Global Artificial Intelligence (AI) / Machine Learning in Medical Devices Market - Where Smart Systems Redefine Clinical Precision and Future Healthcare Possibilities
How Is AI Transforming Device Intelligence Beyond Traditional Mechanical Functionality?
Artificial intelligence and machine learning are evolving from experimental features into foundational pillars within modern medical devices, enabling a shift from static programmed systems to responsive platforms capable of contextual reasoning and adaptive functionality. AI powered imaging devices can learn from previous scans and refine interpretation accuracy over time, while intelligent monitoring platforms detect subtle physiological deviations that would otherwise remain unnoticed. The integration of speech recognition, natural language processing, and contextual understanding into smart devices is reshaping clinical workflows, reducing manual inputs, and improving response time in high acuity environments. This level of augmentation enables devices to play a meaningful role in screening, triage, diagnosis, and planning rather than serving purely mechanical or measurement based functions.The evolution toward autonomous decision support is especially evident in diagnostic imaging, cardiology, laboratory medicine, and connected monitoring solutions, where algorithmic reasoning adds clinical depth to data interpretation. Deep learning enables detection of complex patterns in radiological scans, arrhythmia traces, and hematological profiles with a growing level of consistency. In connected care settings, AI enhanced devices continuously learn from real world datasets, improving their predictive ability, calibration logic, and feedback modeling. As a result, the boundary between device functionality and clinical reasoning is gradually narrowing, positioning AI enabled medical devices as core enablers of predictive care, precision diagnostics, and personalized interventions. The pace of adoption indicates a long term trajectory where AI becomes a standard expectation rather than a premium differentiator across most categories of medical technology.
What Makes the AI Enabled Device Ecosystem Expand Across Hospitals, Remote Care, and Home Health Environments?
The expanding infrastructure surrounding digital health ecosystems is accelerating deployment of AI driven medical devices across diverse care environments. Electronic health record integration, cloud based processing, and real time data synchronization allow devices to operate within interconnected clinical frameworks rather than as isolated equipment categories. Wearable and remote monitoring platforms using machine learning have become pivotal in supporting chronic disease management, post operative follow up, and decentralized care models. AI assists these devices in interpreting continuous physiological inputs, identifying risk inflection points, and initiating alert based workflows without requiring manual oversight. This creates a feedback loop between home based monitoring and clinical intervention, enabling earlier engagement and reducing avoidable complications.Hospitals and diagnostic centers benefit from efficiency gains derived from automation in imaging review, case prioritization, and structured reporting. Predictive modeling embedded into monitoring systems enhances decision accuracy in intensive care and emergency environments, supporting earlier escalation and resource allocation. On the other hand, AI powered software as a medical device solutions operate independently of proprietary hardware, facilitating cross platform deployment and algorithmic scalability. The convergence of embedded intelligence within hardware systems and cloud enabled decision support software is shaping a hybrid model where AI sits at the center of monitoring, diagnostics, and automated therapeutics across institutional and home settings. This dual format adoption signals a shift toward a distributed health technology ecosystem rather than device centric product silos.
How Do Regulation, Model Accountability, and Lifecycle Governance Influence Industry Momentum?
The introduction of adaptive and learning models into medical devices has prompted significant regulatory evolution as oversight frameworks transition from static product certification to lifecycle governance models. Regulatory bodies increasingly require transparency, traceability, explainability, and continuous validation for algorithms deployed in clinical environments. This includes demonstrating model stability, reproducibility, and risk mitigation across demographic populations, input variance, and changing clinical conditions. Lifecycle monitoring frameworks now emphasize real world evidence collection, algorithm drift detection, remote configuration controls, and structured update pathways. This reinforces the requirement that AI driven medical devices remain accountable as their behavior evolves beyond the state in which they were initially assessed.Vendors are also navigating integration challenges related to interoperability, cybersecure data exchanges, and bias mitigation. As devices become increasingly connected, cybersecurity architecture gains prominence, especially for implantables, autonomous monitoring systems, and cloud dependent remote diagnostics. Meanwhile, ethical considerations extend beyond fairness and bias toward responsible deployment and clinical reliance thresholds. These regulatory and ethical dimensions influence vendor development strategies, procurement priorities, and ecosystem partnerships. The result is a competitive environment where successful market participation is linked not only to technological capability but to the strength of compliance infrastructure, post market surveillance strategy, and transparent operational governance.
The Growth in the AI / Machine Learning in Medical Devices Market Is Driven by Several Factors…
The most influential growth catalysts include increased demand for precision diagnostics, expansion of remote and decentralized care models, and rising dependence on automation in clinical workflows experiencing workforce constraints. Adoption accelerates as AI powered devices improve analytical throughput in high volume environments such as radiology and cardiology, enabling faster case prioritization and reducing interpretation fatigue. Software as a medical device solutions grow rapidly as healthcare organizations seek scalable, cloud deployable intelligence without requiring large hardware capital expenditure. Growth is further stimulated by advancements in wearable and implantable sensing platforms where machine learning enhances personalization, supports continuous monitoring, and strengthens long term disease management strategies.Shifts in consumer behavior also accelerate adoption, particularly as remote health monitoring, wellness tracking, and preventive diagnostics gain mainstream relevance. The integration of multimodal datasets such as genomic, metabolic, clinical, and behavioral signals strengthens predictive modeling, enabling more sophisticated risk scoring and adaptive therapy pathways. The rise of interoperable digital health infrastructures and increasing maturity of federated learning, edge computing, natural language interaction layers, and multimodal AI models continue to expand the market addressable scope. As these forces converge, the trajectory of intelligent medical devices points toward deeper autonomy, broader accessibility, and greater influence on global care delivery systems, signaling a transformation in how healthcare intelligence is created, distributed, and operationalized.
Report Scope
The report analyzes the AI / ML in Medical Devices market, presented in terms of market value (US$). The analysis covers the key segments and geographic regions outlined below:- Segments: Product Type (System / Hardware, Software-As-A Medical Device); Clinical Area (Radiology, Cardiology, Hematology, Other Clinical Areas)
- Geographic Regions/Countries: World; United States; Canada; Japan; China; Europe (France; Germany; Italy; United Kingdom; and Rest of Europe); Asia-Pacific; Rest of World.
Key Insights:
- Market Growth: Understand the significant growth trajectory of the System / Hardware segment, which is expected to reach US$11.2 Billion by 2032 with a CAGR of a 18.3%. The Software-As-A Medical Device segment is also set to grow at 22.8% CAGR over the analysis period.
- Regional Analysis: Gain insights into the U.S. market, valued at $1.5 Billion in 2025, and China, forecasted to grow at an impressive 19.0% CAGR to reach $3.1 Billion by 2032. Discover growth trends in other key regions, including Japan, Canada, Germany, and the Asia-Pacific.
Why You Should Buy This Report:
- Detailed Market Analysis: Access a thorough analysis of the Global AI / ML in Medical Devices Market, covering all major geographic regions and market segments.
- Competitive Insights: Get an overview of the competitive landscape, including the market presence of major players across different geographies.
- Future Trends and Drivers: Understand the key trends and drivers shaping the future of the Global AI / ML in Medical Devices Market.
- Actionable Insights: Benefit from actionable insights that can help you identify new revenue opportunities and make strategic business decisions.
Key Questions Answered:
- How is the Global AI / ML in Medical Devices Market expected to evolve by 2032?
- What are the main drivers and restraints affecting the market?
- Which market segments will grow the most over the forecast period?
- How will market shares for different regions and segments change by 2032?
- Who are the leading players in the market, and what are their prospects?
Report Features:
- Comprehensive Market Data: Independent analysis of annual sales and market forecasts in US$ Million from 2025 to 2032.
- In-Depth Regional Analysis: Detailed insights into key markets, including the U.S., China, Japan, Canada, Europe, Asia-Pacific, Latin America, Middle East, and Africa.
- Company Profiles: Coverage of players such as EDAN Instruments, Inc., and more.
- Complimentary Updates: Receive free report updates for one year to keep you informed of the latest market developments.
Some of the companies featured in this AI / ML in Medical Devices market report include:
- EDAN Instruments, Inc.
Domain Expert Insights
This market report incorporates insights from domain experts across enterprise, industry, academia, and government sectors. These insights are consolidated from multilingual multimedia sources, including text, voice, and image-based content, to provide comprehensive market intelligence and strategic perspectives. As part of this research study, the publisher tracks and analyzes insights from 43 domain experts. Clients may request access to the network of experts monitored for this report, along with the online expert insights tracker.Table of Contents
Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- EDAN Instruments, Inc.
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 134 |
| Published | May 2026 |
| Forecast Period | 2025 - 2032 |
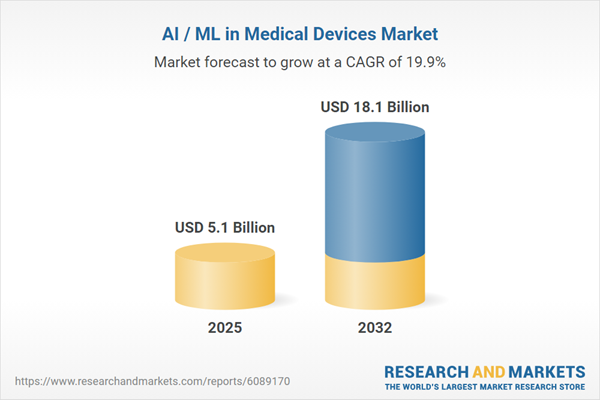
| Estimated Market Value ( USD | $ 5.1 Billion |
| Forecasted Market Value ( USD | $ 18.1 Billion |
| Compound Annual Growth Rate | 19.9% |
| Regions Covered | Global |