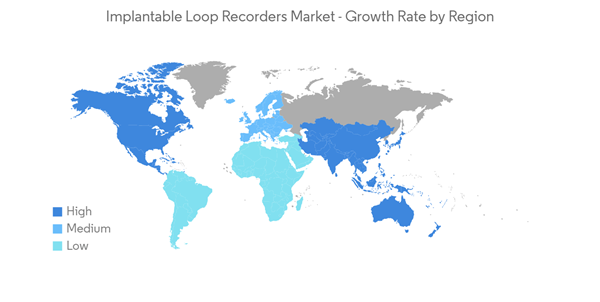
The Implantable Loop Recorders Market is projected to register a CAGR of more than 9% during the forecast period.
The COVID-19 pandemic impacted the production of cardiovascular devices. Furthermore, the delay of elective procedures due to COVID-19-related measures adversely affected the cardiac assist devices market, especially in 2020. While some cardiovascular procedures are considered urgent, others were postponed and delayed at the beginning of the global pandemic. Hence, lifestyle changes and medication were temporarily prescribed instead of cardiovascular procedures for some patients. Another factor negatively impacting the market was the reduction in the diagnosis of cardiovascular diseases due to healthcare resources being reserved for COVID-19 patients. Many medical device companies experienced losses in their cardiovascular device businesses due to the pandemic. For example, Medtronic announced a 33% reduction in its cardiac and vascular business sectors in its 2020 quarterly financial results. The continuous strain of the pandemic negatively impacted the growth of the studied market by decreasing product demand. In addition, the market growth is stabilizing in the current scenario after COVID-19 as the worldwide restrictions have eased down and the disease screening services have been resumed. Moreover, increasing initiatives by most organizers to provide solutions to the surging cardiovascular diseases are the prime boosting factors for market growth during the forecast period.
Certain factors propelling the market growth are the growing prevalence of cardiac disorders, recent technological advancements in cardiac monitoring devices, and increasing demand for remote patient monitoring.
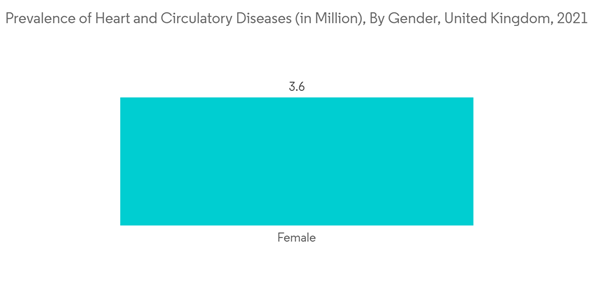
The growing prevalence of heart disorders is significantly augmenting the demand for implantable loop recorders for the treatment of cardiovascular disorders. For instance, according to the British Heart Foundation England Factsheet, published in January 2022, about 6.4 million people were living with cardiovascular diseases in England in 2022. Also, according to AHA, in June 2021, the prevalence rate of heart failure in Europe was between 1% and 2%. Thus, the increasing burden of cardiac disorders among the population is expected to increase the demand for implantable loop recorders to monitor patients’ heart rhythms, thereby fuelling market growth.
Furthermore, recent technological advancements are contributing to market growth. For instance, in February 2021, RhythMedix, a remote cardiac monitoring organization, launched its next-generation RhythmStar wearable device with built-in 4G cellular connectivity. The technology enables rapid ECG analysis and reporting of cardiac abnormalities. Also, in January 2021, Boston Scientific acquired Preventice Solutions for USD 925 million. With this acquisition, Boston expanded its business segment of core cardiac rhythm management and electrophysiology.
Moreover, the increasing adoption of remote patient monitoring (RPM) is contributing to the market growth owing to the introduction of new reimbursement policies. For instance, in January 2021, the Centers for Medicare & Medicaid Services amended the 2021 Physician Fee Schedule, making it possible for increased reimbursement for remote patient monitoring. This indicates wider adoption of remote patient monitoring for cardiac monitoring, thereby increasing the demand for implantable loop recorders. Also, as per the AHA, recent clinical guidelines strongly recommend the use of remote patient monitoring for atrial fibrillation detection in both stroke and non-stroke patients, thereby further increasing the demand for implantable loop recorders, hence boosting the market growth.
However, the high cost of the device and stringent regulations are some of the factors impeding the market growth.
Furthermore, the rising company activities in developing advanced technological devices for monitoring cardiac rhythms and increasing product launches are also contributing to the growth of the market. For instance, in November 2022, Boston Scientific launched the LUX-Dx Insertable Cardiac Monitor (ICM) System, a long-term diagnostic device inserted under the skin of patients to detect arrhythmias associated with conditions such as atrial fibrillation (AF), cryptogenic stroke, and syncope, in Europe.
Therefore, due to the increasing burden of cardiac arrhythmia disorders among the population, as well as increasing company activities and product launches, the studied market is expected to grow over the forecast period.
In addition, the rising geriatric population who are more prone to develop cardiovascular diseases and requires continuous cardiac activity monitoring, increasing the demand for implantable loop recorder devices, thereby boosting the market growth.
The rising burden of cardiovascular diseases such as cardiac arrhythmias, hypertension, and others among the population is expected to increase the demand for implantable loop recorders for measuring and detecting irregular heart rhythms. For instance, according to American Heart Association, in June 2021, the prevalence rate of heart failure in Canada was between 1.5% to 1.9%. Thus, the increased cases of heart failure among the population increase the risk of atrial fibrillation, which requires regular monitoring of heart rhythm and is expected to augment the demand for implantable loop recorders over the forecast period.
In the United States, cardiovascular diseases and hypertension cases account for a major share of the people that require surgery. For instance, the data updated by Cedars-Sinai in January 2022 showed that coronary artery bypass graft surgery (CABG), also known as coronary artery bypass, or bypass surgery, is the most common type of heart surgery, and more than 300,000 people have successful bypass surgery in the United States each year. In addition, according to the CDC, in the United States, the prevalence of atrial fibrillation is estimated to rise to 12.1 million by 2030. This shows a rising burden of cardiovascular diseases in the country, which is expected to demand implantable loop recorders in the United States, driving market growth.
Moreover, the technological advancements in remote patient monitoring devices, as well as the presence of key players in the region, are developing and launching products for helping patients with cardiovascular diseases, which is expected to augment the market growth over the forecast period. For instance, in July 2021, Abbott launched Jot Dx, an insertable cardiac monitor (ICM), in the United States. This technology allows for remote detection and improved diagnostic accuracy of cardiac arrhythmia in patients. In addition, SyncUP is a personalized service that offers one-on-one training and instruction to help patients become connected and stay connected to their ICM. Also, in July 2021, the US FDA approved Medtronic’s two AccuRhythm artificial intelligence (AI) algorithms for use with the LINQ II insertable cardiac monitor (ICM). In addition, AccuRhythm AI improves the information physicians receive to accurately diagnose and treat irregular heart rhythms by applying AI to heart rhythm event data gathered by LINQ II.
Therefore, due to the aforementioned factors, the implantable loop recorder market is expected to grow in the region over the forecast period.
This product will be delivered within 2 business days.
The COVID-19 pandemic impacted the production of cardiovascular devices. Furthermore, the delay of elective procedures due to COVID-19-related measures adversely affected the cardiac assist devices market, especially in 2020. While some cardiovascular procedures are considered urgent, others were postponed and delayed at the beginning of the global pandemic. Hence, lifestyle changes and medication were temporarily prescribed instead of cardiovascular procedures for some patients. Another factor negatively impacting the market was the reduction in the diagnosis of cardiovascular diseases due to healthcare resources being reserved for COVID-19 patients. Many medical device companies experienced losses in their cardiovascular device businesses due to the pandemic. For example, Medtronic announced a 33% reduction in its cardiac and vascular business sectors in its 2020 quarterly financial results. The continuous strain of the pandemic negatively impacted the growth of the studied market by decreasing product demand. In addition, the market growth is stabilizing in the current scenario after COVID-19 as the worldwide restrictions have eased down and the disease screening services have been resumed. Moreover, increasing initiatives by most organizers to provide solutions to the surging cardiovascular diseases are the prime boosting factors for market growth during the forecast period.
Certain factors propelling the market growth are the growing prevalence of cardiac disorders, recent technological advancements in cardiac monitoring devices, and increasing demand for remote patient monitoring.
The growing prevalence of heart disorders is significantly augmenting the demand for implantable loop recorders for the treatment of cardiovascular disorders. For instance, according to the British Heart Foundation England Factsheet, published in January 2022, about 6.4 million people were living with cardiovascular diseases in England in 2022. Also, according to AHA, in June 2021, the prevalence rate of heart failure in Europe was between 1% and 2%. Thus, the increasing burden of cardiac disorders among the population is expected to increase the demand for implantable loop recorders to monitor patients’ heart rhythms, thereby fuelling market growth.
Furthermore, recent technological advancements are contributing to market growth. For instance, in February 2021, RhythMedix, a remote cardiac monitoring organization, launched its next-generation RhythmStar wearable device with built-in 4G cellular connectivity. The technology enables rapid ECG analysis and reporting of cardiac abnormalities. Also, in January 2021, Boston Scientific acquired Preventice Solutions for USD 925 million. With this acquisition, Boston expanded its business segment of core cardiac rhythm management and electrophysiology.
Moreover, the increasing adoption of remote patient monitoring (RPM) is contributing to the market growth owing to the introduction of new reimbursement policies. For instance, in January 2021, the Centers for Medicare & Medicaid Services amended the 2021 Physician Fee Schedule, making it possible for increased reimbursement for remote patient monitoring. This indicates wider adoption of remote patient monitoring for cardiac monitoring, thereby increasing the demand for implantable loop recorders. Also, as per the AHA, recent clinical guidelines strongly recommend the use of remote patient monitoring for atrial fibrillation detection in both stroke and non-stroke patients, thereby further increasing the demand for implantable loop recorders, hence boosting the market growth.
However, the high cost of the device and stringent regulations are some of the factors impeding the market growth.
Key Market Trends
Cardiac Arrhythmia Segment Expected to Witness Significant Growth Over the Forecast Period
The cardiac arrhythmia segment is expected to witness significant growth over the forecast period owing to the factors such as the increasing prevalence of cardiac arrhythmia as well as the increasing adoption of remote patient monitoring devices. For instance, as per a September 2021 update by the National Health Service, in the United Kingdom, more than 2 million people experienced arrhythmias or heart rhythm problems. With the increasing number of heart problems, the demand for electrophysiology devices is increasing, which is likely to augment the growth of the market. Also, as per the 2021 updates of the WHO, ischemic heart disease was responsible for 16% of the world’s total deaths. Thus, increasing myocardial ischemia created an unstable electrical substrate capable of initiating and sustaining arrhythmias, which is likely to increase the monitoring of cardiac activity, which in turn is anticipated to fuel market growth.Furthermore, the rising company activities in developing advanced technological devices for monitoring cardiac rhythms and increasing product launches are also contributing to the growth of the market. For instance, in November 2022, Boston Scientific launched the LUX-Dx Insertable Cardiac Monitor (ICM) System, a long-term diagnostic device inserted under the skin of patients to detect arrhythmias associated with conditions such as atrial fibrillation (AF), cryptogenic stroke, and syncope, in Europe.
Therefore, due to the increasing burden of cardiac arrhythmia disorders among the population, as well as increasing company activities and product launches, the studied market is expected to grow over the forecast period.
North America Dominates the Market and Expected to do Same Over the Forecast Period
North America is expected to account for a major share of the implantable loop recorders market over the forecast period, owing to the increasing prevalence of cardiovascular diseases, rising demand for remote patient monitoring devices, the presence of well-established healthcare infrastructure, and the growing healthcare expenditure.In addition, the rising geriatric population who are more prone to develop cardiovascular diseases and requires continuous cardiac activity monitoring, increasing the demand for implantable loop recorder devices, thereby boosting the market growth.
The rising burden of cardiovascular diseases such as cardiac arrhythmias, hypertension, and others among the population is expected to increase the demand for implantable loop recorders for measuring and detecting irregular heart rhythms. For instance, according to American Heart Association, in June 2021, the prevalence rate of heart failure in Canada was between 1.5% to 1.9%. Thus, the increased cases of heart failure among the population increase the risk of atrial fibrillation, which requires regular monitoring of heart rhythm and is expected to augment the demand for implantable loop recorders over the forecast period.
In the United States, cardiovascular diseases and hypertension cases account for a major share of the people that require surgery. For instance, the data updated by Cedars-Sinai in January 2022 showed that coronary artery bypass graft surgery (CABG), also known as coronary artery bypass, or bypass surgery, is the most common type of heart surgery, and more than 300,000 people have successful bypass surgery in the United States each year. In addition, according to the CDC, in the United States, the prevalence of atrial fibrillation is estimated to rise to 12.1 million by 2030. This shows a rising burden of cardiovascular diseases in the country, which is expected to demand implantable loop recorders in the United States, driving market growth.
Moreover, the technological advancements in remote patient monitoring devices, as well as the presence of key players in the region, are developing and launching products for helping patients with cardiovascular diseases, which is expected to augment the market growth over the forecast period. For instance, in July 2021, Abbott launched Jot Dx, an insertable cardiac monitor (ICM), in the United States. This technology allows for remote detection and improved diagnostic accuracy of cardiac arrhythmia in patients. In addition, SyncUP is a personalized service that offers one-on-one training and instruction to help patients become connected and stay connected to their ICM. Also, in July 2021, the US FDA approved Medtronic’s two AccuRhythm artificial intelligence (AI) algorithms for use with the LINQ II insertable cardiac monitor (ICM). In addition, AccuRhythm AI improves the information physicians receive to accurately diagnose and treat irregular heart rhythms by applying AI to heart rhythm event data gathered by LINQ II.
Therefore, due to the aforementioned factors, the implantable loop recorder market is expected to grow in the region over the forecast period.
Competitive Landscape
The implantable loop recorders market is highly consolidated, and a few companies are currently dominating the market. Some of the companies include Abbott, Boston Scientific Corporation, Medtronic, BIOTRONIK, and Vectorious. The competition in the market is majorly based on technological advances in implantable loop recorders. Additionally, the key players have been involved in various strategic alliances, such as acquisitions and collaborations, along with the launch of advanced products to secure their position in the global market.Additional benefits of purchasing the report:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
1 INTRODUCTION
4 MARKET DYNAMICS
5 MARKET SEGMENTATION (Market Size by Value - USD million)
6 COMPETITIVE LANDSCAPE
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Abbott
- Medtronic
- BIOTRONIK
- Boston Scientific Corporation
- Vectorious
- Angel Medical Systems Inc.