With the emergence of COVID-19, the focus has shifted more toward patient care and comfort. The study published in July 2021 by PLoS One stated that the elective procedure incidence decreased rapidly at the onset of the first COVID-19 wave in March 2020 but recovered in May and June, after which the surgery incidence was 22% higher than in the reference years. With only urgent surgeries being allowed and most of the cardiac procedures canceled or postponed due to the diversion of resources toward COVID-19-affected patients, the market for fetal heart rate monitoring market was tremendously affected. However, with the rise in demand compelling the situation, regulatory authorities like the United States Food and Drug Administration provided some relaxation in the regulations for the launch of medical devices to provide uninterrupted support and care to the mother and fetus during the challenging situation of the pandemic. Thus, COVID-19 is expected to impact the fetal heart rate monitoring device market positively over the post-pandemic era.
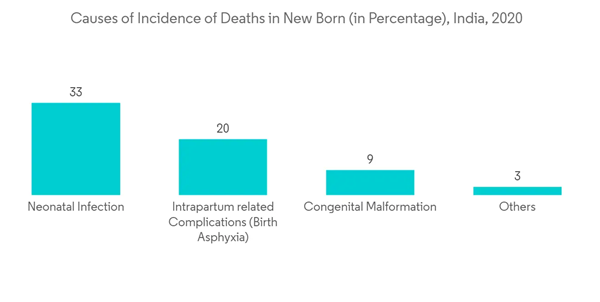
The growth of the market studied is largely attributed to technological advancement in fetal monitoring devices, increasing birth rate and preterm births, and rising government and non-government initiatives to provide better maternal and fetal care. The technological advancement in monitoring devices is likely to drive the market's growth, as it primarily focuses on patient care and comfort. The wireless and in-home monitoring devices introduced in the market enable data sharing with healthcare workers. Preterm birth indicates that a baby is born before the completion of 37 weeks of pregnancy, and lifestyle conditions, such as diabetes and hypertension, are the common causes for this. For instance, as per the article published in July 2020 by the International Journal of Gynecology & Obstetrics, preterm birth is a live birth that occurs before 37 completed weeks of pregnancy, and approximately 15 million babies are born preterm annually worldwide, indicating a global preterm birth rate of about 11%. As per the same source, with 1 million children dying due to preterm birth before the age of 5 years, preterm birth is the leading cause of death among children, accounting for 18% of all deaths among children aged under five years and as many as 35% of all deaths among newborns. Therefore, vigorous heart rate monitoring helps determine the fetus's safety. Hence, owing to the rising number of preterm births and rising usage of these devices in fetal health monitoring, the market is likely to grow with a notable growth rate over the upcoming period.
Various government and non-government agencies also participate to ensure better facilities. For instance, WHO collaborated with ministries of health to strengthen and invest in care, particularly around the time of birth and the first week of life. This is likely to encourage the participation of the organization and boost the market's growth. Due to the aforementioned facts, the fetal heart rate monitoring devices market is expected to witness significant growth over the forecast period.
Various advancements, product approvals, launches, partnerships, and acquisitions by the key market players in cardiac monitoring devices are likely to boost the growth of the market studied over the forecast period. In June 2021, Nuvo Group-a private company commercializing INVU by Nuvo, an FDA-cleared, patent-protected, prescription-initiated remote pregnancy monitoring platform, announced that it had received clearance from the US Food and Drug Administration (FDA) to add a new uterine activity module that provides the capability for remote monitoring of uterine activity (UA). Such product launches are anticipated to result in the growth of the market.
However, stringent regulatory norms for medical devices and the high cost associated with fetal heart rate monitoring devices may act as restraining factors for the market studied.
Fetal Heart Rate Monitoring Devices Market Trends
Internal Heart Rate Monitoring Devices Segment is Expected to Witness Growth over the Forecast Period
Internal fetal heart rate monitoring uses an electronic transducer connected directly to the fetal skin. A wire electrode is attached to the fetal scalp or another body part through the cervical opening and is connected to the monitor. This type of electrode is sometimes called a spiral or scalp electrode.Many technologically advanced medical device organizations are planning to launch such devices or are on the verge of launching them. These wireless or cableless monitoring devices have made prenatal care a better experience, especially in developing countries with a shortage of obstetricians and gynecologists. Additionally, the use of a wireless fetal monitor that can transmit fetal heart rate data via Bluetooth was piloted among healthy patients undergoing labor induction at term and allowed home monitoring for 24 hours. The use of wireless fetal monitoring technology in the home may be found feasible and acceptable to pregnant women. The instrument, like HeraBEAT (smart fetal doppler with data sharing) by H-cube (Her Healthcare at home), enables the expecting mother to hear and monitor the fetus's heart rate and share it with health practitioners. Thus, with the support of technologically advanced solutions for providing better prenatal care, the demand for wireless fetal heart rate monitoring devices is expected to grow in the future.
For instance, according to the article titled 'Neuroblastoma Treatment' published in February 2022, the prevalence of neuroblastoma is about 1 case per 7,000 live births. The incidence is 10.54 cases per 1 million annually in children younger than 15 years. As per the same source, about 37% of patients are diagnosed as infants, and 90% are younger than 5 years at diagnosis, with a median age at diagnosis of 19 months. Such a high burden of disease is propelling the growth of the market.
Thus, due to the aforementioned developments, the market is expected to witness significant growth over the forecast period.
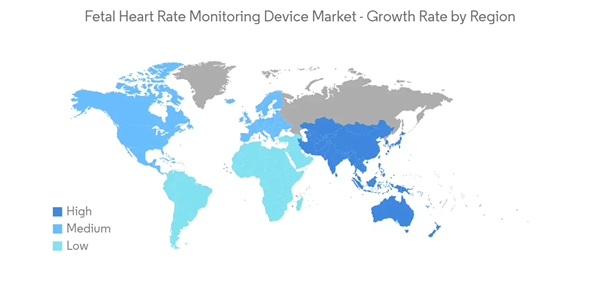
North America is Expected to Witness Growth over the Forecast Period
North America, along with the United States and Canada, is found to have one of the most well-structured, developed, and advanced healthcare systems in the world. With high R&D expenditure for fetal and neonatal care devices, governments of the North American region are also promoting small setups for healthcare research. Therefore, many companies are encouraged to operate in the region, allowing North America to dominate the market across the world.According to the November 2021 update by the CDC, in 2020, preterm birth affected 1 in every 10 infants born in the United States. It also reported that the rate of preterm birth among African-American women (14.4%) was about 50% higher than the rate of preterm birth among white or Hispanic women (9.1% and 9.8%, respectively). Since infants who are born preterm are at an increased risk of heart complications, fetal heart rate monitoring device helps in the monitoring of the conditions, thereby driving the growth of the market.
Moreover, the increasing funding from government organizations for research and development activities in the United States is a major contributing factor to the market's growth in this region. Furthermore, continuous advancements in technology, growing awareness in patients about cardiac monitoring, well-established healthcare infrastructure, and favorable reimbursement policies are expected to boost the cardiac monitoring market in the North American region. For instance, in July 2021 Abbott launched Jot Dx, the insertable cardiac monitor (ICM), in the United States. This technology allows for remote detection and improved diagnostic accuracy of cardiac arrhythmias in patients.
Therefore, owing to the aforementioned factors, the growth of the market studied is anticipated in the North American region.
Fetal Heart Rate Monitoring Devices Industry Overview
The fetal heart rate monitoring devices market is moderately competitive with several global and regional players. In terms of market share, the major players currently dominate the market. With the rising patient awareness levels and high preterm birth rates, many regional players are expected to be part of the fetal heart rate monitoring devices market over the forecast period. The market's major players are GE Healthcare, Fujifilm Holdings Corporation, MedGyn Products Inc., Siemens Healthineers, Cooper Companies Inc, Edan instruments, and Trismed Co. Ltd.Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Koninklijke Philips NV
- GE Healthcare
- Siemens Healthineers
- Cooper Companies Inc.
- MedGyn Products Inc.
- Edan Instruments
- Bionet America
- Trismed Co. Ltd
- Mindray Medical International Limited
- Fujifilm Holdings Corporation
- Natus Medical Inc.