The epilepsy market has been comprehensively analyzed in this report titled "Epilepsy Market: Epidemiology, Industry Trends, Share, Size, Growth, Opportunity, and Forecast 2024-2034". Epilepsy is a neurological condition distinguished by recurrent seizures, which are sudden and uncontrolled bursts of electrical activity in the brain. These seizures are brief periods of uncontrollable movement involving a portion of the body (partial) or the entire body (generalized) and are occasionally followed by loss of awareness and control of bowel or bladder function. Symptoms of epilepsy can vary widely, but common ones include convulsions, loss of consciousness, and muscle spasms. Epilepsy is often diagnosed through a medical history review, physical examination, imaging tests, and diagnostic testing, such as an electroencephalogram (EEG). Diagnosis of epilepsy may also entail ruling out other illnesses that may cause seizures. Treatment for epilepsy typically consists of a combination of medications and lifestyle changes.
The rising prevalence of several associated risk factors, including neurological disorders, birth-related injuries, infections, substance abuse, road accidents, etc., is primarily driving the global epilepsy market. In addition to this, the expanding geriatric population, who are more prone to developing age-related conditions, such as Alzheimer's disease or Parkinson's disease, that can increase the risk of epilepsy, is also augmenting the market growth. Moreover, the widespread adoption of anti-epileptic drugs (AEDs) for treating epilepsy, which stabilize the electrical activity in the brain and prevent seizures, is also creating a positive outlook for the market. Apart from this, the increasing utilization of vagus nerve stimulation (VNS) devices to help reduce the frequency of seizures is further acting as a significant growth-inducing factor. The device can be programmed to deliver stimulation at specific times and intensities, and can be adjusted as needed. Additionally, the ongoing development of extended-release (ER) drug formulations that allow longer dosing intervals while minimizing the fluctuations in serum drug levels, thus improving efficacy as compared to immediate-release (IR) drugs, is also propelling the global market. Besides this, the implementation of numerous government initiatives to raise awareness about the ailment, promote research, and offer active assistance by expediting the development of better treatment options is further augmenting the market growth. Additionally, several key players are making significant investments in R&D activities aimed at introducing novel medications that are less expensive, more effective, and have fewer adverse effects. This, in turn, is expected to drive the global epilepsy market in the coming years.
This report provides an exhaustive analysis of the epilepsy market in the United States, EU5 (Germany, Spain, Italy, France, and United Kingdom) and Japan. This includes treatment practices, in-market, and pipeline drugs, share of individual therapies, market performance across the seven major markets, market performance of key companies and their drugs, etc. The report also provides the current and future patient pool across the seven major markets. According to the report the United States has the largest patient pool for epilepsy and also represents the largest market for its treatment. Furthermore, the current treatment practice/algorithm, market drivers, challenges, opportunities, reimbursement scenario and unmet medical needs, etc. have also been provided in the report. This report is a must-read for manufacturers, investors, business strategists, researchers, consultants, and all those who have any kind of stake or are planning to foray into the epilepsy market in any manner.
Time Period of the Study
- Base Year: 2023
- Historical Period: 2018-2023
- Market Forecast: 2024-2034
Countries Covered
- United States
- Germany
- France
- United Kingdom
- Italy
- Spain
- Japan
Analysis Covered Across Each Country
- Historical, current, and future epidemiology scenario
- Historical, current, and future performance of the epilepsy market
- Historical, current, and future performance of various therapeutic categories in the market
- Sales of various drugs across the epilepsy market
Competitive Landscape:
This report also provides a detailed analysis of the current epilepsy marketed drugs and late-stage pipeline drugs.In-Market Drugs
- Drug Overview
- Mechanism of Action
- Regulatory Status
- Clinical Trial Results
- Drug Uptake and Market Performance
Late-Stage Pipeline Drugs
- Drug Overview
- Mechanism of Action
- Regulatory Status
- Clinical Trial Results
- Drug Uptake and Market Performance
Key Questions Answered in this Report:
Market Insights
- How has the epilepsy market performed so far and how will it perform in the coming years?
- What are the markets shares of various therapeutic segments in 2023 and how are they expected to perform till 2034?
- What was the country-wise size of the epilepsy market across the seven major markets in 2023 and what will it look like in 2034?
- What is the growth rate of the epilepsy market across the seven major markets and what will be the expected growth over the next ten years?
- What are the key unmet needs in the market?
Epidemiology Insights
- What is the number of prevalent cases (2018-2034) of epilepsy across the seven major markets?
- What is the number of prevalent cases (2018-2034) of epilepsy by age across the seven major markets?
- What is the number of prevalent cases (2018-2034) of epilepsy by gender across the seven major markets?
- What is the number of prevalent cases (2018-2034) of epilepsy by type across the seven major markets?
- How many patients are diagnosed (2018-2034) with epilepsy across the seven major markets?
- What is the size of the epilepsy patient pool (2018-2023) across the seven major markets?
- What would be the forecasted patient pool (2024-2034) across the seven major markets?
- What are the key factors driving the epidemiological trend of epilepsy?
- What will be the growth rate of patients across the seven major markets?
Epilepsy: Current Treatment Scenario, Marketed Drugs and Emerging Therapies
- What are the current marketed drugs and what are their market performance?
- What are the key pipeline drugs and how are they expected to perform in the coming years?
- How safe are the current marketed drugs and what are their efficacies?
- How safe are the late-stage pipeline drugs and what are their efficacies?
- What are the current treatment guidelines for epilepsy drugs across the seven major markets?
- Who are the key companies in the market and what are their market shares?
- What are the key mergers and acquisitions, licensing activities, collaborations, etc. related to the epilepsy market?
- What are the key regulatory events related to the epilepsy market?
- What is the structure of clinical trial landscape by status related to the epilepsy market?
- What is the structure of clinical trial landscape by phase related to the epilepsy market?
- What is the structure of clinical trial landscape by route of administration related to the epilepsy market?
This product will be updated with the latest data at the time of order. Consequently, dispatch time for this product will be 7-10 business days.
Table of Contents
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 140 |
| Published | May 2024 |
| Forecast Period | 2023 - 2034 |
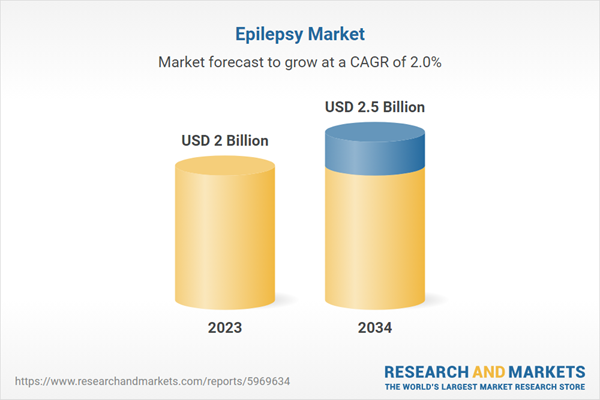
| Estimated Market Value ( USD | $ 2 Billion |
| Forecasted Market Value ( USD | $ 2.5 Billion |
| Compound Annual Growth Rate | 2.0% |
| Regions Covered | Global |