Global Artificial Intelligence (AI) for Drug Development and Discovery Market - Key Trends & Drivers Summarized
How Is Computational Intelligence Replacing Trial And Error In Molecular Discovery?
Artificial intelligence has altered the earliest phase of pharmaceutical research by shifting compound identification from laboratory screening toward predictive molecular design. Traditional discovery depended on testing thousands of compounds experimentally, but learning models now predict biological activity based on structural and physicochemical properties before synthesis occurs. Algorithms evaluate binding affinity between proteins and candidate molecules using structural representations derived from crystallography and cryo electron microscopy datasets. Instead of searching randomly across chemical libraries, researchers generate optimized compounds digitally and select only the most promising candidates for laboratory validation. Chemical space exploration expands dramatically because models evaluate millions of theoretical structures within practical timeframes. Generative molecular models propose new scaffolds that satisfy pharmacological constraints such as solubility and toxicity thresholds. Biological pathway mapping platforms analyze genomic and proteomic datasets to identify disease mechanisms and reveal previously unknown therapeutic targets. The discovery workflow becomes data guided where hypotheses are ranked by predicted efficacy probability. Pharmaceutical organizations increasingly integrate laboratory automation with computational prediction so experimental validation feeds back into model refinement. This iterative loop shortens the time required to reach a viable lead compound. Researchers therefore move from broad screening campaigns toward focused validation strategies guided by computational reasoning. The discovery stage evolves into a predictive design discipline grounded in biological data interpretation.Can Machine Learning Predict Clinical Success Before Human Trials Begin?
Predictive analytics extends beyond molecular identification into assessment of pharmacokinetics, toxicity and therapeutic response likelihood. Models analyze historical clinical trial datasets to estimate probability of success for candidate therapies in specific patient populations. Simulation platforms replicate biological interactions across organs to evaluate absorption, distribution, metabolism and excretion behavior before clinical exposure. Patient stratification algorithms identify biomarkers associated with treatment responsiveness enabling targeted trial recruitment strategies. Safety assessment tools detect off target interactions by comparing molecular patterns against databases of adverse events. Clinical protocol optimization systems recommend dosage ranges and cohort structures to maximize statistical significance while minimizing participant numbers. These capabilities reduce costly late stage failures which historically represented a major burden for pharmaceutical development. Regulatory documentation preparation is assisted by automated analysis of experimental outputs ensuring traceable evidence generation. Continuous learning systems update predictions as new experimental results appear creating dynamic risk evaluation across the development pipeline. The clinical planning stage becomes an informed forecasting process supported by quantitative probabilities rather than empirical estimation. Organizations increasingly treat predictive modeling as a gatekeeper determining which compounds advance to trials.How Are Pharmaceutical Operations Integrating AI Across The Entire R And D Pipeline?
Drug development workflows now connect discovery models, laboratory robotics, clinical analytics and manufacturing optimization within unified data environments. Laboratory instruments automatically upload assay results into centralized platforms where models refine predictions in near real time. Digital twins of biological processes simulate production yield and stability conditions during formulation design. Manufacturing analytics monitor batch variability and predict deviations before they occur ensuring consistent therapeutic quality. Post market surveillance systems analyze patient outcomes and pharmacovigilance reports to detect long term safety patterns and guide label adjustments. Knowledge graphs link scientific publications, patents and experimental datasets to support hypothesis generation by researchers. Collaboration between biotechnology firms and technology providers results in specialized platforms dedicated to antibody engineering, RNA therapies and gene editing applications. Cloud based infrastructure enables secure sharing of research data across geographically distributed teams. Pharmaceutical companies establish dedicated computational biology divisions responsible for maintaining model performance and dataset governance. The research environment transitions into a continuously connected ecosystem where insights propagate rapidly between stages instead of isolated handoffs.What Forces Are Accelerating Adoption Of AI Driven Pharmaceutical Innovation?
The growth in the Artificial Intelligence for drug development and discovery market is driven by several factors including rising availability of genomic sequencing data enabling precise target identification, increasing complexity of biologic therapies requiring computational design assistance, and high failure rates in late stage trials encouraging predictive screening before clinical investment. Adoption is further supported by expansion of rare disease research where limited patient populations demand efficient candidate selection, integration of biomarker guided personalized medicine strategies requiring analytical modeling, and demand for rapid response platforms capable of designing therapies against emerging pathogens. Growth also stems from pharmaceutical partnerships with technology companies providing specialized modeling infrastructure, regulatory encouragement for evidence supported development processes, and use of real world patient data to refine therapeutic indications after approval. The need to optimize manufacturing consistency for advanced therapies such as cell and gene treatments promotes analytical monitoring across production lines. Increasing competitive pressure to shorten development timelines motivates organizations to adopt computational prioritization methods. Together these end use and workflow specific drivers sustain widespread implementation of intelligent platforms across pharmaceutical research and commercialization stages.Report Scope
The report analyzes the AI for Drug Development and Discovery market, presented in terms of market value (US$). The analysis covers the key segments and geographic regions outlined below:- Segments: Type (Target Identification Type, Molecule Screening Type, De Novo Drug Design & Drug Optimization Type, Preclinical & Clinical Testing Type); Indication (Oncology Indication, Infectious Disease Indication, Neurology Indication, Other Indications); End-Use (Pharma & Biotech Companies End-Use, Contract Research Organization End-Use)
- Geographic Regions/Countries: World; USA; Canada; Japan; China; Europe; France; Germany; Italy; UK; Rest of Europe; Asia-Pacific; Rest of World.
Key Insights:
- Market Growth: Understand the significant growth trajectory of the Target Identification Type segment, which is expected to reach US$9.3 Billion by 2032 with a CAGR of a 35.5%. The Molecule Screening Type segment is also set to grow at 26.9% CAGR over the analysis period.
- Regional Analysis: Gain insights into the U.S. market, valued at $790.6 Million in 2025, and China, forecasted to grow at an impressive 29.9% CAGR to reach $2.9 Billion by 2032. Discover growth trends in other key regions, including Japan, Canada, Germany, and the Asia-Pacific.
Why You Should Buy This Report:
- Detailed Market Analysis: Access a thorough analysis of the Global AI for Drug Development and Discovery Market, covering all major geographic regions and market segments.
- Competitive Insights: Get an overview of the competitive landscape, including the market presence of major players across different geographies.
- Future Trends and Drivers: Understand the key trends and drivers shaping the future of the Global AI for Drug Development and Discovery Market.
- Actionable Insights: Benefit from actionable insights that can help you identify new revenue opportunities and make strategic business decisions.
Key Questions Answered:
- How is the Global AI for Drug Development and Discovery Market expected to evolve by 2032?
- What are the main drivers and restraints affecting the market?
- Which market segments will grow the most over the forecast period?
- How will market shares for different regions and segments change by 2032?
- Who are the leading players in the market, and what are their prospects?
Report Features:
- Comprehensive Market Data: Independent analysis of annual sales and market forecasts in US$ Million from 2025 to 2032.
- In-Depth Regional Analysis: Detailed insights into key markets, including the U.S., China, Japan, Canada, Europe, Asia-Pacific, Latin America, Middle East, and Africa.
- Company Profiles: Coverage of players such as Alphabet, Inc., BenevolentAI, Biomap, BioSymetrics, Cloud Pharmaceuticals, Inc. and more.
- Complimentary Updates: Receive free report updates for one year to keep you informed of the latest market developments.
Some of the companies featured in this AI for Drug Development and Discovery market report include:
- Alphabet, Inc.
- BenevolentAI
- Biomap
- BioSymetrics
- Cloud Pharmaceuticals, Inc.
- Deep Genomics
- Euretos Services BV
- IBM Corporation
- Iktos SA
- Insilico Medicine
Domain Expert Insights
This market report incorporates insights from domain experts across enterprise, industry, academia, and government sectors. These insights are consolidated from multilingual multimedia sources, including text, voice, and image-based content, to provide comprehensive market intelligence and strategic perspectives. As part of this research study, the publisher tracks and analyzes insights from 43 domain experts. Clients may request access to the network of experts monitored for this report, along with the online expert insights tracker.Companies Mentioned (Partial List)
A selection of companies mentioned in this report includes, but is not limited to:
- Alphabet, Inc.
- BenevolentAI
- Biomap
- BioSymetrics
- Cloud Pharmaceuticals, Inc.
- Deep Genomics
- Euretos Services BV
- IBM Corporation
- Iktos SA
- Insilico Medicine
Table Information
| Report Attribute | Details |
|---|---|
| No. of Pages | 182 |
| Published | May 2026 |
| Forecast Period | 2025 - 2032 |
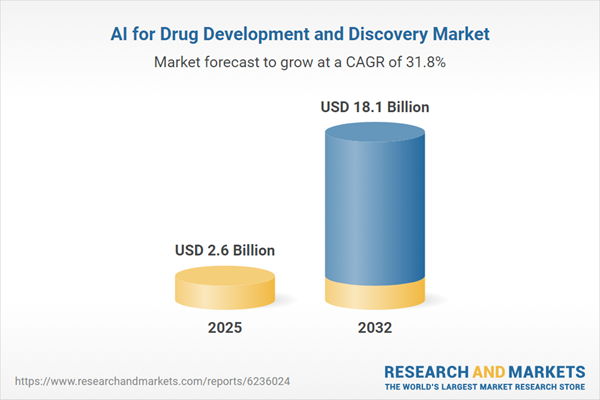
| Estimated Market Value ( USD | $ 2.6 Billion |
| Forecasted Market Value ( USD | $ 18.1 Billion |
| Compound Annual Growth Rate | 31.8% |
| Regions Covered | Global |