Intravascular Temperature Management: Introduction
Intravascular temperature management is a technique that is used for the management of the body's core temperature while undergoing particular medical procedures or conditions. It is also known as cooling or warming technique in medical terms. The body temperature is managed by inserting the specialized catheters or devices into the blood vessels which helps in regulating the body's temperature by maintaining temperature of the blood stream. The main focus of intravascular temperature management technique is to maintain the patient's body temperature within a certain restricted range in order to improve recovery and patient's outcome.Global Intravascular Temperature Management Market Analysis
Intravascular temperature management has multiple applications, depending upon the requirement of the condition. It can be used in multiple clinical settings, such as during the cardiac surgeries, post cardiac care, neurosurgeries, or sepsis, among others. It is also widely used in hypothermia, a condition where prominent organs of the body as brain and other organs are being protected from the effect of a cardiac arrest or traumatic brain injury by using the controlled cooling technique.The several key trends running in the intravascular temperature management market expansion include the development of more advanced devices, such as catheters, with even better temperature control capabilities and easy to use devices. There is an escalating focus on the use of minimally invasive procedures and techniques in intravascular temperature management.
Minimally invasive procedures are known and preferred for their comparatively minimal incisions and to enable them even smaller equipment are required to be developed, hence the development of even smaller catheters and other devices with more flexibility and functionality is ongoing in the market. Some other key trends in the market include personalized temperature management, and involvement of new technologies along with research and studies owing to the intravascular temperature management market growth.
Global Intravascular Temperature Management Market Segmentations
Intravascular Temperature Management Market Report and Forecast 2025-2034 offers a detailed analysis of the market based on the following segments:Market Breakup by Type
- Intravascular Cooling
- Intravascular Warming
Market Breakup by Medical Condition
- Cardiac Arrest
- Traumatic Brain Injury (TBI)
- Stroke
- Myocardial Infection
- Others
Market Breakup by Application
- Pre-Operative Care
- Operative Care
- Post-Operative Care
- Perioperative Care
- Acute/Critical Care
Market Breakup by Region
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East and Africa
Global Intravascular Temperature Management Market Overview
The market for intravascular temperature management has seen evident growth in the past and is expected to be growing rapidly in coming years as well. The growth of the market is driven by various factors, including the involvement of minimally invasive procedures. The increasing prevalence of the chronic diseases and increasing geriatric population has increased the demand for intravascular temperature management solutions in healthcare industry, propelling the intravascular temperature management market development.Additionally, rapidly advancing technologies and utilization of new advanced devices in the procedure of temperature management has also increased the demand for intravascular temperature management in the market.
Intravascular Temperature Management Market: Competitor Landscape
The key features of the market report include patent analysis, grants analysis, clinical trials analysis, funding and investment analysis, partnerships, and collaborations analysis by the leading key players. The major companies in the market are as follows:- Lonza Group
- Bio-Rad Laboratories, Inc.
- Thermo Fisher Scientific
- Qiagen NV
- Promega Corporation
- SignaGen Laboratories
- Merck KGaA
- Altogen Biosystems
- Polyplus-transfection SA
- Mirus Bio
This product will be delivered within 3-5 business days.
Table of Contents
Companies Mentioned
- Lonza Group
- Bio-Rad Laboratories, Inc.
- Thermo Fisher Scientific
- Qiagen NV
- Promega Corporation
- SignaGen Laboratories
- Merck KGaA
- Altogen Biosystems
- Polyplus-transfection SA
- Mirus Bio
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 350 |
| Published | July 2025 |
| Forecast Period | 2025 - 2034 |
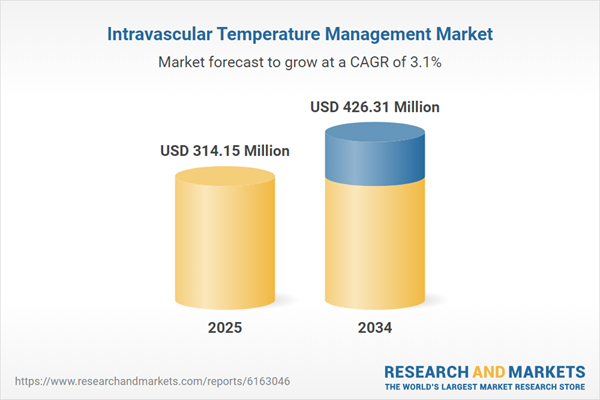
| Estimated Market Value ( USD | $ 314.15 Million |
| Forecasted Market Value ( USD | $ 426.31 Million |
| Compound Annual Growth Rate | 3.1% |
| Regions Covered | Global |
| No. of Companies Mentioned | 10 |