Speak directly to the analyst to clarify any post sales queries you may have.
10% Free customizationThis report comes with 10% free customization, enabling you to add data that meets your specific business needs.
Venture capital funding within the U.S. bioscience sector, which underpins cloning and mutagenesis technologies, achieved $26 billion in 2024, as reported by the Biotechnology Innovation Organization (BIO). Despite this strong financial backing, the market faces considerable obstacles due to the intricate regulatory and ethical frameworks surrounding genetic manipulation, which require strict supervision and widespread public approval to ensure broader adoption.
Market Driver
A major catalyst for the Global Cloning and Mutagenesis Market is the rising use of sophisticated gene editing tools, which greatly improve the accuracy and speed of genetic alterations. Breakthroughs like the CRISPR-Cas system consistently enhance the capacity for precise genomic modifications, facilitating more focused therapeutic and research initiatives. By equipping scientists with robust methods for base editing, gene insertion, and gene knockout, these technological strides expedite both scientific exploration and practical application. Reflecting the substantial financial interest in these foundational technologies, Cure. noted that CRISPR Therapeutics declared an upsized $550 million convertible senior notes offering in March 2026, a move that will further drive innovation and the commercialization of gene editing solutions.At the same time, the escalating need for customized therapeutics and personalized medicine heavily impacts the market, as it demands precise genetic adjustments suited to unique patient characteristics. Methods in cloning and mutagenesis are essential for producing these individualized therapies, playing a role in everything from engineering therapeutic cells to establishing disease models for medication testing.
This strategy tailors healthcare interventions to a person's specific lifestyle, environmental, and genetic elements. The tangible results of this demand are evident; the Personalized Medicine Coalition reported to the FDA in May 2025 that 2024 saw the approval of 18 novel personalized medicines and six cell- and gene-based treatments for specific cancers and genetic disorders. Highlighting the robust nature of the associated biopharma industry, JPMorgan recorded $82.7 billion in announced biopharma licensing values during the first quarter of 2026.
Market Challenge
The elaborate regulatory and ethical frameworks surrounding genetic manipulation pose a major hurdle for the expansion of the Global Cloning and Mutagenesis Market. This complicated setting introduces considerable unpredictability for innovators, resulting in extended product development schedules and higher expenses related to maintaining compliance. Furthermore, differing regulatory interpretations and strict supervisory processes across various regions can obstruct global partnerships and delay market access, ultimately impeding the progress and commercial rollout of novel genetic tools.This obstacle directly affects how quickly new products are approved and introduced to the market. For example, the Alliance for Regenerative Medicine indicated that the United States was on track to approve merely 4 to 5 cell and gene therapies in 2025. Despite continuous scientific breakthroughs in genetic engineering, this low volume of market introductions highlights the extent to which strict and lengthy regulatory procedures restrict the shift from laboratory research to commercial availability. Consequently, such a demanding landscape may discourage essential funding, decelerating the growth of vital applications in the cloning and mutagenesis industry.
Market Trends
The digitization and automation of laboratory operations are profoundly transforming the Global Cloning and Mutagenesis Market by improving data reliability and workflow efficiency. Incorporating cloud networks, sophisticated software, and robotics enables the accurate and consistent performance of intricate genetic procedures, thereby minimizing human mistakes and boosting processing capacity. This shift allows laboratories to manage greater sample sizes and more complex experimental structures, both of which are vital for synthetic biology and pharmaceutical discovery. Thermo Fisher Scientific Inc. highlighted this momentum in its January 29, 2026, earnings call for the fourth quarter of 2025, revealing a 13% revenue growth in its Life Science Solutions division, which supplies laboratory workflow instruments.Another crucial trend is the advancement of seamless, high-throughput cloning techniques, which facilitate the swift and effective assembly of recombinant DNA without the tedious phases of conventional methods. These improvements empower scientists to rapidly produce numerous expression vectors and gene variants for simultaneous testing, hastening the overall trajectory of therapeutic innovation and research. By reducing technical delays, these optimized processes broaden the potential for sophisticated genetic engineering endeavors. Highlighting the need for these scalable biomanufacturing solutions, Sartorius AG reported in its February 3, 2026, preliminary data for 2025 that its Bioprocess Solutions Division experienced a 9.5% rise in sales revenue in constant currencies.
Key Market Players
- Agilent Technologies Inc.
- Eurofins Scientific SE
- Sartorius AG
- Collecta Inc.
- Codex DNA Inc.
- Lonza Group AG
- Charles River Laboratories International Inc.
- TransGene Biotech Co. Ltd.
- Takara Bio, Inc.
- Thermo Fisher Scientific Inc.
Report Scope
In this report, the Global Cloning & Mutagenesis Market has been segmented into the following categories, in addition to the industry trends which have also been detailed below:Cloning & Mutagenesis Market, By Gene Type:
- Standard
- Complex
Cloning & Mutagenesis Market, By Product Type:
- Cloning Kits
- Mutagenesis Kits
Cloning & Mutagenesis Market, By Technique:
- Topo PCR Cloning
- Blunt End Cloning
- Seamless Cloning
- Site-Directed Mutagenesis
- Others
Cloning & Mutagenesis Market, By Application:
- Gene Synthesis
- Gene Expression
- Gene Therapy
- Vaccine Research
- Others
Cloning & Mutagenesis Market, By End User:
- Pharmaceutical & Biotechnology Companies
- Academic & Research Institutions
- Others
Cloning & Mutagenesis Market, By Region:
- North America
- Europe
- Asia Pacific
- South America
- Middle East & Africa
Competitive Landscape
Company Profiles: Detailed analysis of the major companies present in the Global Cloning & Mutagenesis Market.Available Customizations
With the given market data, the publisher offers customizations according to a company's specific needs. The following customization options are available for the report:Company Information
- Detailed analysis and profiling of additional market players (up to five).
This product will be delivered within 1-3 business days.
Table of Contents
Companies Mentioned
- Agilent Technologies Inc.
- Eurofins Scientific SE
- Sartorius AG
- Collecta Inc.
- Codex DNA Inc.
- Lonza Group AG
- Charles River Laboratories International Inc.
- TransGene Biotech Co. Ltd.
- Takara Bio, Inc.
- Thermo Fisher Scientific Inc.
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 180 |
| Published | May 2026 |
| Forecast Period | 2025 - 2031 |
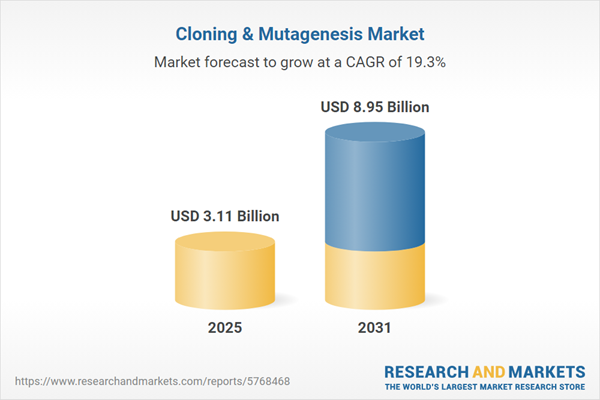
| Estimated Market Value ( USD | $ 3.11 Billion |
| Forecasted Market Value ( USD | $ 8.95 Billion |
| Compound Annual Growth Rate | 19.2% |
| Regions Covered | Global |
| No. of Companies Mentioned | 10 |