The COVID-19 pandemic is expected to have a significant effect on the market. To keep pace with increased testing norms to detect suspected cases of COVID-19, with the emergence of the SARS-CoV-2 virus, already increased lab testing demand grew even more across the world. For instance, the article published in Nature Journal in October 2021 mentioned that a rapid fluorescence in situ hybridization protocol capable of detecting SARS-CoV-2 specifically and quantitatively in approximately 20 minutes in virus cultures combined nasal and throat swabs with added virus and likely patient samples with added virus and likely patient samples without previous purifications. Such proven efficacy of in situ hybridizations in the diagnosis of COVID-19 infection significantly impacted the market growth during the pandemic period. Also, the demand for in situ hybridization is expected to remain intact due to the emergence of various mutant strains of the SARS-CoV-2 virus during the post-pandemic period, thereby driving the growth of the market during the forecast period.
The increasing incidence of cancer, infectious diseases, and genetic disorders and the growing awareness of cancer therapeutics are the major drivers for the growth of the in-situ hybridization market. For instance, the Global Flu data published in February 2022 mentioned that an estimated 1 billion people worldwide are infected by seasonal influenza yearly. The report also mentioned that out of those 1 billion, about 3 to 5 million people have a severe case of flu every year. Likewise, the report published by the World Health Organization in November 2021 reported that more than 1.0 million sexually transmitted infections are acquired globally, and most of them are asymptomatic. It also reported that every year there are an estimated 374.0 million new infections, with 1 out of 4 sexually transmitted infections: gonorrhea, chlamydia, trichomoniasis, and syphilis.
Additionally, as per the WHO factsheet published in February 2022, in 2021, there were an estimated 1.9 million new cancer cases diagnosed in the United States. The report also mentioned that each year, approximately 400,000 children develop cancer globally. Cervical cancer is the most common in 23 countries, such as Eswatini, India, South Africa, Latin America, and others. The most common cancers are breast, lung, colon, rectum, and prostate. Such rising cases of infectious diseases and cancer across the world are expected to drive the demand for advanced and effective diagnosis, thereby contributing to the growth of the market over the forecast period.
Additionally, the rising market players' initiatives, such as partnerships, acquisitions, and mergers to develop in situ hybridization techniques, are also expected to propel the market's growth. For instance, in March 2021, Bio SB announced that through its partnership agreement with ZytoVision, it would distribute a line of CE-approved chromogenic in situ hybridization and fluorescent in situ hybridization probes targeted for use in lung carcinomas.
However, the lack of skilled professionals to perform advanced procedures is expected to restrain the market's growth over the forecast period.
In Situ Hybridization Market Trends
The Fluorescence In Situ Hybridization (FISH) is Expected to Witness a Healthy Growth in the Market Over the Forecast Period
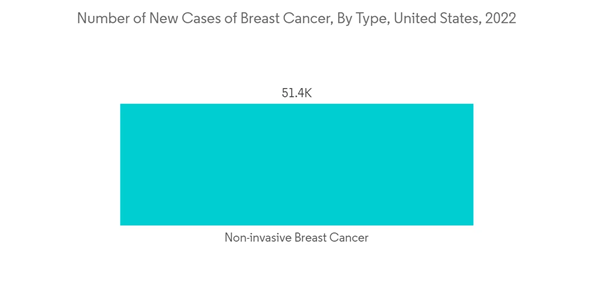
Fluorescence In Situ Hybridization (FISH) is a laboratory technique used to detect and locate a specific DNA sequence on a chromosome from an individual that is affixed to a glass slide and then exposed to a probe, a mall piece of purified DNA tagged with a fluorescent dye. It is utilized to diagnose genetic diseases, cancer, gene mapping, and identification of chromosomal abnormalities. Factors for the growth of the FISH segment are growing genetic disorders, chronic diseases, and increasing demand for efficient diagnostic procedures.For instance, the UN data published in March 2022 stated that the estimated incidence of Down syndrome is between 1 in 1,000 and 1 in 1,100 live births annually. The report also mentioned each year, approximately 3,000 to 5,000 children are born with this chromosome disorder. Also, as per the National Breast Cancer Foundation Inc., data published in June 2022 mentioned that in 2022, an estimated 287,500 new cases of invasive breast cancer is diagnosed in women in the United States and as well as 51,400 new cases of non-invasive breast cancer. The report also mentioned that an estimated 2,170 men are diagnosed with breast cancer in 2022 in the United States. Such rising prevalence of chromosomal disorders and chronic diseases is expected to drive the demand for early and effective diagnosis, thereby contributing to the growth of the studied segment.
Also, the rising product launches by various market players meet the growing demand for FISH techniques, thereby contributing to the market's growth. For instance, in May 2021, Vizgen launched its freely available data set as a part of its newly-launched Vizgen release program. The Vizgen multiplexed error robust fluorescence in situ hybridization mouse brain receptor map is an open-source spatial genomics dataset containing the exact position of transcripts from 483 genes.
Thus, the above-mentioned factors, such as the growing prevalence of various chronic disorders, rising demand for advanced techniques such as FISH, and increasing product launches, are expected to contribute to the growth of the studied segment.
North America is Expected to Hold Significant Share in Situ Hybridization Market Over the Forecast Period
North America is expected to account for the major share in the in situ hybridization market, owing to the higher incidence of cancer in the region and the prevalence of infectious diseases and genetic disorders in the region. Additionally, the rising research and developmental activities, presence of key market players, and frequent product launches are expected to contribute to the market's growth.For instance, according to the American Cancer Society Inc., data published in 2022, in 2021, there were an estimated 1.9 million new cancer cases diagnosed in the United States. In addition, according to the Public Health Agency of Canada data published in October 2021, approximately 2 in 5 Canadians were diagnosed with Cancer in their lifetime. In 2021, an estimated 229,000 Canadians were diagnosed with Cancer. The report also mentioned that lung, breast, colorectal, and prostate cancers were expected to remain the most commonly diagnosed cancers, accounting for nearly 46% of all cancer diagnoses in 2021. Such a high prevalence of cancers in North American countries is expected to contribute to the market's growth. High cancer incidence rates are expected to positively affect market demand for in situ hybridization products during the forecast period.
Additionally, the rising product launches by various key market players in this region are expected to contribute to the market's growth. For instance, in December 2022, OmicsVeu launched an extensive line of spatial biology products along with mRNA and DNA FISH probes and kits. A wide range of over 220 plus fluorophore-tagged miRNA probes enables the generation of multiplex miRNA profiling. Thus, given the above factors, North America is expected to dominate the in situ hybridization market over the forecast period owing to the rising chronic disorders and rising product launches by various key market players.
In Situ Hybridization Market Competitor Analysis
The In Situ Hybridization Market is fragmented and competitive. The major players operating globally in the In Situ Hybridization Market are Abnova Corporation, Agilent Technologies Inc., F. Hoffmann-La Roche Ltd, Biocare Medical LLC., Genemed Biotechnologies Inc., Thermo Fisher Scientific Inc., Biogenex Laboratories, Zytomed Systems GmbH, Creative Bioarray, and Bio SB Inc.Additional benefits of purchasing the report:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Abnova Corporation
- Agilent Technologies Inc.
- F. Hoffmann-La Roche Ltd
- Biocare Medical LLC
- Genemed Biotechnologies Inc.
- Thermo Fisher Scientific Inc.
- Biogenex Laboratories
- Zytomed System GmbH
- Bio-Techne Corporation (Advanced Cell Diagnostics Inc.)
- Bio SB Inc.
- Bio-Techne
- Qiagen NV
- Perkin Elmer
- Abbott Laboratories
- Merck KgaA