Breast Tissue Markers Market Key Insights
- Shift from wires to wire-free localization: Hospitals are transitioning toward RFID-, radar-, and magnetic-based breast tissue localization systems to reduce patient discomfort and procedural bottlenecks. Wire-free methods decouple marker placement from the surgical schedule, improving OR flexibility and reducing same-day workflow stress. The result is shorter localization times, fewer reschedules, and enhanced patient satisfaction.
- Multi-modality visibility is now a baseline spec: Modern markers must maintain visibility across mammography, ultrasound, and MRI while minimizing imaging artifacts. Innovations such as shape-coded identifiers and echogenic surface designs enable faster and more reliable localization. Consistent imaging visibility supports longitudinal tracking, particularly for patients undergoing neoadjuvant therapies.
- Migration resistance and tissue friendliness: Advanced anchoring features - textured coatings, hydrogel carriers, and bioinert alloys - help prevent marker migration and reduce inflammatory response. Low-profile, flexible designs minimize tissue distortion and improve comfort. Stable placement reduces re-localization and accelerates imaging workflows.
- Procedure ecosystems beat standalone SKUs: Hospitals increasingly prefer comprehensive system ecosystems including markers, applicators, detection probes, and sterile kits. Vendor differentiation depends on reader reliability, ergonomic probes, and responsive technical service. Integrated platform adoption enhances standardization and operational continuity across multi-site health systems.
- Neoadjuvant therapy expands durable-mark needs: Prolonged treatment cycles require markers that remain visible and stable over several months. Durable materials and secure anchoring ensure reliable localization even after fibrosis or edema. This durability supports precise margin identification and lowers re-excision rates post-therapy.
- Imaging advances raise the bar: New modalities such as tomosynthesis and contrast-enhanced mammography demand markers that maintain high visibility without causing beam distortion. MRI-compatible options increase patient inclusivity and diagnostic flexibility. Manufacturers must balance radiopacity with artifact minimization to meet imaging quality standards.
- Operating economics and throughput: Wire-free localization can streamline preoperative coordination and increase operating room utilization. Evaluations now emphasize total cost-in-use, including disposable pricing, equipment amortization, and efficiency gains. Outpatient and ASC facilities particularly value workflow speed and predictable scheduling.
- Regulatory and safety considerations: The decline of radioactive seed localization stems from handling restrictions and compliance complexity. Vendors emphasizing non-radioactive alternatives with clear IFUs, MRI-conditional labeling, and verified biocompatibility gain tender advantages. Safety documentation is central to procurement approval.
- Training and standardization as success levers: Successful adoption depends on cross-disciplinary training for radiologists, surgeons, and technologists. Standardized checklists and image-guided placement protocols ensure consistent outcomes. Structured onboarding and refresher modules shorten learning curves and strengthen confidence in wire-free systems.
- Data capture and interoperability: Next-generation systems integrate with hospital IT infrastructure to log placement data, signal IDs, and audit trails. Seamless PACS/EMR connectivity enhances traceability and quality assurance. These data linkages support evidence-based improvements in localization accuracy and surgical efficiency.
Breast Tissue Markers Market Reginal Analysis
North America
Mature screening infrastructure and tomosynthesis penetration drive large biopsy volumes and high adoption of wire-free platforms. Health systems prioritize OR efficiency, same-day discharge pathways, and ASC suitability. Vendors win with comprehensive service models, MRI-conditional labeling, and proven performance in dense breast cohorts. Integrated training across multi-hospital networks is a key differentiator.
Europe
Strong guideline adherence and centralized breast units favor standardized protocols and evidence-backed platforms. Radiation safety rules accelerate the shift away from radio-seeds. Procurement emphasizes MRI compatibility, minimal artifact, and traceable supply chains. Value assessments consider long-term outcomes such as re-excision avoidance and patient-reported comfort.
Asia-Pacific
Screening expansion and investments in imaging capacity increase biopsy volumes, with varied adoption speeds between metros and secondary cities. Cost-sensitive buyers favor durable markers visible across modalities with straightforward deployment. Training partnerships and local distributor support are decisive. Wire-free systems gain traction in tertiary centers, then diffuse to regional hospitals.
Middle East & Africa
Urban centers invest in advanced imaging and breast specialty programs, adopting wire-free localization to streamline surgical scheduling. Hot climates and logistics constraints heighten the value of robust supply and reliable device performance. Private hospitals and regional centers seek turnkey kits with strong vendor training and rapid service response.
South & Central America
Public-private mixes create diverse purchasing criteria; large urban hospitals lead adoption of multi-modality markers. Reliability and ease of use are prioritized to offset staffing variability. Vendors with localized inventory, bilingual training materials, and flexible financing gain share. Gradual protocol standardization supports broader uptake beyond flagship institutions.
Breast Tissue Markers Market Segmentation
By Product- Coil
- Ribbon
- Wing
- U Shaped
- Conic
- Ring
- Heart
- Venus
- Others
- Bio-absorbable
- Non-absorbable
- Biopsy
- Stereotactic
- Ultrasound
- MRI-guided
- Surgical Planning
- Others
- Hospitals
- Ambulatory Surgical Centers
- Diagnostic Centers
- Specialty Clinics
Key Market players
Hologic, Inc., Mammotome (Leica Biosystems), BD (Becton, Dickinson and Company), Cook Medical, Merit Medical Systems, Inc., Endomag (Endomagnetics Ltd.), Elucent Medical, Sirius Medical, IZI Medical Products, Argon Medical Devices, Inc., SOMATEX Medical Technologies (a Hologic company), STERYLAB Srl, Biopsybell Srl, BPB Medica, Theragenics Corporation.Breast Tissue Markers Market Analytics
The report employs rigorous tools, including Porter’s Five Forces, value chain mapping, and scenario-based modelling, to assess supply-demand dynamics. Cross-sector influences from parent, derived, and substitute markets are evaluated to identify risks and opportunities. Trade and pricing analytics provide an up-to-date view of international flows, including leading exporters, importers, and regional price trends.Macroeconomic indicators, policy frameworks such as carbon pricing and energy security strategies, and evolving consumer behaviour are considered in forecasting scenarios. Recent deal flows, partnerships, and technology innovations are incorporated to assess their impact on future market performance.
Breast Tissue Markers Market Competitive Intelligence
The competitive landscape is mapped through proprietary frameworks, profiling leading companies with details on business models, product portfolios, financial performance, and strategic initiatives. Key developments such as mergers & acquisitions, technology collaborations, investment inflows, and regional expansions are analyzed for their competitive impact. The report also identifies emerging players and innovative startups contributing to market disruption.Regional insights highlight the most promising investment destinations, regulatory landscapes, and evolving partnerships across energy and industrial corridors.
Countries Covered
- North America - Breast Tissue Markers market data and outlook to 2034
- United States
- Canada
- Mexico
- Europe - Breast Tissue Markers market data and outlook to 2034
- Germany
- United Kingdom
- France
- Italy
- Spain
- BeNeLux
- Russia
- Sweden
- Asia-Pacific - Breast Tissue Markers market data and outlook to 2034
- China
- Japan
- India
- South Korea
- Australia
- Indonesia
- Malaysia
- Vietnam
- Middle East and Africa - Breast Tissue Markers market data and outlook to 2034
- Saudi Arabia
- South Africa
- Iran
- UAE
- Egypt
- South and Central America - Breast Tissue Markers market data and outlook to 2034
- Brazil
- Argentina
- Chile
- Peru
Research Methodology
This study combines primary inputs from industry experts across the Breast Tissue Markers value chain with secondary data from associations, government publications, trade databases, and company disclosures. Proprietary modeling techniques, including data triangulation, statistical correlation, and scenario planning, are applied to deliver reliable market sizing and forecasting.Key Questions Addressed
- What is the current and forecast market size of the Breast Tissue Markers industry at global, regional, and country levels?
- Which types, applications, and technologies present the highest growth potential?
- How are supply chains adapting to geopolitical and economic shocks?
- What role do policy frameworks, trade flows, and sustainability targets play in shaping demand?
- Who are the leading players, and how are their strategies evolving in the face of global uncertainty?
- Which regional “hotspots” and customer segments will outpace the market, and what go-to-market and partnership models best support entry and expansion?
- Where are the most investable opportunities-across technology roadmaps, sustainability-linked innovation, and M&A-and what is the best segment to invest over the next 3-5 years?
Your Key Takeaways from the Breast Tissue Markers Market Report
- Global Breast Tissue Markers market size and growth projections (CAGR), 2024-2034
- Impact of Russia-Ukraine, Israel-Palestine, and Hamas conflicts on Breast Tissue Markers trade, costs, and supply chains
- Breast Tissue Markers market size, share, and outlook across 5 regions and 27 countries, 2023-2034
- Breast Tissue Markers market size, CAGR, and market share of key products, applications, and end-user verticals, 2023-2034
- Short- and long-term Breast Tissue Markers market trends, drivers, restraints, and opportunities
- Porter’s Five Forces analysis, technological developments, and Breast Tissue Markers supply chain analysis
- Breast Tissue Markers trade analysis, Breast Tissue Markers market price analysis, and Breast Tissue Markers supply/demand dynamics
- Profiles of 5 leading companies-overview, key strategies, financials, and products
- Latest Breast Tissue Markers market news and developments
Additional Support
With the purchase of this report, you will receive:- An updated PDF report and an MS Excel data workbook containing all market tables and figures for easy analysis.
- 7-day post-sale analyst support for clarifications and in-scope supplementary data, ensuring the deliverable aligns precisely with your requirements.
- Complimentary report update to incorporate the latest available data and the impact of recent market developments.
This product will be delivered within 1-3 business days.
Table of Contents
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 160 |
| Published | February 2026 |
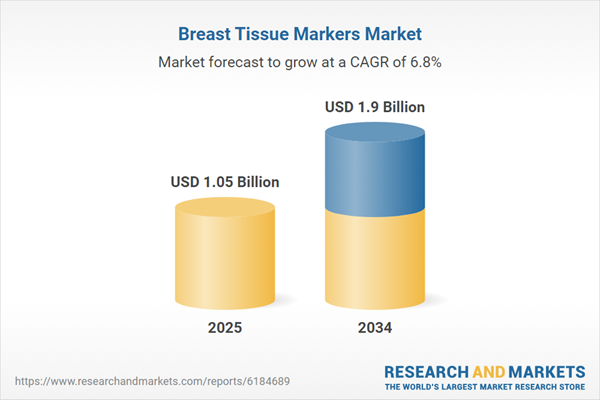
| Forecast Period | 2025 - 2034 |
| Estimated Market Value ( USD | $ 1.05 Billion |
| Forecasted Market Value ( USD | $ 1.9 Billion |
| Compound Annual Growth Rate | 6.8% |
| Regions Covered | Global |
| No. of Companies Mentioned | 15 |