The COVID-19 pandemic had a significant impact on the growth of the market due to a reduction in patients' visits to hospitals and clinics as a preventative step to avoid contracting the virus. The decreased volume of surgical procedures due to the strict regulations imposed during the peak stages of the pandemic impacted the demand for implantable drug delivery devices. The pandemic-related regulations affected the supply chain of implantable drug delivery devices, thereby affecting the market's growth. However, the relaxation of strict guidelines on various surgical procedures involving the implantation of drug-delivery devices during the post-pandemic is expected to drive the growth of the market over the forecast period.
The implantable drug delivery devices market is anticipated to grow due to the increasing prevalence of target diseases such as diabetic retinopathy, cancer, cardiovascular disorders, and other chronic diseases. As per an NCBI article published in November 2021, the prevalence of diabetic retinopathy (DR) among persons with diabetes was 16.9%, the majority of sight-threatening DR (STDR) was 3.6%, and the bulk of mild retinopathy was 11.8%. Thus, the increasing cases of diabetic retinopathy are expected to demand more use of implantable drug delivery devices for the treatment, which is expected to drive the market's growth. As per an MHAF article published in June 2022, HIV prevention tools are inserted, injected, infused, or implanted in a person’s body from once a month to once a year to provide sustained protection from acquiring HIV. These implants are small devices implanted in the body and release an anti-HIV drug at a controlled rate for continuous protection from HIV over time. Thus, the increasing AIDS infections are also expected to promote the usage and growth of implantable drug-delivery devices.
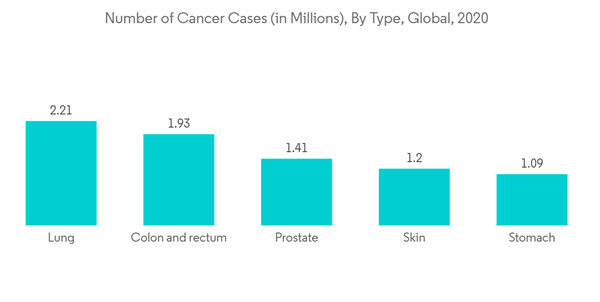
The increase in chronic diseases is also expected to drive the growth of the market over the forecast period. As per the 2021 report of the International Agency for Research on Cancer which estimated the incidence and mortality of 36 cancer in 185 countries, globally, there were an estimated 19.3 million new cases of cancer diagnosed in 2020 all over the world. The same source also mentioned that over 10.1 million cases were reported in males and 9.3 million in females from the total diagnosed cancer cases. Such a rise in chronic diseases is expected to contribute to the growing demand for implantable drug-delivery devices, thereby driving the growth of the market over the forecast period.
However, factors such as the high cost of devices and surgery costs and the low rate of adoption are hindering the growth of the implantable drug delivery devices market.
Implantable Drug Delivery Devices Market Trends
Drug Infusion Pumps Segment is Expected to Hold a Significant Market Share Over the Forecast Period
Infusion pumps are devices used to deliver fluids into a patient’s body in a controlled manner. They can supply medications such as insulin or other hormones, antibiotics, chemotherapy drugs, and pain relievers. Infusion pumps play an important role in delivering drugs in a controlled manner for the corresponding diseases. According to Johns Hopkins Medicine 2022, hospitals use infusion pumps to deliver fluids, nutrients, and medicines to roughly 90% of their patients. Across the United States, healthcare facilities invest in these infusion pumps as they can be programmed to deliver exact dosages at a controlled rate during customizable time intervals. Thus, the comfortable usage of infusion pumps to deliver the drugs is expected to promote their usage and drive the market's growth.According to the WHO data updated in February 2022, each year, approximately 400,000 children develop cancer. The most common cancers vary between countries. In cancer, the infusion pump is connected to your central venous catheter (CVC), and continuous infusion of the chemotherapy flows from the balloon, through the infusion line, and your CVC tubing into the patient’s bloodstream. Thus, the increasing cases of cancer are expected to demand more chemotherapy treatment and drug delivery devices and promote the segment's growth in the market.
The rising number of chronic disorders, such as diabetes cases, and increasing investments in diabetes care and management have helped adopt insulin infusion pumps. Technological advancements, and rising product approvals, coupled with increasing R&D initiatives on gastrointestinal disorders, cancer, neurological problems, and AIDS, are providing lucrative opportunities for infusion pumps in the market. For instance, in August 2022, Baxter International received 510(k) clearance from United States FDA for its new Novum IQ syringe infusion pump with Dose IQ safety software, representing Baxter’s latest developments for infusion therapy.
Therefore, the above-mentioned factors, such as the prevalence of target diseases and innovative product launches, are expected to promote the growth of the segment in the market studied during the forecast period.
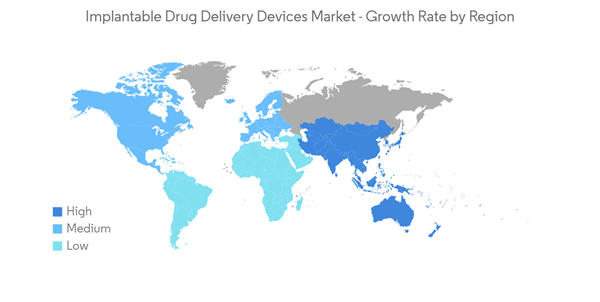
North America is Expected to Hold a Significant Share in the Implantable Drug Delivery Devices Market Over the Forecast Period
North America is expected to hold a significant share in the growth of the market over the forecast period. The increasing prevalence of chronic diseases, rising demand for minimally invasive procedures, and the quick adoption of technologically advanced products are expected to contribute to the market's growth in North America over the forecast period.As per the GLOBOCAN 2020 report, the new breast cancer cases in the United States, Canada, and Mexico were 253,465, 28,026, and 29,929, respectively, in 2020. Such prevalence of cancer in North American countries is expected to drive the demand for implantable drug delivery devices, thereby contributing to the growth of the market. Similarly, the incidence of autoimmune disorders is also expected to contribute to the growth of the market. According to the National Multiple Sclerosis Society report published in September 2021, about 2.8 million people are living with multiple sclerosis, of which about 1 million are living in the United States. Further, as per the same source, the epidemiology of multiple sclerosis has increased over the years. A significant population living with multiple sclerosis were children and young people of the age of under 18 years. Therefore, the demand for efficient implantable drug delivery devices is expected to increase in this region, thereby contributing to the growth of the market.
The increasing product approvals and product launches are expected to contribute to the growth of the market. For instance, in October 2021, Genentech received approval from the FDA for Susvimo, previously called the Port Delivery System (PDS), with ranibizumab, the wet or neovascular age-related macular degeneration treatment. Furthermore, in February 2021, United Therapeutics Corporation commercially launched the Remunity Pump for Remodulin for patients with pulmonary arterial hypertension. Remunity is a small, discreet pump that delivers Remodulin in prefilled cassettes delivered directly to patients, offering significant improvements.
Thus, the above-mentioned factors, such as the prevalence of target diseases, the presence of major market players, and frequent product developments, are expected to contribute to the growth of the market in North America over the forecast period.
Implantable Drug Delivery Devices Market Competitor Analysis
The implantable drug delivery devices market is moderately fragmented and consists of several major market players. The competitive landscape includes an analysis of a few international and local companies that hold market shares and are well known. Some of the major market players include DSM Biomedical, Delpor Inc., Medtronic, Teleflex Incorporated, 3M Company, and others.Additional benefits of purchasing the report:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Allergan PLC
- Boston Scientific Corporation
- Bausch and Lomb Inc.
- Medtronic PLC
- Alcon Inc.
- Teleflex Incorporated
- DSM Biomedical
- Delpor Inc.
- 3M Company
- Biotronik Inc
- Terumo Corporation
- Bayer AG