Speak directly to the analyst to clarify any post sales queries you may have.
10% Free customizationThis report comes with 10% free customization, enabling you to add data that meets your specific business needs.
Key Market Drivers
Increasing Preference for Minimally Invasive Procedures
The preference for minimally invasive treatments has become a defining factor in modern healthcare, and this shift is significantly driving growth in the thyroid ablation devices market. Compared to traditional thyroidectomy, ablation techniques such as radiofrequency ablation (RFA), microwave ablation (MWA), and laser ablation offer quicker recovery times, minimal scarring, and reduced hospitalization. These benefits are particularly attractive for treating benign thyroid nodules and low-risk cancers. For example, a multi-center study in Asia demonstrated that RFA led to an average 74% volume reduction of benign nodules at 12 months, with over 85% of patients reporting symptom relief. The growing availability of outpatient procedures further supports this trend, making thyroid ablation a cost-effective and patient-friendly solution that aligns with modern healthcare demands.Key Market Challenges
Regulatory Approvals
Gaining regulatory approval presents a substantial challenge in the thyroid ablation devices market. These devices must undergo rigorous testing and evaluation to meet safety and efficacy standards established by health authorities. The approval process involves complex documentation, clinical trials, and strict quality controls, often resulting in delays in product launch and market availability. Additionally, each device may require a unique regulatory pathway based on its intended use and mechanism. The rapid pace of technological innovation also creates a disconnect with existing regulatory frameworks, slowing down the commercialization of advanced therapies. These factors collectively impede the swift adoption of newer ablation devices, particularly in markets with stringent compliance protocols.Key Market Trends
Personalized Treatment Approaches
A growing emphasis on personalized medicine is transforming thyroid ablation practices. Modern ablation devices now enable treatment customization based on factors such as the patient’s anatomy, nodule characteristics, and clinical needs. Physicians can tailor the ablation technique - choosing between thermal or non-thermal methods - and adjust device parameters for optimized outcomes. This shift toward individualized care enhances treatment efficacy while minimizing adverse effects, promoting better patient satisfaction. Personalized approaches are further supported by advancements in imaging and real-time procedural guidance, which ensure precision during treatment and help clinicians adapt strategies dynamically to suit patient-specific conditions.Key Market Players
- Medtronic Plc
- StarMed Co. Ltd.
- Terumo Corporation
- Johnson & Johnson
- Theraclion SA
- Integra LifeSciences Corp
- Olympus Corp.
- MedWaves Inc
- BVM Medical System
- Boston Scientific Corporation
Report Scope:
In this report, the Global Thyroid Ablation Devices Market has been segmented into the following categories, in addition to the industry trends which have also been detailed below:Thyroid Ablation Devices Market, By Product:
- Non-Thermal Based Devices
- Thermal-Based Devices
Thyroid Ablation Devices Market, By Application:
- Thyroid Nodules
- Thyroid Cancer
Thyroid Ablation Devices Market, By End User:
- Hospitals & Clinics
- Ambulatory Surgical Centers
- Others
Thyroid Ablation Devices Market, By Region:
- North America
- United States
- Canada
- Mexico
- Asia-Pacific
- China
- India
- South Korea
- Australia
- Japan
- Europe
- Germany
- France
- United Kingdom
- Spain
- Italy
- South America
- Brazil
- Argentina
- Colombia
- Middle East & Africa
- South Africa
- Saudi Arabia
- UAE
Competitive Landscape
Company Profiles: Detailed analysis of the major companies present in the Global Thyroid Ablation Devices Market.Available Customizations:
With the given market data, the publisher offers customizations according to a company's specific needs. The following customization options are available for the report.Company Information
- Detailed analysis and profiling of additional market players (up to five).
This product will be delivered within 1-3 business days.
Table of Contents
Companies Mentioned
- Medtronic Plc
- StarMed Co. Ltd.
- Terumo Corporation
- Johnson & Johnson
- Theraclion SA
- Integra LifeSciences Corp
- Olympus Corp.
- MedWaves Inc
- BVM Medical System
- Boston Scientific Corporation
Table Information
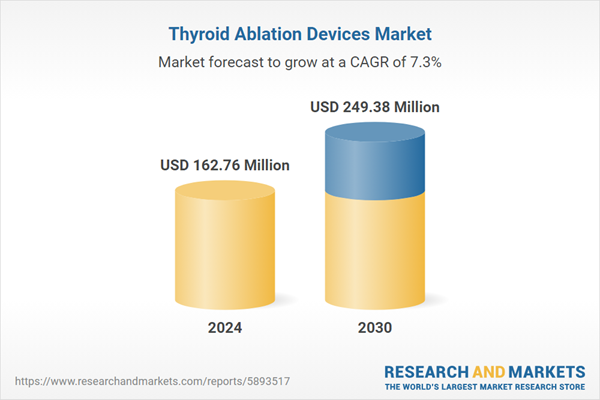
| Report Attribute | Details |
|---|---|
| No. of Pages | 190 |
| Published | June 2025 |
| Forecast Period | 2024 - 2030 |
| Estimated Market Value ( USD | $ 162.76 Million |
| Forecasted Market Value ( USD | $ 249.38 Million |
| Compound Annual Growth Rate | 7.3% |
| Regions Covered | Global |
| No. of Companies Mentioned | 10 |