Global No-Code AI Platform Market - Key Trends & Drivers Summarized
Is Artificial Intelligence Finally Breaking the Code Barrier?
No-code AI platforms are redefining the artificial intelligence landscape by enabling business users, analysts, and domain experts to build, deploy, and iterate AI models without writing a single line of code. These platforms offer intuitive drag-and-drop interfaces, automated data preprocessing tools, and pre-configured machine learning pipelines that democratize AI capabilities across non-technical roles. By lowering the entry barrier to AI development, no-code platforms are enabling departments such as marketing, finance, HR, logistics, and customer service to independently deploy predictive models for demand forecasting, churn analysis, fraud detection, and more. This shift is decentralizing AI innovation within enterprises and accelerating time-to-value for AI-driven initiatives.Enterprise adoption of no-code AI tools is being driven by the operational need for real-time, low-latency decisions without overburdening centralized data science teams. Organizations are increasingly empowering business units with tools to explore data, run experiments, and build customized solutions for local problems without long development cycles or dependency on scarce AI engineers. Leading platforms are embedding features such as automated model explainability, visual model comparison dashboards, and compliance-aware model deployment options, which are particularly useful in regulated industries like banking, healthcare, and insurance. These features ensure that while AI model development is simplified, governance and accuracy are not compromised.
What's Fueling the Enterprise Shift Toward Low-Code and No-Code AI Ecosystems?
The increasing demand for agile data strategies and rapid experimentation has made traditional coding-centric AI pipelines less feasible for many business environments. Companies need to move from proof-of-concept to production in weeks, not months. No-code AI platforms cater to this need by offering ready-to-use templates for classification, regression, clustering, and natural language processing models. These platforms are also integrating with enterprise data warehouses, CRM systems, and cloud-based storage platforms, allowing users to work with live data pipelines in real time. This seamless integration is allowing teams to operationalize machine learning without dealing with infrastructure complexities or versioning headaches.Moreover, as edge computing and embedded AI gain momentum, no-code platforms are being optimized for model deployment across devices ranging from mobile apps to IoT sensors. Edge-optimized no-code tools are enabling field technicians, retail managers, or factory supervisors to interact with localized AI models through web-based dashboards or mobile apps. The ability to build models that are responsive to local data while remaining integrated with central systems is giving rise to a hybrid AI deployment model, blending cloud scalability with edge precision. This trend is particularly visible in industries like retail, telecom, and manufacturing, where decision-making at the point of interaction is critical.
How Are Advancements in AutoML and Embedded AI Powering No-Code Platforms?
No-code AI platforms are evolving rapidly due to the parallel advancements in Automated Machine Learning (AutoML), neural architecture search, and explainable AI frameworks. AutoML, which automates the selection of algorithms, hyperparameter tuning, and model validation, forms the backbone of many no-code platforms. These capabilities allow users to generate high-performing models by simply selecting the problem type and uploading relevant datasets. Platforms are now also incorporating model drift detection, fairness evaluation metrics, and real-time retraining triggers all without requiring programming input. Such features are enabling continuous improvement of AI models in dynamic environments like e-commerce, fintech, and logistics.Integration of natural language processing (NLP) and computer vision models into no-code frameworks is expanding their reach across industries. Businesses can now deploy image recognition tools for defect detection, text classification models for sentiment analysis, or OCR models for document processing using simple UI-based workflows. Moreover, platforms are increasingly embedding explainable AI (XAI) modules, which help users interpret how a model arrived at a particular decision using visualizations like SHAP values or LIME plots. These explainability features are essential for ensuring transparency and trust in AI-driven decisions especially in healthcare diagnostics, insurance underwriting, and loan processing workflows.
What's Propelling the Breakneck Growth of the No-Code AI Platform Market?
The growth in the no-code AI platform market is driven by several factors that reflect the convergence of workforce transformation, digital acceleration, and data strategy modernization. A major growth driver is the acute shortage of skilled data scientists and machine learning engineers. As AI adoption becomes imperative across verticals, enterprises are looking for scalable ways to equip their workforce with tools that require minimal technical training. No-code AI platforms fill this gap by allowing business analysts, product managers, and operations teams to independently develop AI solutions tailored to their specific use cases. This shift is reducing backlogs for IT and data science teams while promoting a culture of innovation across organizations.Another key driver is the proliferation of data across organizational silos. With massive volumes of structured and unstructured data being generated from enterprise apps, IoT devices, and customer touchpoints, there is an urgent need for tools that enable rapid, contextual analysis without requiring data engineering expertise. No-code platforms, with their automated ETL, feature engineering, and model deployment capabilities, are emerging as the default solution for turning raw data into actionable insights at scale. Additionally, the rise of cloud-native infrastructure, API-first platforms, and SaaS-based delivery models is making these tools accessible to SMEs and startups, not just large enterprises.
The evolving regulatory landscape is further amplifying demand for no-code AI solutions that offer compliance-aware features, audit trails, and explainability-by-design. As industries face increasing pressure to justify algorithmic decisions and avoid bias-related risks, platforms that integrate responsible AI capabilities out-of-the-box are seeing heightened traction. With robust vendor ecosystems, expanding third-party integrations, and increasingly domain-specific templates being added every quarter, the global no-code AI market is poised for sustained hypergrowth transforming how businesses harness the power of machine learning in the years ahead.
Key Insights:
- Market Growth: Understand the significant growth trajectory of the Platform segment, which is expected to reach US$10.3 Billion by 2030 with a CAGR of a 20.9%. The Services segment is also set to grow at 28.8% CAGR over the analysis period.
- Regional Analysis: Gain insights into the U.S. market, valued at $1.3 Billion in 2024, and China, forecasted to grow at an impressive 22.7% CAGR to reach $2.7 Billion by 2030. Discover growth trends in other key regions, including Japan, Canada, Germany, and the Asia-Pacific.
Why You Should Buy This Report:
- Detailed Market Analysis: Access a thorough analysis of the Global No-Code AI Platform Market, covering all major geographic regions and market segments.
- Competitive Insights: Get an overview of the competitive landscape, including the market presence of major players across different geographies.
- Future Trends and Drivers: Understand the key trends and drivers shaping the future of the Global No-Code AI Platform Market.
- Actionable Insights: Benefit from actionable insights that can help you identify new revenue opportunities and make strategic business decisions.
Key Questions Answered:
- How is the Global No-Code AI Platform Market expected to evolve by 2030?
- What are the main drivers and restraints affecting the market?
- Which market segments will grow the most over the forecast period?
- How will market shares for different regions and segments change by 2030?
- Who are the leading players in the market, and what are their prospects?
Report Features:
- Comprehensive Market Data: Independent analysis of annual sales and market forecasts in US$ Million from 2024 to 2030.
- In-Depth Regional Analysis: Detailed insights into key markets, including the U.S., China, Japan, Canada, Europe, Asia-Pacific, Latin America, Middle East, and Africa.
- Company Profiles: Coverage of players such as Akkio Inc., Amazon Web Services, Inc., Apple Inc., Caspio, Inc., and more.
- Complimentary Updates: Receive free report updates for one year to keep you informed of the latest market developments.
Some of the 37 companies featured in this No-Code AI Platform market report include:
- Akkio Inc.
- Amazon Web Services, Inc.
- Apple Inc.
- Caspio, Inc.
- Clarifai, Inc.
- DataRobot, Inc.
- Google LLC
- Graphite Note
- H2O.ai
- IBM Corporation
- Levity AI GmbH
- Microsoft Corporation
- MonkeyLearn
- Noogata
- Pecan AI
- Quickbase, Inc.
- Runway ML, Inc.
- Salesforce, Inc.
- SymphonyAI
- Yellow.ai
This edition integrates the latest global trade and economic shifts as of June 2025 into comprehensive market analysis. Key updates include:
- Tariff and Trade Impact: Insights into global tariff negotiations across 180+ countries, with analysis of supply chain turbulence, sourcing disruptions, and geographic realignment. Special focus on 2025 as a pivotal year for trade tensions, including updated perspectives on the Trump-era tariffs.
- Adjusted Forecasts and Analytics: Revised global and regional market forecasts through 2030, incorporating tariff effects, economic uncertainty, and structural changes in globalization. Includes segmentation by product, technology, type, material, distribution channel, application, and end-use, with historical analysis since 2015.
- Strategic Market Dynamics: Evaluation of revised market prospects, regional outlooks, and key economic indicators such as population and urbanization trends.
- Innovation & Technology Trends: Latest developments in product and process innovation, emerging technologies, and key industry drivers shaping the competitive landscape.
- Competitive Intelligence: Updated global market share estimates for 2025, competitive positioning of major players (Strong/Active/Niche/Trivial), and refined focus on leading global brands and core players.
- Expert Insight & Commentary: Strategic analysis from economists, trade experts, and domain specialists to contextualize market shifts and identify emerging opportunities.
- Complimentary Update: Buyers receive a free July 2025 update with finalized tariff impacts, new trade agreement effects, revised projections, and expanded country-level coverage.
Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Akkio Inc.
- Amazon Web Services, Inc.
- Apple Inc.
- Caspio, Inc.
- Clarifai, Inc.
- DataRobot, Inc.
- Google LLC
- Graphite Note
- H2O.ai
- IBM Corporation
- Levity AI GmbH
- Microsoft Corporation
- MonkeyLearn
- Noogata
- Pecan AI
- Quickbase, Inc.
- Runway ML, Inc.
- Salesforce, Inc.
- SymphonyAI
- Yellow.ai
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 237 |
| Published | February 2026 |
| Forecast Period | 2024 - 2030 |
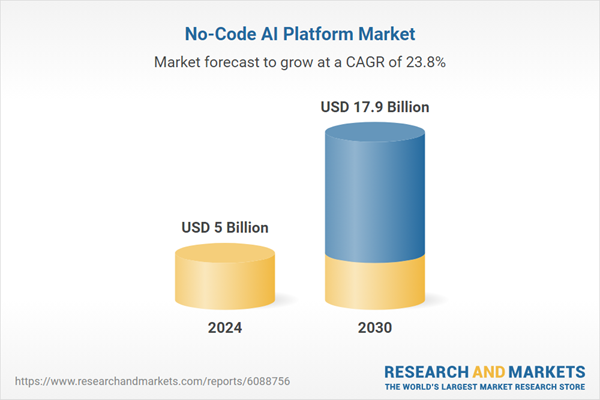
| Estimated Market Value ( USD | $ 5 Billion |
| Forecasted Market Value ( USD | $ 17.9 Billion |
| Compound Annual Growth Rate | 23.8% |
| Regions Covered | Global |