COVID-19 had a profound impact on the glaucoma surgery devices market owing to the cancellations of elective procedures, including glaucoma surgery. For instance, an article published in the International Ophthalmology Journal in April 2022 stated that the surgical volume of glaucoma dropped by 30.1% at the tertiary glaucoma center of the University Hospital of Verona, Italy, in 2020. The study also mentioned a decline in Baerveldt tube implants of 4.9%. General anesthesia was used for just 24.3% of the surgeries, as compared to 41.5% in the pre-pandemic period. However, the market has been recovering well over the last two years since the restrictions were lifted. The rise in glaucoma surgeries worldwide to clear the backlogs of surgical procedures caused by the pandemic restrictions compensated for the market growth in the later phase of the pandemic. The rising awareness about glaucoma treatment and prevention worldwide in recent times is adding to the market's growth. For instance, a Glaucoma Awareness Walk was conducted by the newly established Dr. B. R. Ambedkar State Institute of Medical Sciences, Mohali, in March 2022 for the first time to spread awareness related to the disease's prevention and treatment options. The awareness campaign reported that in India, every eighth individual, or nearly 40 million people, has either glaucoma or is at risk of developing the disease, and 11.2 million Indians suffer from the disease, with 1.1 million being blind, including children. Therefore, the high burden of the disease is anticipated to boost the utility of glaucoma surgery devices during the forecast years.
In addition, the rising prevalence of glaucoma, the increasing geriatric population, and the growing adoption of surgical treatments for glaucoma are actively affecting the growth of the studied market.
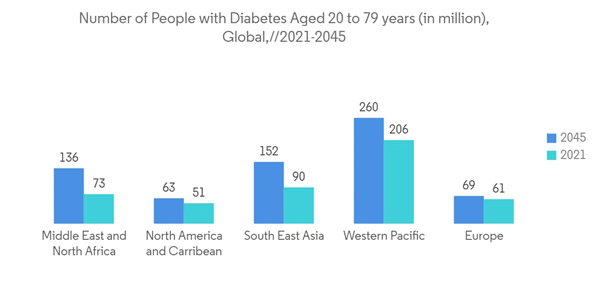
According to the 2022 update from the World Glaucoma Association (WGA), 79.6 million people worldwide were predicted to have glaucoma in 2020. It was estimated that there would be 111.8 million people with glaucoma by 2040. It also stated that at least half of glaucoma patients are unaware of their condition.90% of glaucoma cases in some developing countries go undetected. Thus, the surge in glaucoma is associated with the rise in glaucoma surgery, which is ultimately projected to boost the glaucoma surgery device market.
Also, according to the World Population Prospect 2022 report, the population aged 65 years and over in Sub-Saharan Africa is predicted to be 3.0% in 2022 and projected to reach 3.3% by 2030; in Northern Africa and Western Asia, it is predicted to be 5.5% in 2022 and projected to reach 7.0% by 2030; in Central and Southern Asia, it is estimated to be 6.4% in 2022 and projected to reach 8.1% by 2030; and in Europe and Northern America, it is estimated to be 18.7% in 2022 and projected to reach 22.0% by 2030. Since glaucoma is more common in the geriatric population, the growth of the market segment is likely to increase as the population increases.
Furthermore, glaucoma is the most common cause of irreversible blindness. Various national and international organizations are actively involved in spreading awareness regarding management and treatment options. For instance, the WGA and the World Glaucoma Patient Committee (WGPC) collaborated to commemorate World Glaucoma Week (WGW) from March 7 to March 13, 2021, in an effort to increase public awareness of glaucoma on a global level. Such global awareness initiatives are ultimately projected to boost glaucoma surgery worldwide, which in turn is estimated to propel the market's growth.
Therefore, owing to the aforementioned factors, it is anticipated that the studied market will witness growth over the analysis period. However, the availability of alternative treatment methods is likely to impede market growth.
Glaucoma Surgery Devices Market Trends
Minimal Invasive Glaucoma Surgery is Expected to Witness Growth Over The Forecast Period
Minimally invasive glaucoma surgery is also known as micro-invasive glaucoma surgery (MIGS). The segment is expected to grow owing to the increasing number of glaucoma patients globally, the rise in the geriatric population, and technological advancements in devices.According to a StatPearls article updated in August 2022, glaucoma accounts for a total of 8% of blindness worldwide. Treatments for mild to moderate glaucoma using micro-invasive glaucoma surgery (MIGS) are intended to reduce the burden of pre- and post-operative care while providing an acceptable intraocular pressure (IOP) decrease for patients. With the help of minimally invasive surgery, surgeons and patients are both at ease, and it offers good postoperative recovery with minimal bedtime for patients. The advantages of minimally invasive glaucoma surgery over traditional surgical procedures are majorly impacting the growth of the segment.
Moreover, the strategic initiatives adopted by key players, like technological advancements, product approvals, and launches related to the devices used in minimally invasive glaucoma surgeries, are also expected to boost segment growth. For instance, in August 2022, Glaukos Corporation, an ophthalmic medical technology and pharmaceutical company, received 510(k) clearance from the United States FDA for the iStent infinite trabecular micro-bypass system. The device is indicated for reducing elevated intraocular pressure (IOP) in patients with primary open-angle glaucoma uncontrolled by prior medical and surgical therapy. The device is efficient in glaucoma surgeries. Such launches of technologically advanced products are estimated to propel segment growth over the forecast period.
North America is Expected to Expected to Hold a Major Share in The Glaucoma Surgery Devices Market Over The Forecast Period.
North America is expected to dominate the market owing to factors such as the rising incidence of glaucoma, the growing geriatric population, better healthcare infrastructure, awareness among people and healthcare industry stakeholders about available technologies, and the strong presence of industry players in the region.According to the 2022 update from the World Glaucoma Association (WGA), after heart attacks and cancer, glaucoma-associated blindness will be the third-most feared medical condition in the United States in 2022. Thus, the fear and increasing prevalence of glaucoma in the country are estimated to propel glaucoma surgical procedures, which in turn will propel market growth in the region.
Also, the United Health Foundation 2021 report stated that more than 54 million adults aged 65 and older live in the United States, which accounts for about 16.5% of the nation’s population. The total number of adults aged 65 and older is projected to reach 85.7 million by 2050, or roughly 20% of the overall country's population. Since the geriatric population is prone to diseases like glaucoma and cataracts, the demand for glaucoma surgery is likely to excel in the region, which is projected to boost the growth of the glaucoma surgery devices market in the region.
Furthermore, increasing healthcare expenditure and the presence of well-established healthcare infrastructure are also fueling the growth of the overall regional market to a large extent. For instance, according to Fighting Blindness Canada's May 2021 article, the emerging crisis of preventable blindness in Canada costs almost USD 33 billion annually. The high burden of the country on disease treatment indicates the high demand for glaucoma surgery, which is projected to propel market growth in the region.
Therefore, owing to the above-mentioned factors, the growth of the studied market is anticipated in the North American region.
Glaucoma Surgery Devices Market Competitor Analysis
The glaucoma surgery devices market is slightly consolidated in nature due to the presence of a few companies operating globally as well as regionally. The competitive landscape includes an analysis of a few international as well as local companies that hold large market shares and are well known. Some key market players are Abbott Laboratories Inc., Lumenis, Alcon, Glaukos Corporation, ASICO, LLC., Katalyst Surgical LLC., Allergan, Ziemer Ophthalmic Systems, and Carl Zeiss Meditec.Additional benefits of purchasing the report:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Abbott Laboratories Inc.
- Lumenis
- Alcon
- Glaukos Corporation
- ASICO LLC
- Katalyst Surgical LLC
- Allergan
- Ziemer Ophthalmic Systems
- Carl Zeiss Meditec
- Altomed
- Nova Eye Medical
- Johnson and Johnson