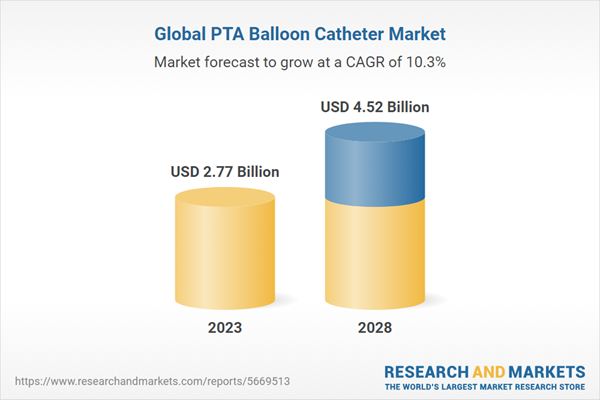
The Global PTA Balloon Catheter Market is estimated to be USD 2.77 Bn in 2023 and is expected to reach USD 4.52 Bn by 2028 growing at a CAGR of 10.3%Open MRI may be the last chance for imaging to support diagnoses of conditions for patients while avoiding the need for general anaesthesia
Market Dynamics
Market dynamics are forces that impact the prices and behaviors of the stakeholders. These forces create pricing signals which result from the changes in the supply and demand curves for a given product or service. Forces of Market Dynamics may be related to macro-economic and micro-economic factors. There are dynamic market forces other than price, demand, and supply. Human emotions can also drive decisions, influence the market, and create price signals.As the market dynamics impact the supply and demand curves, decision-makers aim to determine the best way to use various financial tools to stem various strategies for speeding the growth and reducing the risks.
Market Segmentations
- The Global PTA Balloon Catheter Market is segmented based on Matrerial, Application, End-Users, and Geography.
- By Matrerial, the market is classified into Nylon and Polyurethane.
- By Application, the market is classified into Coronary Artery Disease, and Peripheral Vascular Disease.
- By End-Users, the market is classified into Ambulatory Surgery Centers, Hospitals , and Clinics.
- By Geography, the market is classified into Americas, Europe, Middle-East & Africa, and Asia-Pacific.
Company Profiles
The report provides a detailed analysis of the competitors in the market. It covers the financial performance analysis for the publicly listed companies in the market. The report also offers detailed information on the companies' recent development and competitive scenario. Some of the companies covered in this report are Abbott Laboratories, Andratec GmbH, B. Braun Ag, Biosensors International Group, Ltd., BIOTRONIK SE & Co. KG, Boston Scientific Corp., BrosMed Medical Co., Ltd., Cardinal Health, Inc., etc.Countries Studied
- America (Argentina, Brazil, Canada, Chile, Colombia, Mexico, Peru, United States, Rest of Americas)
- Europe (Austria, Belgium, Denmark, Finland, France, Germany, Italy, Netherlands, Norway, Poland, Russia, Spain, Sweden, Switzerland, United Kingdom, Rest of Europe)
- Middle-East and Africa (Egypt, Israel, Qatar, Saudi Arabia, South Africa, United Arab Emirates, Rest of MEA)
- Asia-Pacific (Australia, Bangladesh, China, India, Indonesia, Japan, Malaysia, Philippines, Singapore, South Korea, Sri Lanka, Thailand, Taiwan, Rest of Asia-Pacific)
Competitive Quadrant
The report includes Competitive Quadrant, a proprietary tool to analyze and evaluate the position of companies based on their Industry Position score and Market Performance score. The tool uses various factors for categorizing the players into four categories. Some of these factors considered for analysis are financial performance over the last 3 years, growth strategies, innovation score, new product launches, investments, growth in market share, etc.Ansoff Analysis
- The report presents a detailed Ansoff matrix analysis for the Global PTA Balloon Catheter Market. Ansoff Matrix, also known as Product/Market Expansion Grid, is a strategic tool used to design strategies for the growth of the company. The matrix can be used to evaluate approaches in four strategies viz. Market Development, Market Penetration, Product Development and Diversification. The matrix is also used for risk analysis to understand the risk involved with each approach.
- The publisher analyses the Global PTA Balloon Catheter Market using the Ansoff Matrix to provide the best approaches a company can take to improve its market position.
- Based on the SWOT analysis conducted on the industry and industry players, the publisher has devised suitable strategies for market growth.
Why buy this report?
- The report offers a comprehensive evaluation of the Global PTA Balloon Catheter Market. The report includes in-depth qualitative analysis, verifiable data from authentic sources, and projections about market size. The projections are calculated using proven research methodologies.
- The report has been compiled through extensive primary and secondary research. The primary research is done through interviews, surveys, and observation of renowned personnel in the industry.
- The report includes an in-depth market analysis using Porter's 5 forces model and the Ansoff Matrix. In addition, the impact of Covid-19 on the market is also featured in the report.
- The report also includes the regulatory scenario in the industry, which will help you make a well-informed decision. The report discusses major regulatory bodies and major rules and regulations imposed on this sector across various geographies.
- The report also contains the competitive analysis using Positioning Quadrants, the Proprietary competitive positioning tool.
Report Highlights:
- A complete analysis of the market, including parent industry
- Important market dynamics and trends
- Market segmentation
- Historical, current, and projected size of the market based on value and volume
- Market shares and strategies of key players
- Recommendations to companies for strengthening their foothold in the market
Table of Contents
1 Report Description
2 Research Methodology
3 Executive Summary
4 Market Dynamics
5 Market Analysis
6 Global PTA Balloon Catheter Market, By Matrerial
7 Global PTA Balloon Catheter Market, By Application
8 Global PTA Balloon Catheter Market, By End-Users
9 Americas’ PTA Balloon Catheter Market
10 Europe’s PTA Balloon Catheter Market
11 Middle East and Africa’s PTA Balloon Catheter Market
12 APAC’s PTA Balloon Catheter Market
13 Competitive Landscape
14 Company Profiles
15 Appendix
Companies Mentioned
- Abbott Laboratories
- Andratec GmbH
- B. Braun Ag
- Biosensors International Group, Ltd.
- BIOTRONIK SE & Co. KG
- Boston Scientific Corp.
- BrosMed Medical Co., Ltd.
- Cardinal Health, Inc.
- Cardionovum GmbH
- Cook Group, Inc.
- Cordis Corp.
- Endovastec (MicroPort Scientific Corp.)
- JOTEC GmbH
- Medtronic, PLC
- Meril Life Sciences Pvt. Ltd.
- Natec Medical, Ltd.
- QT Vascular, Ltd.
- QXMedical, LLC
- Terumo Corp.
- Tokai Medical Products, Inc.