Olympus Corporation, B. Braun Melsungen AG, Johnson & Johnson (Ethicon), STERIS PLC, Karl Storz GmbH & Co. KG, and Ambu A/S.Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Olympus Corporation
- Medtronic Plc
- Hoya Group (PENTAX Medical)
- Karl Storz GmbH & Co. KG
- Stryker Corporation
- FUJIFILM Holdings Corporation
- B. Braun Melsungen AG
- Johnson & Johnson (Ethicon)
- Ambu A/S
- STERIS PLC