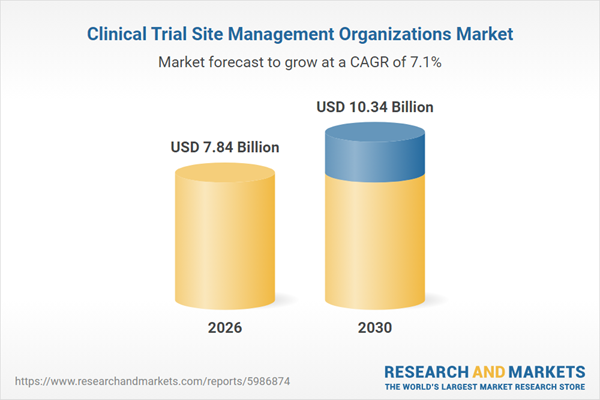
The clinical trial site management organizations market size is expected to see strong growth in the next few years. It will grow to $10.34 billion in 2030 at a compound annual growth rate (CAGR) of 7.1%. The growth in the forecast period can be attributed to increasing adoption of decentralized site operations, rising investments in site digitization tools, growing demand for faster site activation, expansion of complex therapeutic trials, increasing emphasis on data quality and compliance. Major trends in the forecast period include increasing demand for specialized site management support, rising focus on patient recruitment and retention services, growing adoption of centralized site coordination models, expansion of regulatory compliance support services, enhanced use of digital site management tools.
The rising demand for drug discovery is expected to drive the growth of the clinical trial site management organizations market in the coming years. Drug discovery involves the identification and development of new medicines or therapeutic compounds to treat diseases and improve health outcomes. The growing focus on drug discovery is driven by unmet medical needs, emerging diseases, increasing drug resistance, and an aging population, all of which are fueling continuous innovation in pharmaceutical research and development. Clinical trial site management organizations support drug discovery by improving coordination and oversight of clinical trials, ensuring regulatory compliance, enabling efficient patient recruitment, and supporting accurate data collection. For instance, in January 2024, according to Health Economists, a UK-based pharmaceutical and life sciences company, 69 new active substances were approved globally in 2023, representing an increase from the previous year, including 24 first-in-class drugs approved in the United States. This trend highlights the expanding pipeline of innovative therapies and supports market growth.
Companies operating in the clinical trial site management organizations market are increasingly focusing on integrating artificial intelligence into virtual trial platforms to improve recruitment, patient engagement, and overall trial efficiency. AI-enabled clinical trial recruitment and engagement platforms use advanced algorithms to optimize participant outreach, streamline enrollment processes, and enhance engagement throughout the trial lifecycle. For example, in October 2023, Elligo Health Research, a US-based healthcare-enabling research organization, partnered with Avallano, a US-based clinical research platform provider, to launch myTrialsConnect, an AI-driven clinical trial recruitment and engagement platform. The platform enables patients to enroll both as trial participants and healthcare network members and receive notifications when they qualify for relevant clinical trials through automated medical record screening and chatbot-based surveys.
In January 2023, Novotech, an Australia-based provider of clinical development, project management, and site management organization services, acquired EastHoRN Clinical Services for an undisclosed amount. This acquisition allows Novotech to expand its global presence and provide biotech clients with broader access to its comprehensive contract research organization services across Europe, the United States, and the Asia-Pacific region. EastHoRN Clinical Services is a Poland-based clinical research organization specializing in clinical trial management, regulatory affairs support, data management, and biostatistics.
Major companies operating in the clinical trial site management organizations market are IQVIA Holdings Inc., ICON plc, PPD Inc., SGS SA, Syneos Health Inc., Charles River Laboratories International Inc., PAREXEL International Corporation, Medpace Holdings Inc, Premier Research Group Limited, WIRB-Copernicus Group, Worldwide Clinical Trials Inc., ClinChoice Inc., Clinigen Group plc, Precision for Medicine Inc., Synexus Clinical Research, Celerion Inc., Sarah Cannon Research Institute, Clinipace Worldwide Inc., Ergomed plc, Rho Federal Systems Division Inc., Veristat LLC, CTI Clinical Trial & Consulting Services, Clinedge, FOMAT Medical Research INC., Grand Pacific CRO, Xylem CRO, Access Clinical Research LLC, KV Consulting Corporation, Aurum Clinical Research, EthosExcel Inc.
Asia-Pacific was the largest region in the clinical trial site management organizations market in 2025. The regions covered in the clinical trial site management organizations market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in the clinical trial site management organizations market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.
Tariffs are influencing the clinical trial site management organizations market by increasing costs of imported IT systems, monitoring equipment, data capture devices, and site-level infrastructure technologies. Trial sites in North America and Europe are most affected due to reliance on imported digital tools, while Asia-Pacific faces cost pressure on scaling site management operations. These tariffs are increasing operational expenses for site management organizations and influencing service pricing. However, they are also encouraging local sourcing of site technologies, regional service partnerships, and greater adoption of cloud-based and software-driven site management solutions.
The clinical trial site management organizations market research report is one of a series of new reports that provides clinical trial site management organizations market statistics, including clinical trial site management organizations industry global market size, regional shares, competitors with a clinical trial site management organizations market share, detailed clinical trial site management organizations market segments, market trends and opportunities, and any further data you may need to thrive in the clinical trial site management organizations industry. This clinical trial site management organizations market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future scenario of the industry.
Clinical trial site management organizations are specialized entities that provide dedicated support and services to clinical trial sites. These organizations play an essential role in ensuring the efficient and effective execution of clinical trials by assisting sites with various operational, regulatory, and administrative aspects of study management.
The primary service types offered by clinical trial site management organizations include site management, project management, regulatory monitoring, and on-site monitoring. Site management involves the coordination and oversight of clinical trial sites to ensure smooth day-to-day operations and strict adherence to study protocols throughout the trial lifecycle. These services are utilized across all clinical trial phases, including Phase I, Phase II, Phase III, and Phase IV, and support therapeutic areas such as oncology, cardiology, central nervous system disorders, pain management, endocrine conditions, and others.
The clinical trial site management organizations market consist of revenues earned by entities by providing services such as patient recruitment and retention, clinical data management, quality assurance and monitoring, financial management, training, and education. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified).
The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.
This product will be delivered within 1-3 business days.
Table of Contents
Executive Summary
Clinical Trial Site Management Organizations Market Global Report 2026 provides strategists, marketers and senior management with the critical information they need to assess the market.This report focuses clinical trial site management organizations market which is experiencing strong growth. The report gives a guide to the trends which will be shaping the market over the next ten years and beyond.
Reasons to Purchase:
- Gain a truly global perspective with the most comprehensive report available on this market covering 16 geographies.
- Assess the impact of key macro factors such as geopolitical conflicts, trade policies and tariffs, inflation and interest rate fluctuations, and evolving regulatory landscapes.
- Create regional and country strategies on the basis of local data and analysis.
- Identify growth segments for investment.
- Outperform competitors using forecast data and the drivers and trends shaping the market.
- Understand customers based on end user analysis.
- Benchmark performance against key competitors based on market share, innovation, and brand strength.
- Evaluate the total addressable market (TAM) and market attractiveness scoring to measure market potential.
- Suitable for supporting your internal and external presentations with reliable high-quality data and analysis
- Report will be updated with the latest data and delivered to you along with an Excel data sheet for easy data extraction and analysis.
- All data from the report will also be delivered in an excel dashboard format.
Description
Where is the largest and fastest growing market for clinical trial site management organizations? How does the market relate to the overall economy, demography and other similar markets? What forces will shape the market going forward, including technological disruption, regulatory shifts, and changing consumer preferences? The clinical trial site management organizations market global report answers all these questions and many more.The report covers market characteristics, size and growth, segmentation, regional and country breakdowns, total addressable market (TAM), market attractiveness score (MAS), competitive landscape, market shares, company scoring matrix, trends and strategies for this market. It traces the market’s historic and forecast market growth by geography.
- The market characteristics section of the report defines and explains the market. This section also examines key products and services offered in the market, evaluates brand-level differentiation, compares product features, and highlights major innovation and product development trends.
- The supply chain analysis section provides an overview of the entire value chain, including key raw materials, resources, and supplier analysis. It also provides a list competitor at each level of the supply chain.
- The updated trends and strategies section analyses the shape of the market as it evolves and highlights emerging technology trends such as digital transformation, automation, sustainability initiatives, and AI-driven innovation. It suggests how companies can leverage these advancements to strengthen their market position and achieve competitive differentiation.
- The regulatory and investment landscape section provides an overview of the key regulatory frameworks, regularity bodies, associations, and government policies influencing the market. It also examines major investment flows, incentives, and funding trends shaping industry growth and innovation.
- The market size section gives the market size ($b) covering both the historic growth of the market, and forecasting its development.
- The forecasts are made after considering the major factors currently impacting the market. These include the technological advancements such as AI and automation, Russia-Ukraine war, trade tariffs (government-imposed import/export duties), elevated inflation and interest rates.
- The total addressable market (TAM) analysis section defines and estimates the market potential compares it with the current market size, and provides strategic insights and growth opportunities based on this evaluation.
- The market attractiveness scoring section evaluates the market based on a quantitative scoring framework that considers growth potential, competitive dynamics, strategic fit, and risk profile. It also provides interpretive insights and strategic implications for decision-makers.
- Market segmentations break down the market into sub markets.
- The regional and country breakdowns section gives an analysis of the market in each geography and the size of the market by geography and compares their historic and forecast growth.
- Expanded geographical coverage includes Taiwan and Southeast Asia, reflecting recent supply chain realignments and manufacturing shifts in the region. This section analyzes how these markets are becoming increasingly important hubs in the global value chain.
- The competitive landscape chapter gives a description of the competitive nature of the market, market shares, and a description of the leading companies. Key financial deals which have shaped the market in recent years are identified.
- The company scoring matrix section evaluates and ranks leading companies based on a multi-parameter framework that includes market share or revenues, product innovation, and brand recognition.
Report Scope
Markets Covered:
1) By Services: Site Management; Project Management; Regulatory; Onsite Monitoring2) By Phase: Phase I; Phase II; Phase III; Phase IV
3) By Therapeutic Area: Oncology; Cardiology; Central Nervous System (CNS); Pain Management; Endocrine; Other Therapeutic Areas
Subsegments:
1) By Site Management: Site Selection And Feasibility; Patient Recruitment And Retention; Site Coordination And Administration2) By Project Management: Project Planning And Scheduling; Budget And Resource Management; Risk Management And Compliance
3) By Regulatory: Regulatory Submissions And Approvals; Ethics And Institutional Review Board (IRB) Submissions; Regulatory Compliance And Audits
4) By Onsite Monitoring: Site Visits And Inspections; Data Quality Monitoring; Safety And Protocol Compliance Monitoring
Companies Mentioned: IQVIA Holdings Inc.; ICON plc; PPD Inc.; SGS SA; Syneos Health Inc.; Charles River Laboratories International Inc.; PAREXEL International Corporation; Medpace Holdings Inc; Premier Research Group Limited; WIRB-Copernicus Group; Worldwide Clinical Trials Inc.; ClinChoice Inc.; Clinigen Group plc; Precision for Medicine Inc.; Synexus Clinical Research; Celerion Inc.; Sarah Cannon Research Institute; Clinipace Worldwide Inc.; Ergomed plc; Rho Federal Systems Division Inc.; Veristat LLC; CTI Clinical Trial & Consulting Services; Clinedge; FOMAT Medical Research INC.; Grand Pacific CRO; Xylem CRO; Access Clinical Research LLC; KV Consulting Corporation; Aurum Clinical Research; EthosExcel Inc.
Countries: Australia; Brazil; China; France; Germany; India; Indonesia; Japan; Taiwan; Russia; South Korea; UK; USA; Canada; Italy; Spain.
Regions: Asia-Pacific; South East Asia; Western Europe; Eastern Europe; North America; South America; Middle East; Africa
Time Series: Five years historic and ten years forecast.
Data: Ratios of market size and growth to related markets, GDP proportions, expenditure per capita.
Data Segmentation: Country and regional historic and forecast data, market share of competitors, market segments.
Sourcing and Referencing: Data and analysis throughout the report is sourced using end notes.
Delivery Format: Word, PDF or Interactive Report + Excel Dashboard
Added Benefits:
- Bi-Annual Data Update
- Customisation
- Expert Consultant Support
Companies Mentioned
The companies featured in this Clinical Trial Site Management Organizations market report include:- IQVIA Holdings Inc.
- ICON plc
- PPD Inc.
- SGS SA
- Syneos Health Inc.
- Charles River Laboratories International Inc.
- PAREXEL International Corporation
- Medpace Holdings Inc
- Premier Research Group Limited
- WIRB-Copernicus Group
- Worldwide Clinical Trials Inc.
- ClinChoice Inc.
- Clinigen Group plc
- Precision for Medicine Inc.
- Synexus Clinical Research
- Celerion Inc.
- Sarah Cannon Research Institute
- Clinipace Worldwide Inc.
- Ergomed plc
- Rho Federal Systems Division Inc.
- Veristat LLC
- CTI Clinical Trial & Consulting Services
- Clinedge
- FOMAT Medical Research INC.
- Grand Pacific CRO
- Xylem CRO
- Access Clinical Research LLC
- KV Consulting Corporation
- Aurum Clinical Research
- EthosExcel Inc.
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 250 |
| Published | February 2026 |
| Forecast Period | 2026 - 2030 |
| Estimated Market Value ( USD | $ 7.84 Billion |
| Forecasted Market Value ( USD | $ 10.34 Billion |
| Compound Annual Growth Rate | 7.1% |
| Regions Covered | Global |
| No. of Companies Mentioned | 31 |