Many companies developing COVID-19 vaccines were using viral vectors to ensure effective vaccine delivery. However, the production of viral vectors was a cumbersome and challenging process, and any improvement to that process could result in more vaccines being made available faster. The viral vector-producing cells need to be transfected with multiple plasmids carrying various viral genes and the vaccine. For instance, according to an article published by PubMed in July 2022, a transfection procedure was used for the purification of the SARS-CoV-2 spike (S) protein ectodomain. As the research and development of COVID-19 vaccines gained pace during the initial pandemic, transfection reagents and equipment were increasingly used in the production and research of vaccines. Thus, the pandemic had a significant impact on the market growth, however as the pandemic has subsided currently, the studied market is expected to have stable growth during the forecast period of the study.
The major factors driving the market growth include technological advancements in transfection, increased R&D expenditure and activities by pharmaceutical and biotech companies, and a rise in demand for synthetic genes.
The introduction of transfection technologies has laid the foundation for the genetic dissection of several causative agents of several diseases. The technological advancements in transfection procedures is a major factor driving the market growth. For instance, according to an article published by PubMed Central in October 2022, it has been observed that recently, strategies to improve human mesenchymal stem cells (hMSCs) transfection have been developed by innovating nanocarriers, nucleic acid cargos, and by priming hMSCs chemically and physically for more efficient transfection. Thus, these types of technological advancements in transfection are expected to boost the market growth.
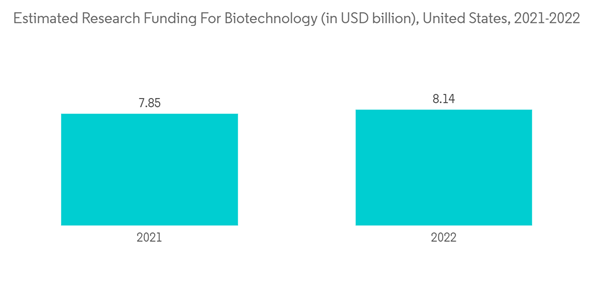
Moreover, the increasing R&D expenditure and research activities by pharmaceutical and biotech companies are also expected to boost market growth. For instance, according to the 2021 annual report of Pfizer Inc., the company invested USD 13,829 million in FY 2021 which increased heavily as compared to USD 9,343 million in the previous year. Similarly, as per a press release published in April 2022 by Biocon Biologics Limited, a subsidiary of biopharmaceutical market player Biocon, the company expects its R&D expenses to increase heavily between 10-15% in the next financial year (FY 2023), as the company is advancing its pipeline to drive future growth. Thus, the rising R&D expenditure of such pharmaceutical and biotechnology companies is expected to boost market growth.
Additionally, the rising developments by various market players are also expected to boost market growth. For instance, in May 2021, Lonza Group AG launched the next generation of its popular Nucleofecto Platform. Nucleofector Technology is an effective non-viral cell transfection method, which can be used even for hard-to-transfect cells, such as primary cells and pluripotent stem cells.
Hence, the aforementioned factors such as the rising technological advancements in transfection, rising R&D expenditure of such pharmaceutical and biotechnology companies, and the increasing developments by various market players are expected to boost the market growth. However, the high cost of transfection reagents and selective effectiveness and certain limitations of transfection reagents can restrain the market growth over the forecast period.
Transfection Reagents and Equipment Market Trends
Pharmaceutical & Biotechnology Companies Segment is Expected to Hold a Significant Share in the Market Over the Forecast Period
The use of biopharmaceutical products has remarkably increased over the years for the treatment of multiple pathological conditions, such as genetic diseases, cancer, etc. With the growing burden of diseases, the need for early diagnosis and treatments also increases. The pharmaceutical and biotechnology companies have increased their investment and research for such therapies, and as transfection reagents and equipment are highly required in such research processes, the increasing demand for transfection reagents and equipment is expected to enhance the segment's growth.The increasing research and development expenses by various pharmaceutical and biotechnology companies are a major factor driving the segment growth. For instance, as per the annual report 2021 of Novartis AG, one of the global market players in the pharmaceutical industry, the company invested USD 14,886 million in R&D in FY 2021 which increased from USD 14,197 million in the previous year. Similarly, according to the annual report of Eli Lilly and Company in 2021, the research and development expenses of the company increased to USD 7,025.9 million in 2021 which is very high compared to USD 6,085.7 million in R&D expenses in the previous year.
Additionally, the research and development collaborations for the development of new genetic products are also expected to enhance the segment growth. For instance, in December 2022, Eli Lilly and Company and ProQR Therapeutics N.V. announced the expansion of their licensing and collaboration agreement focused on the discovery, development and commercialization of new genetic medicines.
Thus, the aforementioned factors such as the increasing research and development expenses, and the increasing collaborations and agreements for developing newer genetic products are expected to boost the segment growth.
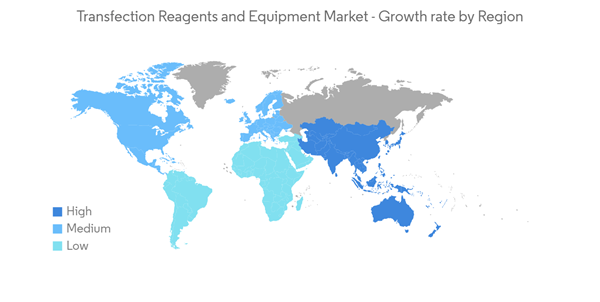
North America is Expected to Hold a Significant Share in the Market Over the Forecast Period
Factors such as increasing investments in research and development in genomics, increasing healthcare expenditure, rising developments by major market players, and the availability of advanced technologies are expected to fuel market growth in North America.For instance, in September 2021, the National Institutes of Health awarded USD 16 million to the University of Washington School of Medicine's scientists and their collaborators to fund two projects within the consortium. The first project aims at studying the more than one million known regulatory elements in the genome and their variants to determine how they affect gene expression. The second project aims at studying the effect of about 200,000 single nucleotide variants, which contain a change of a single DNA molecule in a gene sequence in about 32 genes that are witnessed in specific diseases, including cancer.
Similarly, in October 2022, the Government of Canada invested over USD 5.6 million to help early-stage genomics companies bring new products to market Thus, the rising investments in genomic research are expected to boost the market growth in the region.
Moreover, the rising developments by key market players are also expected to enhance market growth. For instance, in August 2021, Mirus Bio, a United States-based company, expanded its TransIT VirusGen platform, which is compliant with good manufacturing practices (GMP) viral vector manufacturing, to support cell and gene therapy development, process activities, and commercial production. The expansion, referred to as TransIT VirusGen GMP transfection reagent, is designed to enhance the delivery of packaging and transfer vector DNA to suspension and adherent HEK 293 cell types to increase the production of recombinant adeno-associated virus and lentivirus vectors.
Thus, the aforementioned factors such as the increasing funding for genomics research, and the increasing developments by key market players are expected to boost the market growth in the region.
Transfection Reagents and Equipment Industry Overview
The transfection reagents and equipment market is moderately competitive and consists of a number of major players. Companies like Bio-Rad Laboratories, Lonza Group, MaxCyte Inc., Merck KGaA, Mirus Bio, Polyplus-transfection SA, Promega Corporation, QIAGEN NV, Thermo Fisher Scientific Inc., among others, hold a substantial share in the transfection reagents and equipment market.Additional Benefits:
- The market estimate (ME) sheet in Excel format
- 3 months of analyst support
This product will be delivered within 2 business days.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Bio-Rad Laboratories Inc.
- Lonza Group
- MaxCyte Inc.
- Merck KGaA
- Mirus Bio LLC
- Polyplus-transfection SA
- Promega Corporation
- Qiagen NV
- Thermo Fisher Scientific Inc.