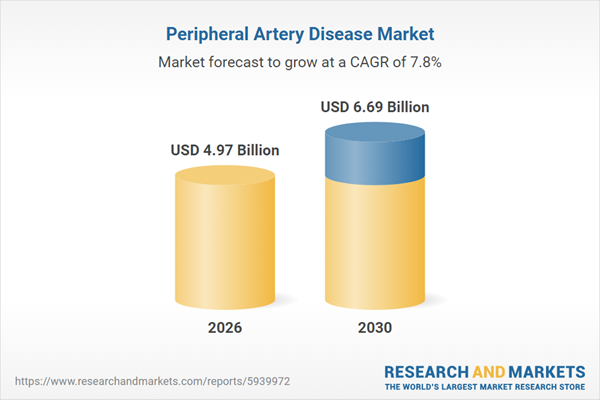
The peripheral artery disease market size is expected to see strong growth in the next few years. It will grow to $6.69 billion in 2030 at a compound annual growth rate (CAGR) of 7.8%. The growth in the forecast period can be attributed to advancements in minimally invasive peripheral interventions, growing adoption of drug-eluting balloons and stents, expansion of hospitals and specialty vascular clinics, rising patient awareness and early detection initiatives, increasing use of combination therapies including lifestyle modification and pharmacotherapy. Major trends in the forecast period include increasing use of ai for early pad diagnosis and risk stratification, rising adoption of cloud-based patient monitoring systems, growing focus on genomic-guided personalized pad therapies, increasing integration of iot devices for post-intervention tracking, rising implementation of smart manufacturing in pad device production.
The increasing prevalence of peripheral artery disease is expected to drive the growth of the peripheral artery disease market in the coming years. Peripheral artery disease involves the narrowing or obstruction of arteries outside the heart, most commonly in the legs, which leads to reduced blood flow and an increased risk of complications. Treatment for peripheral artery disease helps alleviate symptoms, improve mobility, and lower the likelihood of serious complications, thereby enhancing patients’ overall quality of life. For instance, in June 2023, according to the American Heart Association, a US-based nonprofit organization, it was estimated that between 7 million and 12 million individuals in the United States and around 200 million people worldwide are affected by peripheral artery disease. Therefore, the rising prevalence of peripheral artery disease is contributing to the growth of the peripheral artery disease market.
Major companies operating in the peripheral artery disease market are concentrating on the development of technologically advanced solutions, such as dissolvable drug-eluting scaffold systems, to address the growing demand for safer, more durable, and minimally invasive treatment options for below-the-knee arterial blockages. These resorbable scaffold technologies are designed to restore blood flow and then gradually dissolve, offering advantages over permanent metallic stents. For example, in April 2024, Abbott Laboratories, a US-based medical device company, launched the Esprit BTK Everolimus Eluting Resorbable Scaffold System, a first-of-its-kind dissolvable stent therapy for below-the-knee peripheral artery disease. The system delivers everolimus to help prevent vessel re-narrowing, while its fully resorbable structure dissolves over time, reducing long-term risks associated with metal implants. It provides physicians with an advanced alternative to traditional angioplasty or permanent stenting, particularly benefiting patients with long lesions or critical limb ischemia. By combining targeted drug delivery with temporary structural support, the Esprit BTK System aims to improve vessel healing, reduce complications, and expand treatment options for complex peripheral artery disease cases.
In April 2023, Abbott Laboratories, a US-based medical device company, acquired Cardiovascular Systems Inc. for an undisclosed amount. Through this acquisition, Abbott expanded and strengthened its peripheral and coronary artery disease portfolio by adding Cardiovascular Systems Inc.’s atherectomy technologies. Cardiovascular Systems Inc. is a US-based medical device company engaged in the development and manufacturing of products for the treatment of peripheral artery disease.
Major companies operating in the peripheral artery disease market are Cardinal Health Inc., Vascular Solutions Inc., Bayer AG, Abbott Laboratories, Medtronic plc., Koninklijke Philips N.V. (Royal Philips), B. Braun Melsungen AG., Terumo Corporation, Edwards Lifesciences, Becton Dickinson and Company (BD), Boston Scientific Corporation Incorporated, Cook Group Incorporated, Merit Medical Systems Inc., NuVasive Inc., Arjo AB, Penumbra Inc., BIOTRONIK SE and Co. KG, AngioDynamics Inc., Shockwave Medical Inc., Cardiovascular Systems Inc., Endologix LLC., LeMaitre Vascular Inc., Silk Road Medical Inc., WL Gore and Associates Inc., MicroPort Scientific Corporation, Bard Peripheral Vascular Inc., Avinger Inc.
North America was the largest region in the peripheral artery disease market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the peripheral artery disease market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in the peripheral artery disease market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.
Tariffs have influenced the peripheral artery disease market by raising costs of imported devices, catheters, stents, and drugs, particularly in North America and Europe. This has temporarily slowed adoption in hospitals and specialty clinics, while also encouraging local manufacturing, fostering innovation in cost-efficient PAD devices, and strengthening supply chain resilience for interventional treatments and therapies.
The peripheral artery disease market research report is one of a series of new reports that provides peripheral artery disease market statistics, including peripheral artery disease industry global market size, regional shares, competitors with a peripheral artery disease market share, detailed peripheral artery disease market segments, market trends and opportunities, and any further data you may need to thrive in the peripheral artery disease industry. This peripheral artery disease market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future scenario of the industry.
Peripheral artery disease (PAD) is a condition in which the arteries supplying blood to the limbs, most often the legs, become narrowed or blocked. It is commonly caused by atherosclerosis, a process marked by the buildup of fatty deposits, or plaques, within the arterial walls, leading to reduced blood flow to the affected areas.
The primary products used in the treatment of peripheral artery disease include peripheral angioplasty balloons, peripheral stents, peripheral catheters, inferior vena cava (IVC) filters, plaque modification devices, hemodynamic flow alteration devices, and peripheral accessories. Peripheral angioplasty balloons are medical devices designed to widen narrowed or obstructed peripheral arteries and are used in PAD treatment by inflating the balloon to compress plaque and expand the artery, thereby restoring blood flow. The drugs used for managing PAD include lipid-lowering agents, blood pressure-lowering drugs, glucose-regulating medications, blood clot-preventing drugs, inotropes, thrombolytic agents, and anti-inflammatory agents. These therapies are administered through routes such as oral, parenteral, and others, and are utilized by end users including hospitals, specialty clinics, and others.
The peripheral artery disease market consists of revenues earned by entities by providing peripheral artery disease management services and therapies such as diagnosis, angioplasty, atherectomy, lithoplasty, endovascular, vascular surgery, and rehabilitation. The market value includes the value of related goods sold by the service provider or included within the service offering. The peripheral artery disease market also includes sales of guidewires, bare metal, drug-eluting, compression stockings, and exercise equipment. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified).
The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.
This product will be delivered within 1-3 business days.
Table of Contents
Executive Summary
Peripheral Artery Disease Market Global Report 2026 provides strategists, marketers and senior management with the critical information they need to assess the market.This report focuses peripheral artery disease market which is experiencing strong growth. The report gives a guide to the trends which will be shaping the market over the next ten years and beyond.
Reasons to Purchase:
- Gain a truly global perspective with the most comprehensive report available on this market covering 16 geographies.
- Assess the impact of key macro factors such as geopolitical conflicts, trade policies and tariffs, inflation and interest rate fluctuations, and evolving regulatory landscapes.
- Create regional and country strategies on the basis of local data and analysis.
- Identify growth segments for investment.
- Outperform competitors using forecast data and the drivers and trends shaping the market.
- Understand customers based on end user analysis.
- Benchmark performance against key competitors based on market share, innovation, and brand strength.
- Evaluate the total addressable market (TAM) and market attractiveness scoring to measure market potential.
- Suitable for supporting your internal and external presentations with reliable high-quality data and analysis
- Report will be updated with the latest data and delivered to you along with an Excel data sheet for easy data extraction and analysis.
- All data from the report will also be delivered in an excel dashboard format.
Description
Where is the largest and fastest growing market for peripheral artery disease? How does the market relate to the overall economy, demography and other similar markets? What forces will shape the market going forward, including technological disruption, regulatory shifts, and changing consumer preferences? The peripheral artery disease market global report answers all these questions and many more.The report covers market characteristics, size and growth, segmentation, regional and country breakdowns, total addressable market (TAM), market attractiveness score (MAS), competitive landscape, market shares, company scoring matrix, trends and strategies for this market. It traces the market’s historic and forecast market growth by geography.
- The market characteristics section of the report defines and explains the market. This section also examines key products and services offered in the market, evaluates brand-level differentiation, compares product features, and highlights major innovation and product development trends.
- The supply chain analysis section provides an overview of the entire value chain, including key raw materials, resources, and supplier analysis. It also provides a list competitor at each level of the supply chain.
- The updated trends and strategies section analyses the shape of the market as it evolves and highlights emerging technology trends such as digital transformation, automation, sustainability initiatives, and AI-driven innovation. It suggests how companies can leverage these advancements to strengthen their market position and achieve competitive differentiation.
- The regulatory and investment landscape section provides an overview of the key regulatory frameworks, regularity bodies, associations, and government policies influencing the market. It also examines major investment flows, incentives, and funding trends shaping industry growth and innovation.
- The market size section gives the market size ($b) covering both the historic growth of the market, and forecasting its development.
- The forecasts are made after considering the major factors currently impacting the market. These include the technological advancements such as AI and automation, Russia-Ukraine war, trade tariffs (government-imposed import/export duties), elevated inflation and interest rates.
- The total addressable market (TAM) analysis section defines and estimates the market potential compares it with the current market size, and provides strategic insights and growth opportunities based on this evaluation.
- The market attractiveness scoring section evaluates the market based on a quantitative scoring framework that considers growth potential, competitive dynamics, strategic fit, and risk profile. It also provides interpretive insights and strategic implications for decision-makers.
- Market segmentations break down the market into sub markets.
- The regional and country breakdowns section gives an analysis of the market in each geography and the size of the market by geography and compares their historic and forecast growth.
- Expanded geographical coverage includes Taiwan and Southeast Asia, reflecting recent supply chain realignments and manufacturing shifts in the region. This section analyzes how these markets are becoming increasingly important hubs in the global value chain.
- The competitive landscape chapter gives a description of the competitive nature of the market, market shares, and a description of the leading companies. Key financial deals which have shaped the market in recent years are identified.
- The company scoring matrix section evaluates and ranks leading companies based on a multi-parameter framework that includes market share or revenues, product innovation, and brand recognition.
Report Scope
Markets Covered:
1) By Type: Peripheral Angioplasty Balloons; Peripheral Stents; Peripheral Catheters; Inferior Vena Cava (IVC) Filters; Plaque Modification Devices; Hemodynamic Flow Alteration Devices; Peripheral Accessories2) By Drugs: Lipid-Lowering Drugs; Blood Pressure Lowering Drugs; Glucose Regulating Drugs; Blood Clot Preventing Drugs; Inotropes; Thrombolytics Agents; Anti-Inflammatory Agents
3) By Route Of Administration: Oral; Parenteral; Other Routes Of Administrations
4) By End Users: Hospitals; Specialty Clinics; Other End Users
Subsegments:
1) By Peripheral Angioplasty Balloons: Drug-Eluting Balloons; Non-Drug-Eluting Balloons; Scoring Balloons; Cutting Balloons2) By Peripheral Stents: Drug-Eluting Stents; Bare Metal Stents; Covered Stents
3) By Peripheral Catheters: Sheath Introducers; Guidewires; Diagnostic Catheters; Delivery Catheters
4) By Inferior Vena Cava (IVC) Filters: Retrievable IVC Filters; Permanent IVC Filters
5) By Plaque Modification Devices: Atherectomy Devices; Laser Atherectomy Devices; Rotational Atherectomy Devices
6) By Hemodynamic Flow Alteration Devices: Flow Diversion Devices; Flow Restriction Devices
7) By Peripheral Accessories: Diagnostic Imaging Accessories; Accessory Kits For Interventional Procedures; Support Devices
Companies Mentioned: Cardinal Health Inc.; Vascular Solutions Inc.; Bayer AG; Abbott Laboratories; Medtronic plc.; Koninklijke Philips N.V. (Royal Philips); B. Braun Melsungen AG.; Terumo Corporation; Edwards Lifesciences; Becton Dickinson and Company (BD); Boston Scientific Corporation Incorporated; Cook Group Incorporated; Merit Medical Systems Inc.; NuVasive Inc.; Arjo AB; Penumbra Inc.; BIOTRONIK SE and Co. KG; AngioDynamics Inc.; Shockwave Medical Inc.; Cardiovascular Systems Inc.; Endologix LLC.; LeMaitre Vascular Inc.; Silk Road Medical Inc.; WL Gore and Associates Inc.; MicroPort Scientific Corporation; Bard Peripheral Vascular Inc.; Avinger Inc.
Countries: Australia; Brazil; China; France; Germany; India; Indonesia; Japan; Taiwan; Russia; South Korea; UK; USA; Canada; Italy; Spain.
Regions: Asia-Pacific; South East Asia; Western Europe; Eastern Europe; North America; South America; Middle East; Africa
Time Series: Five years historic and ten years forecast.
Data: Ratios of market size and growth to related markets, GDP proportions, expenditure per capita.
Data Segmentation: Country and regional historic and forecast data, market share of competitors, market segments.
Sourcing and Referencing: Data and analysis throughout the report is sourced using end notes.
Delivery Format: Word, PDF or Interactive Report + Excel Dashboard
Added Benefits:
- Bi-Annual Data Update
- Customisation
- Expert Consultant Support
Companies Mentioned
The companies featured in this Peripheral Artery Disease market report include:- Cardinal Health Inc.
- Vascular Solutions Inc.
- Bayer AG
- Abbott Laboratories
- Medtronic plc.
- Koninklijke Philips N.V. (Royal Philips)
- B. Braun Melsungen AG.
- Terumo Corporation
- Edwards Lifesciences
- Becton Dickinson and Company (BD)
- Boston Scientific Corporation Incorporated
- Cook Group Incorporated
- Merit Medical Systems Inc.
- NuVasive Inc.
- Arjo AB
- Penumbra Inc.
- BIOTRONIK SE and Co. KG
- AngioDynamics Inc.
- Shockwave Medical Inc.
- Cardiovascular Systems Inc.
- Endologix LLC.
- LeMaitre Vascular Inc.
- Silk Road Medical Inc.
- WL Gore and Associates Inc.
- MicroPort Scientific Corporation
- Bard Peripheral Vascular Inc.
- Avinger Inc.
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 250 |
| Published | February 2026 |
| Forecast Period | 2026 - 2030 |
| Estimated Market Value ( USD | $ 4.97 Billion |
| Forecasted Market Value ( USD | $ 6.69 Billion |
| Compound Annual Growth Rate | 7.8% |
| Regions Covered | Global |
| No. of Companies Mentioned | 28 |