Implantable Medical Device: Introduction
Implantable medical devices are surgically placed inside the body to treat or manage various medical conditions. These devices include orthopaedic implants, cardiac implants, neurostimulators, cochlear implants, intraocular lenses, and contraceptive implants, among others. Implantable devices are designed to provide long-term therapeutic benefits, improve patients' quality of life, or replace or support the function of ailing organs. Recent advancements in the field have led to the development of innovative technologies such as bioresorbable stents, miniaturized neurostimulators, and 3D-printed implants, enhancing the safety and efficacy of these treatments.Implantable Medical Device Market Scenario
The global implantable medical device market has witnessed significant growth in recent years, driven by factors such as the increasing prevalence of chronic diseases, the aging population, and advancements in implantable device technologies.North America: The largest market for implantable medical devices, driven by factors such as advanced healthcare infrastructure, high prevalence of chronic diseases, and significant investments in research and development.
Europe: The second-largest market, with increasing demand for implantable devices due to a growing aging population, a supportive regulatory framework, and an expanding healthcare sector.
Asia-Pacific: Expected to witness the highest growth rate due to factors such as a rapidly growing population, increasing prevalence of chronic diseases, and growing investments in healthcare infrastructure and research.
Implantable Medical Device Market Segmentations
Market Breakup by Product
- Orthopaedic Implants
- Dental Implants
- Cardiovascular Implants
- Facial Implants
- Breast Implants
- Others
Market Breakup by Material
- Polymers
- Metals
- Ceramics
- Biologics
Market Breakup by End User
Hospitals
- +500 Beds
- 200 - 499 Beds
- Less than 200 Beds
- Ambulatory Surgical Centers (ASCs)
- Clinics
Market Breakup by Region
North America
- United States of America
- Canada
Europe
- United Kingdom
- Germany
- France
- Italy
- Others
Asia Pacific
- China
- Japan
- India
- ASEAN
- Australia
- Others
Latin America
- Brazil
- Argentina
- Mexico
- Others
Middle East and Africa
- Saudi Arabia
- United Arab Emirates
- Nigeria
- South Africa
- Others
Key Trends in the Implantable Medical Device Market
Some key trends of the market are as follows:- Increasing prevalence of chronic diseases: The rising incidence of conditions such as cardiovascular diseases, neurological disorders, and orthopaedic issues has led to a growing demand for implantable medical devices, driving market growth
- Aging population: The growing aging population worldwide has increased the risk of age-related diseases and disorders, further contributing to the demand for implantable devices
- Advancements in implantable device technologies: The development of new and more effective implantable devices, such as bioresorbable stents, miniaturized neurostimulators, and 3D-printed implants, has improved the efficiency and safety of these treatments, further driving market growth
Implantable Medical Device Market: Competitor Landscape
The key features of the market report include patent analysis, grants analysis, clinical trials analysis, funding and investment analysis, partnerships, and collaborations analysis by the leading key players. The major companies in the implantable medical device market are as follows:- Abbott
- Biotronik, Inc
- Boston Scientific Corporation
- Stryker
- Medtronic
- Danaher Corporation
- Dentsply Sirona
- Institut Straumann AG
- Johnson & Johnson
- Smith and Nephew
- Terumo Corporation
- Zimmer Biomet
Table of Contents
Companies Mentioned
- Abbott
- Biotronik, Inc.
- Boston Scientific Corporation
- Stryker
- Medtronic
- Danaher Corporation
- Dentsply Sirona
- Institut Straumann AG
- Johnson & Johnson
- Smith and Nephew
- Terumo Corporation
- Zimmer Biomet
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 140 |
| Published | May 2023 |
| Forecast Period | 2023 - 2031 |
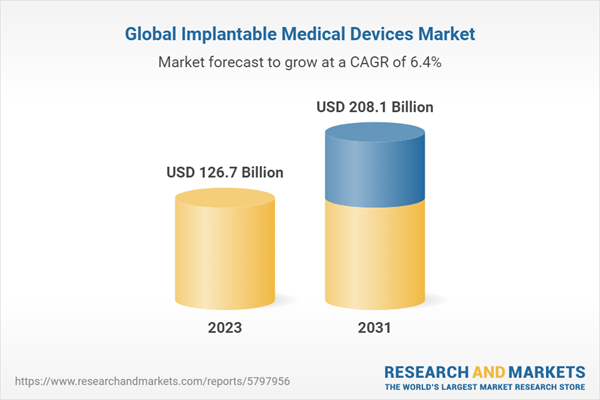
| Estimated Market Value ( USD | $ 126.7 Billion |
| Forecasted Market Value ( USD | $ 208.1 Billion |
| Compound Annual Growth Rate | 6.4% |
| Regions Covered | Global |
| No. of Companies Mentioned | 12 |