Global In Vitro Diagnostics (IVD) Quality Control Market - Key Trends and Drivers Summarized
Why Is In Vitro Diagnostics (IVD) Quality Control Critical for Accurate Healthcare Diagnostics?
In Vitro Diagnostics (IVD) quality control plays a pivotal role in ensuring the accuracy, reliability, and consistency of diagnostic tests performed in laboratories. IVD tests are essential tools for diagnosing diseases, monitoring health conditions, and guiding treatment decisions. These tests include blood tests, molecular diagnostics, and microbiological assays, and they are used across various medical disciplines. Quality control (QC) measures in IVD are crucial because they help detect errors or inconsistencies in test results, which could otherwise lead to incorrect diagnoses or treatments. QC in IVD involves the regular calibration of diagnostic equipment, validation of test procedures, and monitoring of test performance through control samples, ensuring that the diagnostic outcomes are reliable and trustworthy.How Are Technological Advancements Shaping the IVD Quality Control Market?
The IVD quality control market is undergoing significant transformation, thanks to advancements in technology. Automation in laboratories is increasingly being adopted, reducing human error and improving the accuracy of test results. The development of advanced control materials, including multi-analyte controls, has improved the ability to monitor multiple parameters simultaneously, ensuring comprehensive quality checks. Additionally, molecular diagnostics and point-of-care testing are growing rapidly, leading to the need for more sophisticated quality control processes that can handle the complexity and specificity of these newer technologies. Moreover, AI-driven data analysis is being integrated into IVD quality control systems, enabling laboratories to predict potential issues and adjust processes in real-time, further enhancing the reliability of diagnostics.How Do Market Segments Define the Growth of the IVD Quality Control Market?
Product types include quality control materials, data management software, and instruments, with quality control materials holding the largest market share due to their wide applicability in various diagnostic tests. Applications of IVD quality control include clinical chemistry, immunoassay, hematology, and molecular diagnostics, with molecular diagnostics witnessing the fastest growth as personalized medicine and genetic testing gain traction. End-users include hospitals, clinical laboratories, and academic and research institutions, with clinical laboratories being the largest segment due to their high volume of testing and the critical need for consistent, reliable results. The market is also expanding globally, with North America and Europe leading in adoption, while the Asia-Pacific region is experiencing rapid growth due to increasing healthcare infrastructure investments.What Factors Are Driving the Growth in the IVD Quality Control Market?
The growth in the IVD quality control market is driven by several factors, including the increasing demand for accurate diagnostics, the rising prevalence of chronic and infectious diseases, and stringent regulatory requirements for diagnostic accuracy. As healthcare systems strive to provide better outcomes, the need for precise diagnostic tools is paramount, pushing laboratories to invest in robust quality control measures. The increasing incidence of diseases such as cancer, diabetes, and cardiovascular conditions is also contributing to higher demand for diagnostic tests, thus driving the need for quality control in IVD. Furthermore, regulatory agencies such as the FDA and the European Medicines Agency (EMA) have implemented stricter guidelines, mandating rigorous QC practices to ensure patient safety and test reliability, further fueling market growth.Report Scope
The report analyzes the In Vitro Diagnostics (IVD) Quality Control market, presented in terms of market value (US$). The analysis covers the key segments and geographic regions outlined below:- Segments: Application (Clinical Chemistry, Immunochemistry, Molecular Diagnostics, Hematology, Other Applications); End-Use (Hospitals, Clinical Laboratories, Other End-Uses).
- Geographic Regions/Countries: World; United States; Canada; Japan; China; Europe (France; Germany; Italy; United Kingdom; Spain; Russia; and Rest of Europe); Asia-Pacific (Australia; India; South Korea; and Rest of Asia-Pacific); Latin America (Argentina; Brazil; Mexico; and Rest of Latin America); Middle East (Iran; Israel; Saudi Arabia; United Arab Emirates; and Rest of Middle East); and Africa.
Key Insights:
- Market Growth: Understand the significant growth trajectory of the Clinical Chemistry segment, which is expected to reach US$399.9 Million by 2032 with a CAGR of 4.9%. The Immunochemistry segment is also set to grow at 3.6% CAGR over the analysis period.
- Regional Analysis: Gain insights into the U.S. market, valued at $290.8 Million in 2025, and China, forecasted to grow at an impressive 7.7% CAGR to reach $301.0 Million by 2032. Discover growth trends in other key regions, including Japan, Canada, Germany, and the Asia-Pacific.
Why You Should Buy This Report:
- Detailed Market Analysis: Access a thorough analysis of the Global In Vitro Diagnostics (IVD) Quality Control Market, covering all major geographic regions and market segments.
- Competitive Insights: Get an overview of the competitive landscape, including the market presence of major players across different geographies.
- Future Trends and Drivers: Understand the key trends and drivers shaping the future of the Global In Vitro Diagnostics (IVD) Quality Control Market.
- Actionable Insights: Benefit from actionable insights that can help you identify new revenue opportunities and make strategic business decisions.
Key Questions Answered:
- How is the Global In Vitro Diagnostics (IVD) Quality Control Market expected to evolve by 2032?
- What are the main drivers and restraints affecting the market?
- Which market segments will grow the most over the forecast period?
- How will market shares for different regions and segments change by 2032?
- Who are the leading players in the market, and what are their prospects?
Report Features:
- Comprehensive Market Data: Independent analysis of annual sales and market forecasts in US$ Million from 2025 to 2032.
- In-Depth Regional Analysis: Detailed insights into key markets, including the U.S., China, Japan, Canada, Europe, Asia-Pacific, Latin America, Middle East, and Africa.
- Company Profiles: Coverage of players such as Abbott Laboratories, Becton, Dickinson And Company (BD), Bio-Rad Laboratories, Inc., Danaher Corporation, Eurotrol B.V. and more.
- Complimentary Updates: Receive free report updates for one year to keep you informed of the latest market developments.
Some of the companies featured in this In Vitro Diagnostics (IVD) Quality Control market report include:
- Abbott Laboratories
- Becton, Dickinson And Company (BD)
- Bio-Rad Laboratories, Inc.
- Danaher Corporation
- Eurotrol B.V.
- F. Hoffmann -La Roche Ltd.
- Fortress Diagnostics Limited
- Helena Biosciences Europe
- Maine Molecular Quality Controls, Inc.
- Merck KGaA
- Omega Diagnostics Group Plc
- Ortho Clinical Diagnostics
- Randox Laboratories Ltd.
- Seracare Life Sciences Inc.
- Sero As
- Siemens Ag
- Sun Diagnostics, LLC.
- Technopath Clinical Diagnostics
- Thermo Fisher Scientific Inc.
- Zeptometrix Corporation
Domain Expert Insights
This market report incorporates insights from domain experts across enterprise, industry, academia, and government sectors. These insights are consolidated from multilingual multimedia sources, including text, voice, and image-based content, to provide comprehensive market intelligence and strategic perspectives. As part of this research study, the publisher tracks and analyzes insights from 2,866 domain experts. Clients may request access to the network of experts monitored for this report, along with the online expert insights tracker.Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Abbott Laboratories
- Becton, Dickinson And Company (BD)
- Bio-Rad Laboratories, Inc.
- Danaher Corporation
- Eurotrol B.V.
- F. Hoffmann -La Roche Ltd.
- Fortress Diagnostics Limited
- Helena Biosciences Europe
- Maine Molecular Quality Controls, Inc.
- Merck KGaA
- Omega Diagnostics Group Plc
- Ortho Clinical Diagnostics
- Randox Laboratories Ltd.
- Seracare Life Sciences Inc.
- Sero As
- Siemens Ag
- Sun Diagnostics, LLC.
- Technopath Clinical Diagnostics
- Thermo Fisher Scientific Inc.
- Zeptometrix Corporation
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 275 |
| Published | May 2026 |
| Forecast Period | 2025 - 2032 |
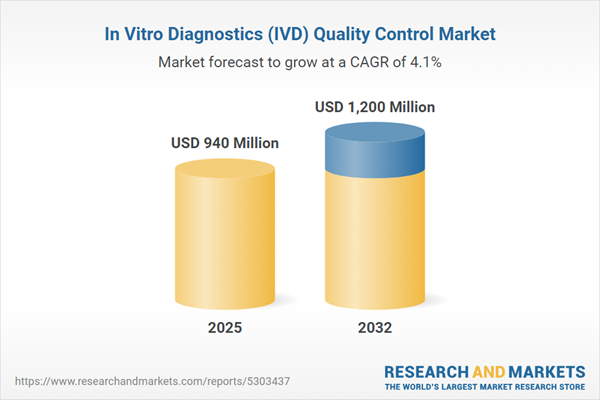
| Estimated Market Value ( USD | $ 940 Million |
| Forecasted Market Value ( USD | $ 1200 Million |
| Compound Annual Growth Rate | 4.1% |
| Regions Covered | Global |