Pediatric Interventional Cardiology Industry Overview
The pediatric interventional cardiology industry focuses on the diagnosis and minimally invasive treatment of congenital and acquired heart conditions in infants, children, and adolescents. This specialized field addresses a wide range of structural heart defects, such as septal defects, valve abnormalities, and vascular obstructions, using catheter-based techniques rather than open-heart surgery. The industry plays a critical role in improving clinical outcomes, reducing recovery times, and lowering procedural risks for pediatric patients.Pediatric interventional cardiology combines advanced medical devices, imaging technologies, and specialized clinical expertise. Key products include catheters, guidewires, balloons, stents, occlusion devices, and implantable valves designed specifically for smaller anatomies and growing patients. Continuous innovation is essential, as devices must balance durability, flexibility, and biocompatibility while accommodating future growth. Imaging guidance technologies, including fluoroscopy, echocardiography, and three-dimensional mapping, are integral to procedural accuracy and safety.
The industry is driven by increasing awareness and early diagnosis of congenital heart disease, improvements in neonatal and pediatric care, and rising survival rates of children with complex cardiac conditions. As more patients live into adolescence and adulthood, demand for repeat and staged interventions continues to grow. Pediatric interventional cardiology is also expanding into hybrid procedures that combine catheter-based and surgical techniques, further broadening treatment options.
Hospitals, specialty pediatric cardiac centers, and academic medical institutions are the primary end users, supported by highly trained interventional cardiologists and multidisciplinary care teams. The industry places strong emphasis on physician training, clinical evidence, and regulatory compliance due to the sensitive nature of pediatric care and the high standards required for device approval.
Despite its advancements, the industry faces challenges such as high development costs, limited patient populations compared to adult cardiology, and stringent regulatory pathways for pediatric-specific devices. These factors can slow innovation and market entry. However, collaborations between medical device manufacturers, healthcare providers, and research institutions continue to support progress.
Overall, the pediatric interventional cardiology industry is a highly specialized and innovation-driven sector that significantly enhances the quality of life and long-term outcomes for children with heart disease. Its continued growth is shaped by technological advancements, clinical expertise, and a strong commitment to patient-centered care.
Growth Drivers for the Pediatric Interventional Cardiology Market
Increasing Prevalence and Early Diagnosis of Congenital Heart Disease
A primary growth driver for the pediatric interventional cardiology market is the increasing prevalence and improved early diagnosis of congenital heart disease (CHD). Advancements in prenatal screening, fetal echocardiography, and neonatal diagnostic technologies have enabled earlier identification of structural heart defects, allowing timely intervention and improved clinical outcomes. Early diagnosis reduces complications associated with delayed treatment and increases the number of patients eligible for minimally invasive procedures rather than open-heart surgery. Additionally, heightened awareness among parents, caregivers, and healthcare professionals contributes to more frequent screening and referrals to specialized pediatric cardiac centers. Improved survival rates of premature infants and children with complex cardiac anomalies further expand the patient population requiring interventional procedures. With growing recognition of the benefits of catheter-based interventions - including shorter recovery times, reduced hospital stays, and lower procedural risks - the demand for pediatric interventional cardiology services continues to rise. Collectively, earlier detection and a larger patient base significantly drive market growth in this specialized healthcare sector.Technological Advancements in Devices and Imaging
Technological innovation is a key driver of growth in the pediatric interventional cardiology market. The development of specialized devices - such as occluders, stents, balloons, and implantable valves designed for smaller anatomies - has expanded treatment options for children with structural heart defects. These devices are increasingly engineered to accommodate patient growth, improve durability, and reduce the risk of complications, enhancing clinical outcomes. Advanced imaging technologies, including 3D echocardiography, fluoroscopy, and hybrid imaging systems, enable precise procedural guidance and improve procedural safety and efficacy. Minimally invasive catheter-based techniques, coupled with robotics and navigation systems, allow for greater procedural accuracy while reducing recovery time and hospital stays. The continuous introduction of innovative products and techniques attracts more hospitals and specialized centers to adopt interventional procedures. As technology evolves, physicians are better equipped to perform complex interventions in younger and high-risk pediatric patients, directly expanding the market for interventional cardiology devices and procedures.Growing Specialized Pediatric Cardiac Centers and Expertise
The expansion of specialized pediatric cardiac centers and the availability of trained interventional cardiologists are significant growth drivers for the pediatric interventional cardiology market. Hospitals and medical institutions are increasingly investing in dedicated pediatric cardiology units equipped with state-of-the-art catheterization labs and advanced imaging systems. These centers provide comprehensive care for children with congenital or acquired heart conditions, offering both diagnostic and therapeutic procedures under multidisciplinary supervision. The presence of highly skilled interventional cardiologists, nurses, and support staff ensures high procedural success rates and patient safety, which encourages greater adoption of minimally invasive interventions. Additionally, medical education programs, fellowships, and continuing professional development initiatives are enhancing the expertise available in this niche field. The growth of such specialized centers also improves access to care in previously underserved regions, expanding the potential patient population. As more institutions adopt advanced interventional techniques and build expertise, the overall demand for pediatric interventional cardiology devices and procedures continues to rise, driving market growth.Challenges in the Pediatric Interventional Cardiology Market
Limited Pediatric-Specific Devices and High Development Costs
A major challenge in the pediatric interventional cardiology market is the limited availability of pediatric-specific devices. Unlike adult cardiology, pediatric cardiology requires devices that accommodate smaller anatomies and allow for patient growth over time, which significantly narrows the market size for individual products. Developing these specialized devices involves extensive research, complex engineering, and rigorous preclinical testing to ensure safety and efficacy. The small patient population for rare congenital heart defects further limits potential returns on investment, making it financially challenging for manufacturers to justify development costs. Regulatory requirements for pediatric devices are stringent, often requiring long clinical trials and extensive safety documentation, which prolongs time to market and adds to expenses. These factors can slow innovation and restrict access to cutting-edge technologies for pediatric patients. Consequently, hospitals and interventional cardiologists may face limited device choices, potentially impacting procedural outcomes and patient care, while companies navigate a high-cost, high-risk development environment.Regulatory and Reimbursement Challenges
Regulatory and reimbursement complexities pose another significant challenge in the pediatric interventional cardiology market. Pediatric medical devices and procedures are subject to strict oversight from authorities such as the U.S. Food and Drug Administration (FDA) and similar bodies worldwide. Regulatory pathways for pediatric-specific devices often require additional clinical studies, demonstrating safety and efficacy in small, heterogeneous patient populations, which can delay approvals and increase costs. In addition, obtaining insurance coverage and reimbursement for advanced interventional procedures can be challenging, particularly in cases involving novel devices or minimally invasive techniques. Variability in payer policies, reimbursement rates, and coding practices can create financial uncertainty for hospitals and healthcare providers, potentially limiting patient access. Furthermore, the cost-intensive nature of pediatric cardiac interventions, combined with small patient volumes, makes it difficult for hospitals to maintain cost-effectiveness. These regulatory and reimbursement hurdles can slow the adoption of innovative therapies, limit market expansion, and restrict broader availability of life-saving interventional cardiology treatments for children.United States Pediatric Interventional Cardiology Market
The United States pediatric interventional cardiology market is a specialized and growing segment of the broader cardiovascular industry, focused on minimally invasive treatment of congenital and acquired heart defects in infants, children, and adolescents. The market is driven by increasing prevalence and early diagnosis of congenital heart disease, advancements in imaging technologies, and the development of pediatric-specific devices such as occluders, stents, and balloons designed for small anatomies. Leading hospitals and dedicated pediatric cardiac centers provide advanced catheterization facilities and highly skilled interventional cardiologists, supporting high procedural success and improved patient outcomes. Technological innovations, including 3D imaging, hybrid procedures, and live navigation systems, further enhance treatment safety and effectiveness. Challenges such as limited pediatric device availability, stringent regulatory requirements, and high development costs persist, but growing awareness, rising survival rates, and expanding specialized centers continue to drive adoption. Overall, the U.S. market is poised for steady growth, fueled by clinical innovation and increased patient access.Germany Pediatric Interventional Cardiology Market
The Germany pediatric interventional cardiology market is a well-established segment of the country’s advanced healthcare system, focused on minimally invasive treatment of congenital and acquired heart conditions in children. Germany’s strong medical infrastructure, specialized pediatric cardiac centers, and skilled interventional cardiologists support high-quality care and positive clinical outcomes. The market benefits from early diagnosis of congenital heart disease through widespread neonatal screening and advanced imaging technologies such as echocardiography, fluoroscopy, and 3D mapping. Pediatric-specific devices, including occluders, stents, balloons, and implantable valves designed for small anatomies, enable minimally invasive procedures that reduce hospital stays and recovery times. The country’s emphasis on clinical research, technological innovation, and regulatory compliance drives adoption of advanced interventional techniques. However, challenges such as high device development costs and strict European regulatory requirements limit rapid market expansion. Overall, Germany’s pediatric interventional cardiology market is characterized by technological advancement, specialized care, and steady growth, addressing increasing demand for safe and effective minimally invasive cardiac treatments for children.India Pediatric Interventional Cardiology Market
The India pediatric interventional cardiology market is an emerging and rapidly growing segment, driven by increasing awareness of congenital heart disease (CHD), expanding healthcare infrastructure, and rising demand for minimally invasive procedures in children. India has witnessed significant improvements in neonatal and pediatric cardiac care, supported by specialized hospitals and cardiac centers in major cities such as Mumbai, Delhi, Bangalore, and Chennai. Advances in diagnostic imaging, including echocardiography, fluoroscopy, and 3D mapping, enable early detection and precise treatment of structural heart defects. Pediatric-specific devices like occluders, balloons, stents, and valves are increasingly being adopted, allowing fewer invasive procedures with shorter hospital stays and improved outcomes. Government initiatives, rising disposable incomes, and growing health insurance penetration further support market growth. Challenges such as high device costs, limited availability of trained interventional cardiologists, and uneven access to care in rural areas remain, but continued investment and awareness are expected to drive steady expansion of the pediatric interventional cardiology market in India.United Arab Emirates Pediatric Interventional Cardiology Market
The United Arab Emirates (UAE) pediatric interventional cardiology market is a developing and promising segment of the country’s advanced healthcare system. The market is driven by increasing awareness of congenital heart disease (CHD), improvements in pediatric healthcare infrastructure, and rising demand for minimally invasive cardiac procedures. Leading hospitals and specialized pediatric cardiac centers in cities like Dubai and Abu Dhabi offer state-of-the-art catheterization labs and advanced imaging technologies, including echocardiography, fluoroscopy, and 3D mapping, enabling accurate diagnosis and treatment. Pediatric-specific devices such as occluders, stents, balloons, and implantable valves support minimally invasive interventions, reducing hospital stays, recovery times, and procedural risks. Government initiatives to enhance healthcare quality, medical tourism, and rising healthcare expenditure further boost market growth. Challenges include high device costs, limited trained specialists, and dependence on imported medical equipment. Overall, the UAE pediatric interventional cardiology market is poised for growth, supported by technological advancements, specialized care, and increasing adoption of minimally invasive pediatric cardiac procedures.Recent Developments in Pediatric Interventional Cardiology Market
- June 2025: Canid completed a $10 million Series Investor confidence in pediatric healthcare technology solutions that improve patient outcomes and streamline clinical workflows is demonstrated by the funding to develop its pediatric vaccine management platform.
- April 2025: The FDA approved Abbott's Tendyne transcatheter mitral valve replacement technology, which is the first of its kind and will help patients with severe mitral annular calcification.
- January 2025: Boston Scientific successfully acquired Bolt Medical for up to USD 664 million, increasing its capacity to treat complex calcified artery disease in both adult and pediatric populations using intravascular lithotripsy technology.
- A major development in pediatric mechanical circulatory support occurred in December 2024 when Johnson & Johnson was granted FDA clearance to extend the Impella heart pump's indications for young patients with symptomatic acute decompensated heart failure and cardiogenic shock.
Market Segmentation
Device Type
- Transcatheter Heart Valves
- Atherectomy Devices
- Catheters
- Closure Devices
- Balloons
- Stents
- Others
Procedure
- Catheter- Based Valve I mplation
- Congenital Heart Defect Correction
- Angioplasty
- Coronary Thrombectomy
- Others
End Use
- Hospitals
- Ambulatory Surgical Centers
- Specialty Clinic
Countries
North America
- United States
- Canada
Europe
- France
- Germany
- Italy
- Spain
- United Kingdom
- Belgium
- Netherlands
- Turkey
Asia Pacific
- China
- Japan
- India
- Australia
- South Korea
- Thailand
- Malaysia
- Indonesia
- New Zealand
Latin America
- Brazil
- Mexico
- Argentina
Middle East & Africa
- South Africa
- Saudi Arabia
- United Arab Emirates
Rest of the World
Company Insights:
- Overview
- Key Persons
- Recent Development
- SWOT Analysis
- Revenue Analysis
Competitive Landscape
- Abbott Laboratories
- Boston Scientific Corporation
- General Electric
- Medtronic
- Siemens AG
- Cardinal Health Inc.
Table of Contents
Companies Mentioned
The companies featured in this Pediatric Interventional Cardiology market report include:- Abbott Laboratories
- Boston Scientific Corporation
- General Electric
- Medtronic
- Siemens AG
- Cardinal Health Inc.
Methodology
In this report, for analyzing the future trends for the studied market during the forecast period, the publisher has incorporated rigorous statistical and econometric methods, further scrutinized by secondary, primary sources and by in-house experts, supported through their extensive data intelligence repository. The market is studied holistically from both demand and supply-side perspectives. This is carried out to analyze both end-user and producer behavior patterns, in the review period, which affects price, demand and consumption trends. As the study demands to analyze the long-term nature of the market, the identification of factors influencing the market is based on the fundamentality of the study market.
Through secondary and primary researches, which largely include interviews with industry participants, reliable statistics, and regional intelligence, are identified and are transformed to quantitative data through data extraction, and further applied for inferential purposes. The publisher's in-house industry experts play an instrumental role in designing analytic tools and models, tailored to the requirements of a particular industry segment. These analytical tools and models sanitize the data & statistics and enhance the accuracy of their recommendations and advice.
Primary Research
The primary purpose of this phase is to extract qualitative information regarding the market from the key industry leaders. The primary research efforts include reaching out to participants through mail, tele-conversations, referrals, professional networks, and face-to-face interactions. The publisher also established professional corporate relations with various companies that allow us greater flexibility for reaching out to industry participants and commentators for interviews and discussions, fulfilling the following functions:
- Validates and improves the data quality and strengthens research proceeds
- Further develop the analyst team’s market understanding and expertise
- Supplies authentic information about market size, share, growth, and forecast
The researcher's primary research interview and discussion panels are typically composed of the most experienced industry members. These participants include, however, are not limited to:
- Chief executives and VPs of leading corporations specific to the industry
- Product and sales managers or country heads; channel partners and top level distributors; banking, investment, and valuation experts
- Key opinion leaders (KOLs)
Secondary Research
The publisher refers to a broad array of industry sources for their secondary research, which typically includes, however, is not limited to:
- Company SEC filings, annual reports, company websites, broker & financial reports, and investor presentations for competitive scenario and shape of the industry
- Patent and regulatory databases for understanding of technical & legal developments
- Scientific and technical writings for product information and related preemptions
- Regional government and statistical databases for macro analysis
- Authentic new articles, webcasts, and other related releases for market evaluation
- Internal and external proprietary databases, key market indicators, and relevant press releases for market estimates and forecasts

LOADING...
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 200 |
| Published | February 2026 |
| Forecast Period | 2025 - 2034 |
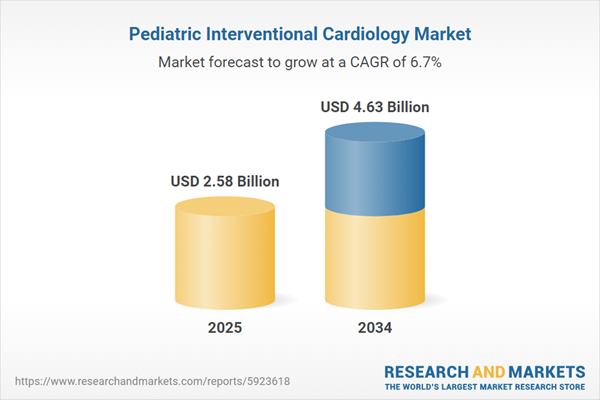
| Estimated Market Value ( USD | $ 2.58 Billion |
| Forecasted Market Value ( USD | $ 4.63 Billion |
| Compound Annual Growth Rate | 6.7% |
| Regions Covered | Global |
| No. of Companies Mentioned | 7 |