Speak directly to the analyst to clarify any post sales queries you may have.
10% Free customizationThis report comes with 10% free customization, enabling you to add data that meets your specific business needs.
However, the high expenses linked to cutting-edge molecular testing platforms and equipment pose a major obstacle to market growth. These significant financial requirements, combined with ongoing gaps in reimbursement coverage for novel genomic assays, place a disproportionate burden on developing nations and smaller laboratories. As a result, the widespread availability and integration of these vital diagnostic technologies are considerably limited.
Market Driver
Rising cancer rates worldwide serve as a primary catalyst for the molecular oncology diagnostics market, driving the need for sophisticated tools to assist in early detection, prognosis, and treatment direction. As global demographics shift toward aging populations and risk factor exposures change, the overall cancer burden continues to grow, fostering a steady requirement for advanced diagnostic options. Research featured in The Lancet and referenced by the Institute for Health Metrics and Evaluation in its September 2025 publication, "Q&A: Global burden of cancer and forecasts to 2025," noted over 18 million new cancer cases globally in 2023. This immense volume of diagnoses emphasizes the vital necessity for molecular testing capable of precisely characterizing cancer types and their unique molecular signatures, a critical component for optimizing patient care and ongoing monitoring.Market growth is additionally fueled by the increasing embrace of personalized and precision oncology, methodologies that depend intrinsically on comprehensive molecular profiling to customize therapies based on a patient's distinct traits. Consequently, there is a surging need for companion diagnostics designed to pinpoint specific biomarkers and facilitate targeted treatments. Major industry participants are directing substantial capital toward this transition to accommodate the rising demand.
In an April 2025 BioSpace article titled 'Roche to invest USD 50 billion in pharmaceuticals and diagnostics in the United States over the next five years,' Roche revealed plans to channel USD 50 billion into the U.S. over five years to establish new manufacturing and research facilities for its advanced diagnostics and medical portfolio. This investment demonstrates how deeply molecular data is becoming embedded in standard treatment protocols. Moreover, Roche's investor data indicates that 31 billion diagnostic tests were supplied globally in 2025, illustrating the widespread utilization of diagnostic tools across modern healthcare and their escalating relevance in the field of oncology.
Market Challenge
High expenses tied to sophisticated molecular testing equipment and platforms act as a formidable hurdle to the expansion of the global molecular oncology diagnostics market. The massive capital investments required to secure such advanced machinery impose a heavy financial burden, especially on laboratories in developing nations and those operating on a smaller scale. Additionally, ongoing deficiencies in reimbursement policies for cutting-edge genomic assays worsen the situation, hindering healthcare providers from fully recouping the substantial expenses incurred while administering these intricate and specialized diagnostic procedures.These monetary constraints directly hinder broad accessibility to vital diagnostic advancements. The American Clinical Laboratory Association warns that unless Congress intervenes, roughly 800 laboratory tests will face rate reductions of up to 15% under the Clinical Laboratory Fee Schedule starting January 31, 2026. Such cuts in reimbursement pose a threat to the financial stability of laboratories, limiting their ability to fund new molecular oncology tests or broaden their current offerings. Ultimately, this dynamic stifles the uptake of modern diagnostic instruments, limits market reach, and decelerates the overarching growth trajectory of the molecular oncology diagnostics sector.
Market Trends
A defining development in molecular oncology diagnostics is the widespread adoption of Next-Generation Sequencing (NGS) for extensive genomic profiling. This shift is marked by the growing incorporation of comprehensive genomic evaluations into standard clinical practice, transitioning away from single-gene assessments to deliver a complete picture of a tumor's genetic makeup. Thorough genomic profiling facilitates the discovery of actionable mutations, directs customized therapeutic interventions, and offers a more precise understanding of how the disease advances. Highlighting this momentum, 1stOncology reported on February 5, 2026, that Illumina's Q4 2025 earnings showed a 20% increase in clinical consumables revenue outside of China, a surge attributed to the wider embrace of sequencing-reliant diagnostics.Another revolutionary trend heavily influencing the industry is the rising utilization of liquid biopsies for the non-invasive management of cancer. By examining circulating tumor DNA and other biomarkers in blood samples, liquid biopsies present a less intrusive substitute for conventional tissue biopsies when diagnosing cancer, tracking treatment efficacy, and monitoring for relapse. This approach is particularly advantageous when securing tissue samples is challenging or when continuous observation is necessary, granting immediate perspective on how a tumor is evolving. Demonstrating this rising clinical acceptance and practicality, Natera experienced a 54.7% year-over-year jump in its oncology test volume during the fourth quarter of 2025, processing roughly 233,300 tests, as noted in a February 26, 2026, Stock Titan report detailing the company's 8-K filing on revenue and cash flow.
Key Market Players
- Sysmex Corporation
- F. Hoffmann-La Roche Ltd.
- Myriad Genetics, Inc.
- HTG Molecular Diagnostics, Inc.
- Danaher Corporation
- QIAGEN N.V.
- Bio-Rad Laboratories, Inc.
- Agilent Technologies, Inc.
- Illumina, Inc.
- Bayer AG
Report Scope
In this report, the Global Molecular Oncology Diagnostics Market has been segmented into the following categories, in addition to the industry trends which have also been detailed below:Molecular Oncology Diagnostics Market, By Product Type:
- Instruments
- Kits and Assays
- Software
Molecular Oncology Diagnostics Market, By Technology:
- Fluorescence In-Situ Hybridization (FISH)
- Next-Generation Sequencing (NGS)
- Polymerase Chain Reaction (PCR)
- Immunohistochemistry (IHC)
- Flow Cytometry
- Other Technologies
Molecular Oncology Diagnostics Market, By Application:
- Research
- Clinical
Molecular Oncology Diagnostics Market, By Cancer Type:
- Solid Tumor
- Blood Cancers
Molecular Oncology Diagnostics Market, By End User:
- Hospitals and Diagnostic Centers
- Reference Laboratories
- Pharmaceutical and Biotechnology Companies
- Academic
- Research Institutes
Molecular Oncology Diagnostics Market, By Region:
- North America
- Europe
- Asia Pacific
- South America
- Middle East & Africa
Competitive Landscape
Company Profiles: Detailed analysis of the major companies present in the Global Molecular Oncology Diagnostics Market.Available Customizations
With the given market data, the publisher offers customizations according to a company's specific needs. The following customization options are available for the report:Company Information
- Detailed analysis and profiling of additional market players (up to five).
This product will be delivered within 1-3 business days.
Table of Contents
Companies Mentioned
- Sysmex Corporation
- F. Hoffmann-La Roche Ltd.
- Myriad Genetics, Inc.
- HTG Molecular Diagnostics, Inc.
- Danaher Corporation
- QIAGEN N.V.
- Bio-Rad Laboratories, Inc.
- Agilent Technologies, Inc.
- Illumina, Inc.
- Bayer AG
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 182 |
| Published | May 2026 |
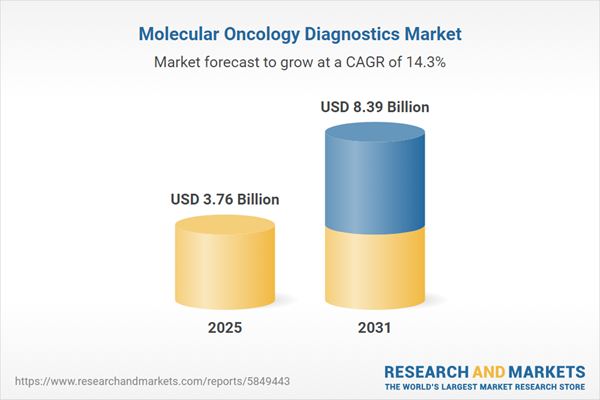
| Forecast Period | 2025 - 2031 |
| Estimated Market Value ( USD | $ 3.76 Billion |
| Forecasted Market Value ( USD | $ 8.39 Billion |
| Compound Annual Growth Rate | 14.3% |
| Regions Covered | Global |
| No. of Companies Mentioned | 10 |