Cancer Drugs Market Key Insights
- Precision and biomarker stewardship now decide market share: Growth concentrates in agents tethered to validated biomarkers, with therapy selection guided by comprehensive genomic profiling, PD-L1 or tumor mutational burden measures, and MRD dynamics; winners equip providers with testing access, turnaround, and interpretation tools while designing trials that secure label breadth across lines and histologies
- Combination architecture is the new product strategy: Checkpoints, targeted inhibitors, and ADCs are increasingly developed as modular backbones with rational add-ons to overcome resistance, deepen responses, and move earlier in care; success depends on mechanistic complementarity, staggered dosing to manage overlapping toxicities, and adaptive trial designs that rapidly cull non-additive pairs
- ADCs and bispecifics rewire the efficacy-tolerability frontier: Smarter linkers, stable payloads, and tumor-selective targets enable high drug delivery to cancer cells while limiting off-tumor toxicity; bispecifics recruit immune effector function or dual-block signaling, but require proactive safety operations to manage cytokine and ocular or hematologic risks without disrupting dose intensity
- Cell and gene therapies push beyond hematology: Autologous platforms refine manufacturing and turnaround, while allogeneic and in vivo approaches seek scalability; expansion into solid tumors hinges on trafficking, microenvironment modulation, and safety switches, with integrated apheresis logistics, vein-to-vein analytics, and regional manufacturing footprints becoming competitive moats
- Access is shaped by value frameworks and real-world evidence: Payers prioritize overall survival, progression-free survival, quality-adjusted life years, and hospital utilization; programs that pair pivotal data with pragmatic outcomes, PROs, and resource use analytics enable indication-based pricing, outcomes contracts, and earlier pathway inclusion
- Toxicity management is commercial strategy, not just medicine: Clear AE playbooks, prophylaxis kits, dose-modification algorithms, and remote monitoring reduce discontinuations; vendors that train infusion centers, provide nurse hotlines, and integrate alerts into EHRs protect dose intensity, improve experience, and differentiate in competitive classes
- Manufacturing agility underwrites launch and growth: For biologics and cell therapies, yield, comparability, and cold-chain reliability are decisive; dual-site drug substance, redundant fill-finish, and real-time release analytics mitigate shortages and support rapid indication expansions and geographic rollouts
- Diagnostics-drug-data ecosystems compress time to treat: Partnerships with labs and hospital networks standardize sample flows, payer pre-auth, and result integration; embedding test orders in oncology pathways and funding reflex testing improves identification rates, expands eligible populations, and sustains market penetration
- Digital adherence and care orchestration drive outcomes: Apps, ePRO capture, and remote vitals flag AEs, guide symptom management, and schedule labs; adherence nudges and pharmacy coordination reduce dose gaps, while analytics inform next-best-action for clinicians and case managers, improving both clinical and economic performance
- Policy and affordability pressures require proactive design: Tiered pricing, patient support, and compassionate use balance access with sustainability; transparent evidence plans, post-marketing commitments, and participation in generics/biosimilars transition planning preserve trust and reduce volatility at loss of exclusivity
Cancer Drugs Market Reginal Analysis
North America
High adoption of genomic profiling, strong clinical trial infrastructure, and broad access to innovative regimens drive early uptake; payer scrutiny channels use through step edits, prior authorization, and outcomes contracts, while integrated delivery networks emphasize pathway adherence, toxicity stewardship, and specialty-pharmacy coordination to manage total cost of care
Europe
Centralized health technology assessments and price-value negotiations shape launch cadence and indication breadth; national tumor boards, guideline harmonization, and robust real-world registries support evidence-based adoption, with increasing preference for combination strategies that demonstrate clear incremental benefit and manageable budget impact
Asia-Pacific
Rapid capacity growth in oncology centers, expanding reimbursement, and domestic innovation pipelines accelerate access; large incident populations enable swift enrollment and real-world data generation, while variability in testing infrastructure and funding drives tiered portfolios, local partnerships, and patient-assistance models to reduce time from diagnosis to therapy start
Middle East & Africa
Centres of excellence in major cities expand access to advanced biologics and targeted therapies, supported by government procurement and private insurance; constraints in molecular testing and infusion capacity spur hub-and-spoke models, with emphasis on guideline adoption, pharmacist-led adherence programs, and reliable cold-chain for temperature-sensitive products
South & Central America
Public-private mix influences uptake, with leading cancer institutes piloting novel regimens and broader systems adopting as budget permits; access strategies combine named-patient use, tiered pricing, and diagnostic partnerships, while supply continuity, clinician education, and navigation services are prioritized to improve adherence and reduce treatment discontinuities
Cancer Drugs Market Segmentation
By Type- Chemotherapy
- Targeted Therapy
- Immunotherapy
- Hormonal Therapy
- Others
- Blood Cancer
- Breast Cancer
- Gastrointestinal Cancer
- Prostate Cancer
- Respiratory/Lung Cancer
- Others
Key Market players
Roche (Genentech), Novartis, Bristol Myers Squibb, Merck & Co. (MSD), Pfizer, AstraZeneca, Johnson & Johnson (Janssen), Eli Lilly, Amgen, GSK, Sanofi, Takeda, Bayer, AbbVie, Gilead Sciences, Daiichi Sankyo, Astellas Pharma, BeiGene, Eisai, SeagenCancer Drugs Market Analytics
The report employs rigorous tools, including Porter’s Five Forces, value chain mapping, and scenario-based modelling, to assess supply-demand dynamics. Cross-sector influences from parent, derived, and substitute markets are evaluated to identify risks and opportunities. Trade and pricing analytics provide an up-to-date view of international flows, including leading exporters, importers, and regional price trends.Macroeconomic indicators, policy frameworks such as carbon pricing and energy security strategies, and evolving consumer behaviour are considered in forecasting scenarios. Recent deal flows, partnerships, and technology innovations are incorporated to assess their impact on future market performance.
Cancer Drugs Market Competitive Intelligence
The competitive landscape is mapped through proprietary frameworks, profiling leading companies with details on business models, product portfolios, financial performance, and strategic initiatives. Key developments such as mergers & acquisitions, technology collaborations, investment inflows, and regional expansions are analyzed for their competitive impact. The report also identifies emerging players and innovative startups contributing to market disruption.Regional insights highlight the most promising investment destinations, regulatory landscapes, and evolving partnerships across energy and industrial corridors.
Countries Covered
- North America - Cancer Drugs market data and outlook to 2034
- United States
- Canada
- Mexico
- Europe - Cancer Drugs market data and outlook to 2034
- Germany
- United Kingdom
- France
- Italy
- Spain
- BeNeLux
- Russia
- Sweden
- Asia-Pacific - Cancer Drugs market data and outlook to 2034
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Malaysia
- Vietnam
- Middle East and Africa - Cancer Drugs market data and outlook to 2034
- Saudi Arabia
- South Africa
- Iran
- UAE
- Egypt
- South and Central America - Cancer Drugs market data and outlook to 2034
- Brazil
- Argentina
- Chile
- Peru
Research Methodology
This study combines primary inputs from industry experts across the Cancer Drugs value chain with secondary data from associations, government publications, trade databases, and company disclosures. Proprietary modeling techniques, including data triangulation, statistical correlation, and scenario planning, are applied to deliver reliable market sizing and forecasting.Key Questions Addressed
- What is the current and forecast market size of the Cancer Drugs industry at global, regional, and country levels?
- Which types, applications, and technologies present the highest growth potential?
- How are supply chains adapting to geopolitical and economic shocks?
- What role do policy frameworks, trade flows, and sustainability targets play in shaping demand?
- Who are the leading players, and how are their strategies evolving in the face of global uncertainty?
- Which regional “hotspots” and customer segments will outpace the market, and what go-to-market and partnership models best support entry and expansion?
- Where are the most investable opportunities-across technology roadmaps, sustainability-linked innovation, and M&A-and what is the best segment to invest over the next 3-5 years?
Your Key Takeaways from the Cancer Drugs Market Report
- Global Cancer Drugs market size and growth projections (CAGR), 2024-2034
- Impact of Russia-Ukraine, Israel-Palestine, and Hamas conflicts on Cancer Drugs trade, costs, and supply chains
- Cancer Drugs market size, share, and outlook across 5 regions and 27 countries, 2023-2034
- Cancer Drugs market size, CAGR, and market share of key products, applications, and end-user verticals, 2023-2034
- Short- and long-term Cancer Drugs market trends, drivers, restraints, and opportunities
- Porter’s Five Forces analysis, technological developments, and Cancer Drugs supply chain analysis
- Cancer Drugs trade analysis, Cancer Drugs market price analysis, and Cancer Drugs supply/demand dynamics
- Profiles of 5 leading companies-overview, key strategies, financials, and products
- Latest Cancer Drugs market news and developments
Additional Support
With the purchase of this report, you will receive:- An updated PDF report and an MS Excel data workbook containing all market tables and figures for easy analysis.
- 7-day post-sale analyst support for clarifications and in-scope supplementary data, ensuring the deliverable aligns precisely with your requirements.
- Complimentary report update to incorporate the latest available data and the impact of recent market developments.
This product will be delivered within 1-3 business days.
Table of Contents
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 160 |
| Published | February 2026 |
| Forecast Period | 2025 - 2034 |
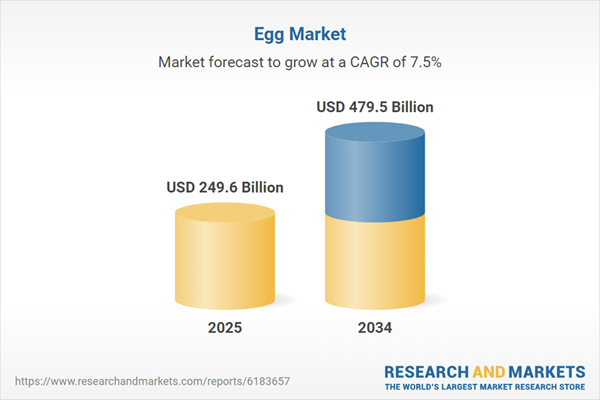
| Estimated Market Value ( USD | $ 249.6 Billion |
| Forecasted Market Value ( USD | $ 479.5 Billion |
| Compound Annual Growth Rate | 7.5% |
| Regions Covered | Global |
| No. of Companies Mentioned | 20 |