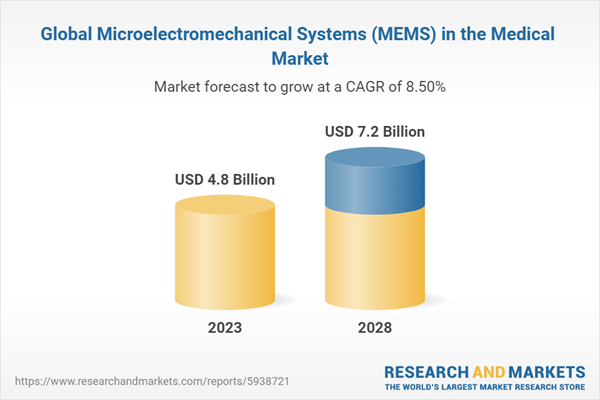
Chapter 2 Summary and Highlights
- Market Outlook
- Market Insights
Chapter 3 Market Overview
- Introduction
- Technology Evolution and Advantages
- MEMS Devices and Applications for the Medical Market
- Regulatory Environment
Chapter 4 Market Dynamics
- Overview
- Market Drivers
- Widening Range of Applications
- Increasing Use of MEMS Devices in Healthcare
- Advances in Technology
- Rising Prevalences of Infectious and Chronic Diseases
- Market Restraints
- Lengthy and Expensive Product Development Cycles
- Lack of a Standardized Fabrication Process for MEMS
- Substituting Nanoelectromechanical Systems (NEMS) for MEMS
- Market Opportunities
- Increasing Government Investment in Healthcare
- R&D of MEMS Devices for Medical Purposes
Chapter 5 Market Breakdown by Product
- Introduction
- MEMS Sensors
- Pressure Sensors
- Optical Sensors
- Inertial Sensors
- Other Sensors
- Microfluidics/Lab-on-a-Chip
- Clinical In vitro Diagnostics
- Analytical In vitro Diagnostics
- Point-Of-Care In vitro Diagnostics
- Medical Research
- Drug Delivery Devices
- Micropumps
- Microneedles
- Inhalers/Nebulizers
Chapter 6 Market Breakdown by Application
- Introduction
- Home Healthcare Market
- Pharmaceutical/Life Sciences Research Market
- High-Throughput Screening
- Genomics
- Cellular Analysis
- Proteomics
- Other Research Applications
- In-vitro Diagnostics Market
- Molecular Diagnostics
- Immunology
- Biochemistry
- Other In vitro Diagnostic Applications
- Medical Devices
- Diagnosis and Patient Monitoring
- Surgical and Medical Equipment
Chapter 7 Market Breakdown by Region
- Overview
- North America
- Europe
- Asia-Pacific
- Rest of the World
Chapter 8 ESG Development
- Introduction to ESG
- ESG Performance in the MEMS Market
- Environmental Impact
- Social Responsibility
- Governance
- Consumer Attitudes Towards ESG in the MEMS Industry
- Case Study
Chapter 9 Emerging Technologies
- Overview
- Emerging Trends
- Miniaturization
- MEMS Mirrors
- Devices
- MEMS-based Piezoelectric Microphones
- Nanomaterials in MEMS
- MEMS Technology
Chapter 11 Competitive Landscape
- Competitive Analysis
- M&A Analysis
Chapter 12 Company Profiles
- Abbott Laboratories
- All Sensors Corp.
- Analog Devices Inc.
- Honeywell International Inc.
- Integrated Sensing Systems Inc.
- Knowles Electronics Llc
- Memscap
- Silex Microsystems Ab
- STMicroelectronics
- TE Connectivity
- Teledyne Dalsa
- Texas Instruments Inc.
List of Tables
Summary Table: Global Market for MEMS Devices in Medical Applications, by Region, Through 2028
Table 1: Global Market for MEMS Devices in Medical Applications, by Type, Through 2028
Table 2: BioMEMS Devices and their Applications
Table 3: Global Market for MEMS Devices in Medical Applications, by Product, Through 2028
Table 4: Global Market for MEMS Devices in Medical Applications, by Region, Through 2028
Table 5: Global Market for MEMS Devices in Medical Applications for Sensors, by Type, Through 2028
Table 6: Global Market for MEMS Devices in Medical Applications for Sensors, by Region, Through 2028
Table 7: Global Market for MEMS Devices in Medical Applications for Pressure Sensors, by Application, Through 2028
Table 8: Global Market for MEMS Devices in Medical Applications for Pressure Sensors, by Region, Through 2028
Table 9: Global Market for MEMS Devices in Medical Applications for Blood Pressure Monitoring, by Region, Through 2028
Table 10: Global Market for MEMS Devices in Medical Applications for Other Pressure Sensors, by Region, Through 2028
Table 11: Global Market for MEMS Devices in Medical Applications for Optical Sensors, by Region, Through 2028
Table 12: Global Market for MEMS Devices in Medical Applications for Inertial Sensors, by Region, Through 2028
Table 13: Global Market for MEMS Devices in Medical Applications for Other Sensors, by Type, Through 2028
Table 14: Global Market for MEMS Devices in Medical Applications for Other Sensors, by Region, Through 2028
Table 15: Global Market for MEMS Devices Used in Medical Applications for Microfluidics/LOC, by Application, Through 2028
Table 16: Global Market for MEMS Devices Used in Medical Applications for Microfluidics/LOC MEMS, by Region, Through 2028
Table 17: Global Market for MEMS Devices Used in Medical Applications for Clinical In vitro Diagnostics Applications, by Region, Through 2028
Table 18: Global Market for MEMS Devices Used in Medical Applications for Analytical In vitro Diagnostics Applications, by Region, Through 2028
Table 19: Global Market for MEMS Devices Used in Medical Applications for Point-of-Care In vitro Diagnostics Applications, by Region, Through 2028
Table 20: Global Market for MEMS Devices Used in Medical Applications for Medical Research Applications, by Region, Through 2028
Table 21: Global Market for MEMS Devices Used in Medical Applications for Drug Delivery Systems, by Type, Through 2028
Table 22: Global Market for MEMS Devices Used in Medical Applications for Drug Delivery Systems, by Region, Through 2028
Table 23: Global Market for MEMS Devices in Medical Applications, by Application, Through 2028
Table 24: Global Market for MEMS Devices for Home Healthcare Applications, by Region, Through 2028
Table 25: Global Market for MEMS Devices in Pharmaceutical and Life Sciences Research Applications, by Type, Through 2028
Table 26: Global Market for MEMS Devices in Pharmaceutical and Life Sciences Research Applications, by Region, Through 2028
Table 27: Global Market for MEMS Devices in High-Throughput Screening Applications, by Region, Through 2028
Table 28: Global Market for MEMS Devices in Genomic Applications, by Region, Through 2028
Table 29: Global Market for MEMS Devices in Cellular Analysis Applications, by Region, Through 2028
Table 30: Global Market for MEMS Devices in Proteomics Applications, by Region, Through 2028
Table 31: Global Market for MEMS Devices in Other Pharmaceutical and Life Sciences Research Applications, by Region, Through 2028
Table 32: Global Market for MEMS Devices in In-vitro Diagnostics Applications, by Type, Through 2028
Table 33: Global Market for MEMS Devices in In-vitro Diagnostics Applications, by Region, Through 2028
Table 34: Global Market for MEMS Devices in Molecular Diagnostics Applications, by Region, Through 2028
Table 35: Global Market for MEMS Devices in Immunology Applications, by Region, Through 2028
Table 36: Global Market for MEMS Devices in Biochemistry Applications, by Region, Through 2028
Table 37: Global Market for MEMS Devices in Other In vitro Diagnostics Applications, by Region, Through 2028
Table 38: Global Market for MEMS Devices in Medical Devices Applications, by Type, Through 2028
Table 39: Global Market for MEMS Devices in Medical Devices Applications, by Region, Through 2028
Table 40: Global Market for MEMS Devices in Diagnosis and Patient Monitoring Applications, by Region, Through 2028
Table 41: Global Market for MEMS Used in Surgical and Medical Equipment Applications, by Region, Through 2028
Table 42: Global Market for MEMS Devices in Medical Applications, by Region, Through 2028
Table 43: ESG Rankings for MEMS Companies, 2023*
Table 44: Comparison of Parameters of MEMS Mirrors
Table 45: Patents Granted for MEMS in Medical Applications, 2021-2023
Table 46: Leading Makers of MEMS in Medical Applications, 2022
Table 47: Recent Developments in the MEMs Market, 2024
Table 48: Abbott: Company Snapshot
Table 49: Abbott: Annual Revenue, 2021 and 2022
Table 50: Abbott: Recent Developments, 2022
Table 51: All Sensors Corp.: Company Snapshot
Table 52: All Sensors Corp.: MEMS Offerings
Table 53: All Sensors Corp.: Recent Developments, 2020
Table 54: Analog Devices Inc.: Company Snapshot
Table 55: Analog Devices Inc.: Annual Revenue, 2022
Table 56: Analog Devices Inc.: Recent Developments, 2023
Table 57: Honeywell International Inc.: Company Snapshot
Table 58: Honeywell International Inc.: MEMS Offerings
Table 59: Honeywell International Inc.: Annual Revenue, 2022
Table 60: Integrated Sensing Systems Inc.: Company Snapshot
Table 61: Integrated Sensing Systems Inc.: MEMS Offerings
Table 62: Knowles Electronics LLC: Company Snapshot
Table 63: Knowles Electronics LLC: MEMS Offerings
Table 64: Knowles Electronics LLC: Annual Revenue, 2022
Table 65: Knowles Electronics LLC: Recent Developments, 2023
Table 66: MEMSCAP: Company Snapshot
Table 67: MEMScap: MEMS Offerings
Table 68: MEMScap: Recent Developments,2022
Table 69: Silex Microsystems AB: Company Snapshot
Table 70: Silex Microsystems AB: MEMS Offerings
Table 71: Silex Microsystems AB: Recent Development, 2023
Table 72: STMicroelectronics: Company Snapshot
Table 73: STMicroelectronics: MEMS Offerings
Table 74: STMicroelectronics: Annual Revenue, 2022
Table 75: STMicroelectronics: Recent Developments, 2023
Table 76: TE Connectivity: Company Snapshot
Table 77: TE Connectivity: Annual Revenue, 2022
Table 78: TE Connectivity: Recent Developments,2023
Table 79: Teledyne DALSA: Company Snapshot
Table 80: Teledyne Dalsa: Recent Developments, 2023
Table 81: Texas Instruments Inc.: Company Snapshot
Table 82: Texas Instruments Inc.: Annual Revenue, 2022
Table 83: Texas Instruments Inc.: Recent Developments, 2023
List of Figures
Summary Figure: Global Market Shares of MEMS Devices in Medical Applications, by Region, 2022
Figure 1: Flowchart of FDA Approval Steps
Figure 2: Microelectromechanical Systems: Market Dynamics
Figure 3: CAGRs for MEMS Devices in Medical Applications, by Region
Figure 4: How a Strong ESG Proposition Benefits Businesses
Figure 5: Levels of ESG Adoption Across All Industries, 2021 and 2022
Figure 6: Honeywell International: ESG Highlights, 2022 and 2023
Figure 7: Honeywell International: ESG-Oriented Solutions
Figure 8: Patent Document, 2021 to November 2023
Figure 9: Total Capital Raised and Number of Deals by BioMEMS Investors, 2014-2023
Figure 10: Abbott: Annual Revenue, 2021 and 2022
Figure 11: Abbott: Revenue Shares, by Business Unit, 2022
Figure 12: Abbott: Revenue Shares, by Country/Region, 2022
Figure 13: Analog Devices Inc.: Annual Revenue, 2021 and 2022
Figure 14: Analog Devices Inc.: Revenue Shares, by Business Unit, 2022
Figure 15: Analog Devices Inc.: Revenue Shares, by Country/Region, 2022
Figure 16: Honeywell International Inc.: Annual Revenue, 2021 and 2022
Figure 17: Honeywell International Inc.: Revenue Shares, by Segment, 2022
Figure 18: Honeywell International Inc.: Revenue Shares, by Country/Region, 2022
Figure 19: Knowles Electronics LLC: Annual Revenue, 2021 and 2022
Figure 20: Knowles Electronics LLC: Revenue Shares, by Segment, 2022
Figure 21: STMicroelectronics: Annual Revenue, 2021 and 2022
Figure 22: STMicroelectronics: Revenue Shares, by Region, 2022
Figure 23: TE Connectivity: Annual Revenue, 2021 and 2022
Figure 24: TE Connectivity: Revenue Shares, by Segment, 2022
Figure 25: TE Connectivity: Revenue Shares, by Region, 2022
Figure 26: Texas Instruments Inc.: Annual Revenue, 2021 and 2022
Figure 27: Texas Instruments Inc.: Revenue Shares, by Product, 2022
Figure 28: Texas Instruments Inc.: Revenue Shares, by Country/Region, 2022