United States Fluorescence In Situ Hybridization Market
FISH is a cornerstone ISH technique in the U.S., valued for its sensitivity, spatial resolution, and clinical utility in oncology and genetic diagnostics. This technique allows for the direct detection of chromosomal rearrangements, gene amplifications, and specific RNA targets within the tissue architecture, thus guiding targeted therapies and risk stratification. Multiplex FISH chemistries and improved fluorophores have increased assay throughput and allow complex karyotypic analyses. Clinical reliance on FISH for hematologic malignancies, breast cancer HER2 testing, ALK/ROS1 rearrangements, and other actionable targets drives steady demand. FISH is often performed as a reflex or confirmatory test in academic centers and commercial laboratories when the sequencing or IHC results are ambiguous.United States Chromogenic In Situ Hybridization Market
CISH uses nucleic acid detection combined with brightfield histology, yielding colorimetric signals observable on standard microscopes-a feature advantageous for most pathology laboratories. CISH offers permanent, durable staining that is easily integrated with regular H&E review, allowing the pathologist to correlate molecular signals with morphology without specialized fluorescence equipment. Ease of interpretation without specialized equipment has made CISH an attractive alternative, especially in community hospitals and labs with limited resources for fluorescence imaging. In diagnostic workflows where long-term archiving and ease of interpretation are highly valued, such as HER2 amplification testing, CISH remains a valuable complement or alternative to FISH. Whereas multiplexing is more difficult than in fluorescence methods, recent advances in chromogenic chemistries and signal amplification have improved sensitivity and expanded applications.United States Infectious Diseases In Situ Hybridization Market
ISH applications in infectious disease diagnostics allow the direct visualization of pathogen nucleic acids within tissue context, aiding in the identification of viral, bacterial, fungal, and parasitic infections - especially in those cases when culture or PCR yields limited information. ISH has been used to diagnose tissue-invasive infections, to confirm etiologies in biopsy specimens, and to provide information on pathogen tropism. In transplant medicine, neuropathology, and histopathologic evaluation of granulomatous disease, ISH provides critical localization data. During outbreak investigations and research, ISH can map viral distribution and tissue reservoirs, informing pathogenesis studies. Laboratories leverage ISH for organisms that are difficult to culture or require spatial resolution to confirm disease association. This method is complementary to molecular PCR testing because it correlates the presence of pathogen RNA/DNA with histologic changes, thereby enhancing diagnostic specificity.United States Genetic & Rare Disorders In Situ Hybridization Market
ISH has an important role in the diagnosis of genetic conditions and rare diseases where the localization of gene expression or chromosomal aberrations in tissues is informative. For conditions caused by mosaicism, focal developmental anomalies, or organ-specific changes in gene expression, ISH offers a spatially resolved complement to genomic sequencing. In the prenatal pathology and neonatal diagnostic setting, ISH identifies structural chromosomal anomalies and gene amplifications within tissue sections. In the study of rare diseases, ISH maps aberrant expression patterns in affected tissues to enable mechanistic study and biomarker discovery. Reference laboratories frequently use ISH during confirmatory testing when karyotype or sequencing results require anatomic correlation. As interest in precision diagnostics for rare conditions increases and as therapeutic development targets tissue-specific mechanisms, ISH continues to serve as a valuable tool for clinical assessment and study in the U.S. rare disease community.United States In Situ Hybridization Diagnostic Laboratories Market
These include large reference labs, hospital-based molecular pathology departments, and specialized academic centers. Centralized labs offer validated panels, multiplex assays, and high-throughput workflows that smaller hospitals often send out because of the cost and complexity of setting up such operations. Reference laboratories will invest in automation, quality management, and the diversification of assays, thus offering rapid turnaround for oncology biomarkers, infectious agents, and research studies. Hospital laboratories with in-house ISH capabilities will continue to support immediate clinical needs, surgical pathology correlation, and urgent oncology cases. Partnerships between diagnostics providers and therapeutic developers will also create testing-as-a-service models for clinical trials and companion diagnostics. Overall, quality assurance programs, proficiency testing, and adherence to regulations are major differentiators in diagnostic labs. With the demand for tissue-based molecular testing continuing to grow, diagnostic laboratories that offer a combination of validated assays, expert interpretation, and robust logistics will lead the U.S. ISH service market.California In Situ Hybridization Market
California's ISH market benefits from its dense network of academic institutions, biotech companies, and leading oncology centers. Research-intensive universities and translational labs often use ISH for developmental biology, neuroscience, and cancer research, thus creating strong demand for probes, instruments, and specialized services. Biotech and pharmaceutical firms in the state incorporate ISH in preclinical studies and biomarker development for drug candidates. Large cancer centers and hospital systems employ ISH for diagnostic assays and clinical trial endpoints, thus fueling investment in automation and digital pathology. The state's emphasis on innovation and early adoption of advanced molecular diagnostics undergirds growth in both clinical and research ISH segments. Moreover, tech companies and life-science labs are collaborating in teleradiology and clinical diagnostics to accelerate software-driven image analysis and AI applications in ISH interpretation, further enhancing California's leadership.New York In Situ Hybridization Market
New York is home to many of the largest medical centers, research hospitals, and clinical laboratories that drive the utilization of ISH in oncology, infectious disease pathology, and translational studies. Academic medical centers tend to emphasize novel applications of ISH for challenging case work and clinical trials, while urban hospital networks support a core role for ISH in confirmatory diagnosis and therapy selection. The dense healthcare landscape and large patient volumes in the state support the availability of high-order ISH services, such as multiplex assays and rapid reflex testing. Reference labs based in the region provide send-out services to smaller hospitals throughout the Northeast. In addition, New York's strong clinical research environment facilitates the integration of ISH study endpoints into trial design, resulting in significant demand for validated assays and harmonized reporting. Taken together, complexity of practice, large caseloads, and research activity ensure consistent levels of ISH market activity within the state.Washington In Situ Hybridization Market
The growing biotech industry, research institutions, and specific clinical centers throughout Washington State support the state's ISH market. Smaller in size compared to California or New York, the State of Washington's numerous biotech companies and medical research institutions fuel demand for ISH in translational projects and diagnostic development. Regional hospitals and academic laboratories apply ISH in targeted oncology testing and infectious disease diagnosis. Collaboration between research groups and technology companies advances the development of imaging and analysis tools that improve ISH workflows. Innovation and interdepartmental collaboration are highlighted within the state, where ISH services grow selectively to support applications in which tissue-based molecular insight is complementary to regional research priorities.Arizona In Situ Hybridization Market
Arizona's ISH market is a segment with continuous growth, as its healthcare infrastructure, oncology services, and regional diagnostic laboratories expand. Concomitantly, as molecular pathology testing capacities increase at various hospitals and cancer treatment centers, ISH is increasingly indicated for confirmatory diagnostics and biomarker testing. Community hospitals frequently rely on send-out arrangements with large regional reference laboratories for sophisticated ISH assays, hence creating demand for centralized services. The state's growing population and investment in medical facilities continue to favor further adoption of ISH for the diagnosis of infectious diseases, tissue-based genetic testing, and clinical trial participation. Training programs and telepathology collaborations extend specialized interpretive expertise to more rural and suburban areas, enabling access to ISH diagnostics across wider swaths of Arizona.Market Segmentations
Product
- Analytical Instruments
- Probes, Kits & Reagents
- Software & Services
- Other Products
Technique
- Fluorescence ISH (FISH)
- Chromogenic ISH (CISH)
- Amplified RNA-ISH (HCR, RNAscope)
- In-situ Sequencing (ISS)
Application
- Cancer Diagnostics & Research
- Infectious Diseases
- Genetic & Rare Disorders
- Neurological & Developmental Biology
- Other Applications
End User
- Diagnostic Laboratories
- Academic & Research Institutes
- Pharma-Biotech & CROs
- Veterinary & Environmental Labs
Top States
- California
- Texas
- New York
- Florida
- Illinois
- Pennsylvania
- Ohio
- Georgia
- New Jersey
- Washington
- North Carolina
- Massachusetts
- Virginia
- Michigan
- Maryland
- Colorado
- Tennessee
- Indiana
- Arizona
- Minnesota
- Wisconsin
- Missouri
- Connecticut
- South Carolina
- Oregon
- Louisiana
- Alabama
- Kentucky
- Rest of United States
All companies have been covered with 5 Viewpoints
- Overviews
- Key Person
- Recent Developments
- SWOT Analysis
- Revenue Analysis
Company Analysis:
- PerkinElmer, Inc.
- Thermo Fisher Scientific, Inc.
- BioView
- Agilent Technologies, Inc.
- Merck KGaA
- Bio-Rad Laboratories, Inc.
- Oxford Gene Technology IP Limited
- Neogenomics Laboratories, Inc.
- Advanced Cell Diagnostics, Inc.
Table of Contents
Companies Mentioned
The companies featured in this United States In Situ Hybridization market report include:- PerkinElmer, Inc.
- Thermo Fisher Scientific, Inc.
- BioView
- Agilent Technologies, Inc.
- Merck KGaA
- Bio-Rad Laboratories, Inc.
- Oxford Gene Technology IP Limited
- Neogenomics Laboratories, Inc.
- Advanced Cell Diagnostics, Inc.
Methodology
In this report, for analyzing the future trends for the studied market during the forecast period, the publisher has incorporated rigorous statistical and econometric methods, further scrutinized by secondary, primary sources and by in-house experts, supported through their extensive data intelligence repository. The market is studied holistically from both demand and supply-side perspectives. This is carried out to analyze both end-user and producer behavior patterns, in the review period, which affects price, demand and consumption trends. As the study demands to analyze the long-term nature of the market, the identification of factors influencing the market is based on the fundamentality of the study market.
Through secondary and primary researches, which largely include interviews with industry participants, reliable statistics, and regional intelligence, are identified and are transformed to quantitative data through data extraction, and further applied for inferential purposes. The publisher's in-house industry experts play an instrumental role in designing analytic tools and models, tailored to the requirements of a particular industry segment. These analytical tools and models sanitize the data & statistics and enhance the accuracy of their recommendations and advice.
Primary Research
The primary purpose of this phase is to extract qualitative information regarding the market from the key industry leaders. The primary research efforts include reaching out to participants through mail, tele-conversations, referrals, professional networks, and face-to-face interactions. The publisher also established professional corporate relations with various companies that allow us greater flexibility for reaching out to industry participants and commentators for interviews and discussions, fulfilling the following functions:
- Validates and improves the data quality and strengthens research proceeds
- Further develop the analyst team’s market understanding and expertise
- Supplies authentic information about market size, share, growth, and forecast
The researcher's primary research interview and discussion panels are typically composed of the most experienced industry members. These participants include, however, are not limited to:
- Chief executives and VPs of leading corporations specific to the industry
- Product and sales managers or country heads; channel partners and top level distributors; banking, investment, and valuation experts
- Key opinion leaders (KOLs)
Secondary Research
The publisher refers to a broad array of industry sources for their secondary research, which typically includes, however, is not limited to:
- Company SEC filings, annual reports, company websites, broker & financial reports, and investor presentations for competitive scenario and shape of the industry
- Patent and regulatory databases for understanding of technical & legal developments
- Scientific and technical writings for product information and related preemptions
- Regional government and statistical databases for macro analysis
- Authentic new articles, webcasts, and other related releases for market evaluation
- Internal and external proprietary databases, key market indicators, and relevant press releases for market estimates and forecasts

LOADING...
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 200 |
| Published | February 2026 |
| Forecast Period | 2025 - 2034 |
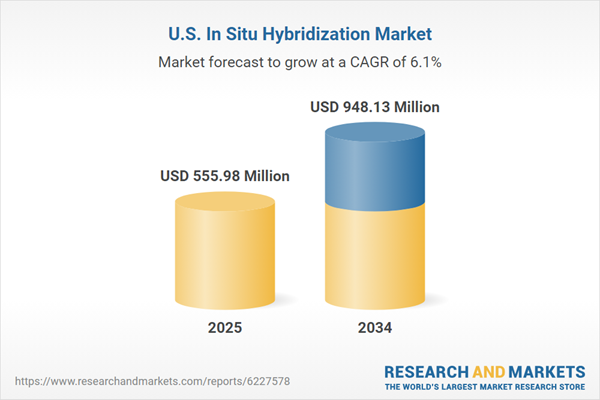
| Estimated Market Value ( USD | $ 555.98 Million |
| Forecasted Market Value ( USD | $ 948.13 Million |
| Compound Annual Growth Rate | 6.1% |
| Regions Covered | United States |
| No. of Companies Mentioned | 10 |